AP Syllabus focus: ‘If the forward rate is greater than the reverse rate, there is net conversion of reactants to products; if the reverse rate is greater, products convert back to reactants.’
Chemical reactions that can proceed in both directions are understood by comparing forward and reverse reaction rates at a given moment. This rate comparison predicts whether reactants are being consumed overall or regenerated overall.
Core idea: net direction comes from comparing rates
In a reversible reaction, reactants form products (forward process) while products can simultaneously re-form reactants (reverse process). The system’s net direction at any instant is determined only by which rate is larger.
If the forward rate is greater than the reverse rate, there is net conversion of reactants to products.
If the reverse rate is greater than the forward rate, there is net conversion of products to reactants.
If the rates are equal, there is no net change in the amounts of reactants and products (even though both processes can still be occurring).
This is a moment-by-moment comparison; as amounts change, the rates typically change, and the net direction can eventually switch.
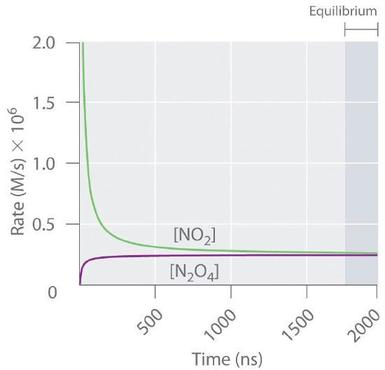
Forward and reverse reaction rates are plotted as functions of time for a reversible system. The net direction at any instant is determined by which curve is higher: implies net product formation, and the point where the curves become equal corresponds to zero net rate (dynamic equilibrium). Source
Reaction rate language you must use precisely
Reaction rate: the change in concentration (or partial pressure for gases) of a species per unit time, typically expressed as (or ).
Because rates track how fast concentrations are changing, the sign of the net change depends on whether product-forming events outnumber reactant-forming events per unit time.
Net rate as a compact way to express direction
A helpful way to formalize “which way is it going overall?” is to define a net rate.
= forward reaction rate (units of concentration/time)
= reverse reaction rate (units of concentration/time)
A positive net rate means the reaction mixture is changing in the forward direction overall; a negative net rate means it is changing in the reverse direction overall. A net rate of zero corresponds to no overall composition change.
What makes one direction faster than the other?
Forward and reverse rates depend on how frequently effective molecular encounters occur (and, for more advanced treatments, on the pathway and activation energy).
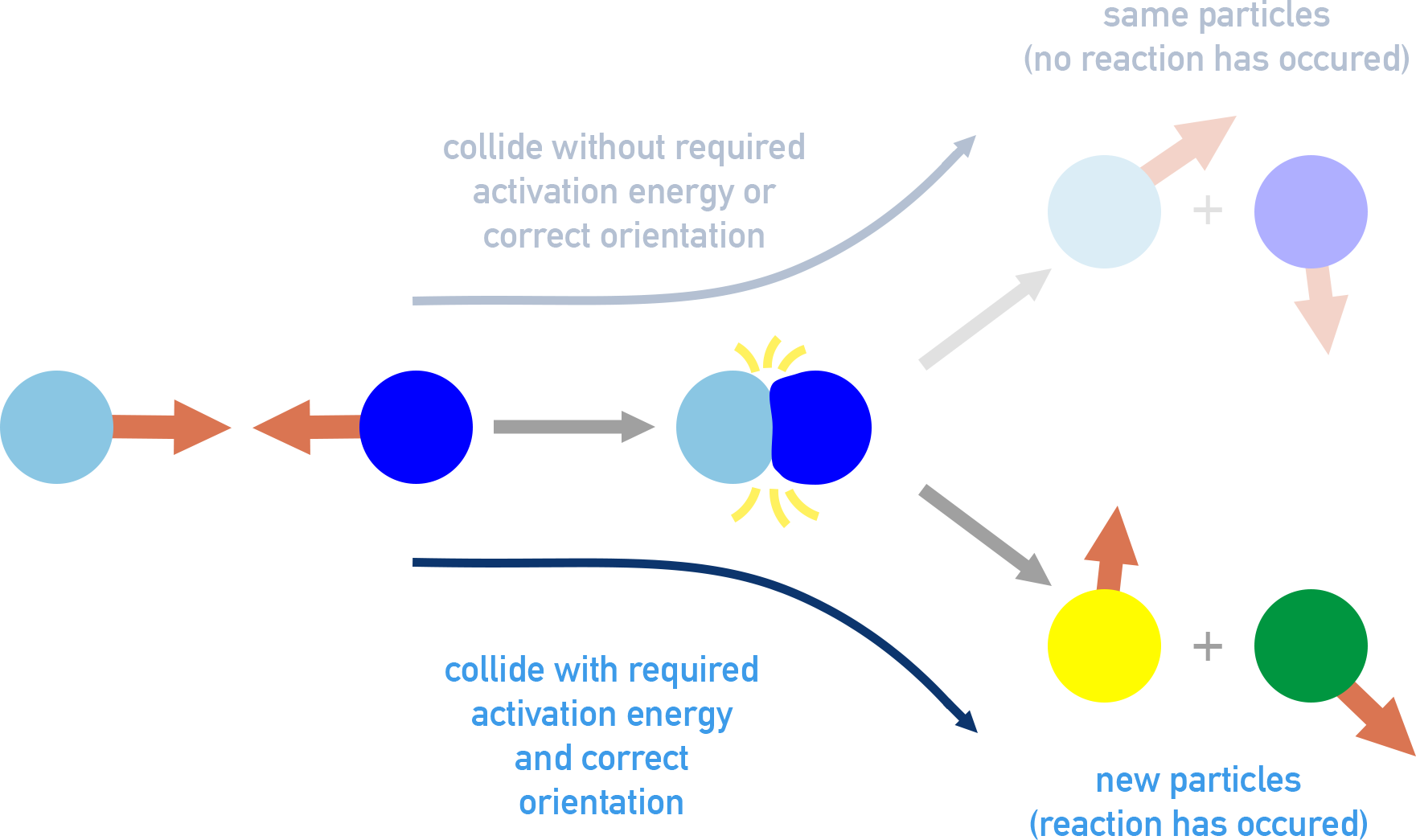
The diagram contrasts collisions that do and do not lead to reaction, emphasizing that correct orientation is required for an effective collision. This provides a molecular-level interpretation of reaction rate as “successful reactive events per unit time,” not merely the number of collisions. It also reinforces why changing conditions can alter rates without changing the overall balanced equation. Source
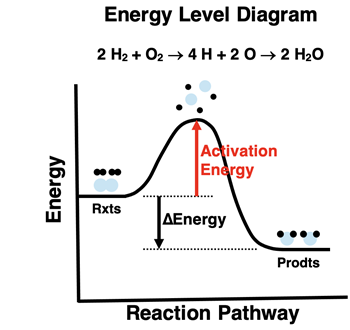
Potential energy is shown along the reaction coordinate, with the activation energy barrier labeled between reactants and products. Collisions must supply at least this activation energy to access the transition state, so the barrier height helps explain why some directions (or conditions) correspond to faster rates. The energy difference between reactants and products is also indicated to connect kinetics diagrams to thermodynamic change. Source
For AP-level reasoning, focus on these practical ideas:
Relative amounts present: More reactant particles generally increases the chance of reactant-to-product events, raising the forward rate; more product particles generally raises the reverse rate.
