AP Syllabus focus: ‘An equilibrium state is reached when the forward and reverse reaction rates become equal, producing no net change in the amounts of reactants and products.’
Chemical reactions that can proceed in both directions may settle into an equilibrium state. This page explains exactly when equilibrium is established, what “equal rates” means, and how that creates constant amounts of reactants and products.
What it means for equilibrium to be established
Equilibrium is defined by rates, not by “stopping”
When equilibrium is established, the reaction has not stopped.
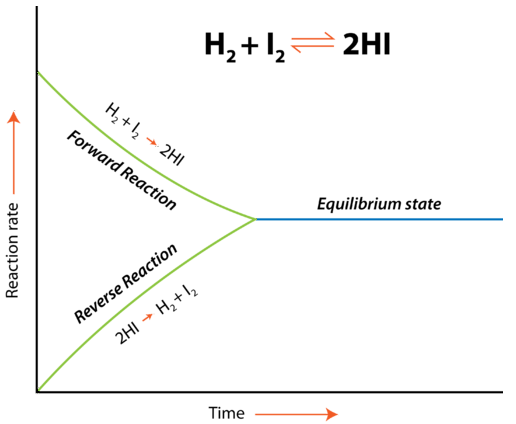
Reaction-rate diagram for showing forward and reverse rates converging and then remaining equal at the labeled equilibrium state. The flat, shared rate after convergence emphasizes that equilibrium is dynamic: both directions still occur, but at matched rates so there is no net compositional change. Source
Instead, two opposite processes occur simultaneously at the same speed.
Equilibrium state: A condition in a reversible reaction where the forward and reverse reaction rates are equal, so the amounts of reactants and products show no net change over time.
This definition matches the syllabus focus: equal rates lead directly to no net change in amounts.
Forward and reverse rates become equal
In a reversible reaction, reactants form products (forward direction) while products can also re-form reactants (reverse direction).
Forward reaction rate: The speed at which reactants are converted to products in the forward direction, measured as a change in amount (or concentration/pressure) per unit time.
Reverse reaction rate: The speed at which products are converted back to reactants in the reverse direction, measured as a change in amount (or concentration/pressure) per unit time.
Equal rates are the criterion for equilibrium, regardless of the starting mixture.
= forward reaction rate (e.g., amount or concentration change per second)
= reverse reaction rate (e.g., amount or concentration change per second)
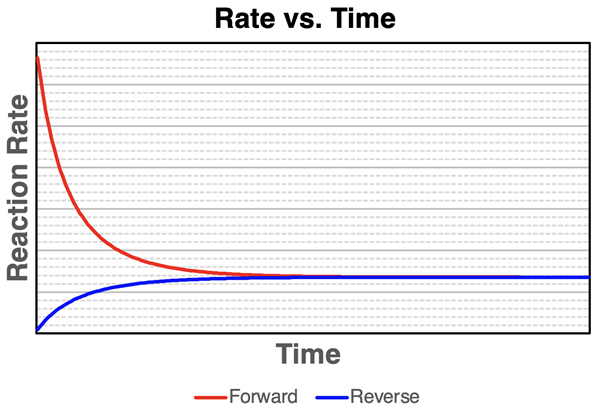
Rate–time curves for a reversible reaction: the forward rate starts high and decreases as reactants are consumed, while the reverse rate starts near zero and increases as products accumulate. The intersection point marks equilibrium, where and macroscopic amounts stop changing even though reactions continue microscopically. Source
Why equal rates cause “no net change” in amounts
Net change depends on the difference between the two rates
The amounts of reactants and products change only when one direction “wins” overall.
If , products form faster than they are consumed
If , reactants re-form faster than they are consumed
If , amounts are replenished and consumed equally, creating no net change
At equilibrium, reactant and product amounts can remain constant even though molecules continue reacting in both directions.
“No net change” does not mean “equal amounts”
A common misconception is that equilibrium implies equal amounts of reactants and products. In fact, equilibrium only requires equal rates, not equal concentrations or equal moles.
Some equilibria have mostly products at equilibrium
Others have mostly reactants
Both cases can still satisfy
Conditions for establishing equilibrium in practice
The system must allow both directions to occur
Equilibrium can only be established for a reversible reaction under conditions where:
products are not permanently removed as they form
reactants are not continuously added in a way that prevents settling
If a product escapes (for example, a gas leaving an open container), the reverse reaction may be suppressed, and the system may not reach equilibrium.
The system must be effectively closed
To observe constant amounts over time, the reacting mixture is typically in a closed system so matter is not lost or gained overall. Otherwise, measured amounts can change due to leakage, evaporation, or venting rather than chemistry.
Reaching equilibrium takes time
Equilibrium is established when the rates become equal, but that equality may occur:
quickly (fractions of a second) for some reactions
slowly (minutes to days) for others
Even after equilibrium is established, microscopic reacting continues; what changes is that the forward and reverse rates have adjusted to match.
How equilibrium can be recognised experimentally (conceptually)
Because equilibrium is defined by equal rates (hard to measure directly), it is often inferred from observations consistent with no net change in amounts:
a measurable composition that becomes constant over time (in a closed system)
properties tied to composition (such as pressure in a sealed gas reaction) that stabilise
These observations support that the system has reached the equilibrium state described in the syllabus focus.
FAQ
They use isotopic labelling or tracers to distinguish molecules formed via the forward pathway from those formed via the reverse pathway.
By monitoring labelled species over time, they can determine that both directional rates are non-zero yet equal.
A catalyst lowers activation energy for both directions.
It typically makes equilibrium established sooner (rates become equal faster), but it does not change the equilibrium condition itself ($r_f = r_r$) or the final equilibrium composition.
It can, but poor mixing can create concentration gradients.
Local regions may temporarily behave as if not at equilibrium even if the overall system is approaching it; stirring helps the whole container reach a uniform state where equal rates apply throughout.
If matter enters or leaves, amounts can change for reasons unrelated to the forward/reverse chemistry.
A closed system ensures