Starting conditions matter: If you begin with mostly reactants and little product, the forward rate tends to dominate initially. If you begin with mostly products, the reverse rate tends to dominate.
Rates change as the reaction proceeds: As reactants are consumed, the forward rate often decreases; as products accumulate, the reverse rate often increases. The net direction may therefore weaken over time.
Interpreting “net conversion” in words
When you state the net direction, tie it to observable composition change:
Net forward: reactant concentrations decrease overall while product concentrations increase overall.
Net reverse: product concentrations decrease overall while reactant concentrations increase overall.
Be careful: “net forward” does not mean the reverse reaction stops; it means the forward process is occurring faster than the reverse process at that moment.
Common pitfalls when predicting net direction from rates
Confusing equal concentrations with equal rates: Equal amounts of reactant and product do not guarantee equal rates; rate depends on how the system is behaving at that moment.
Assuming the faster direction stays faster forever: Because rates typically depend on current composition, the larger rate can change as the reaction mixture changes.
Mixing up direction with equilibrium language: For this skill, focus strictly on comparing and to decide net direction, rather than invoking other equilibrium tools.
FAQ
Chemists can use tracer methods (for example, isotopic labelling) to distinguish product molecules formed via the forward process from those formed via the reverse process.
They may also use rapid spectroscopy to track formation and consumption pathways on short timescales.
A catalyst speeds up both forward and reverse processes by lowering activation barriers for each direction.
At the same instant after adding a catalyst, the larger rate may remain larger, but the system approaches the point where rates match more quickly.
Because the composition changes as the reaction proceeds, and rates typically depend on current amounts.
As reactants drop and products rise, $r_\text{f}$ can fall while $r_\text{r}$ rises until their ordering changes.
Yes, for an intermediate or for a species involved in multiple simultaneous processes, production and consumption can balance.
This is a steady-state idea and is different from the overall reversible reaction’s forward/reverse rate comparison.
The “reaction rate” can be defined consistently using stoichiometry (e.g., normalising by coefficients) so one rate describes the progress of reaction.
Species-specific concentration changes relate by factors: $|d[A]/dt| = 2,|d[B]/dt|$ for the same extent change.
Practice Questions
(2 marks) For a reversible reaction at a particular moment, the forward rate is and the reverse rate is . State the net direction and whether the amounts of reactants and products are changing overall.
1 mark: Correct net direction (net forward; reactants to products).
1 mark: States that amounts are changing overall (reactants decreasing overall and/or products increasing overall), so not steady at that moment.
(5 marks) Consider . At time , extra is injected into the container. Immediately after , predict and explain what happens to: (a) the forward rate, (b) the reverse rate, and (c) the net direction of change.
1 mark: Forward rate increases immediately.
1 mark: Reverse rate is initially unchanged immediately after injection (before any new forms).
1 mark: Net direction becomes forward (net ).
1 mark: Explanation linking forward rate increase to increased amount/concentration of (more effective collisions).
1 mark: Explanation that reverse rate depends on amount, which has not yet increased at the instant after .

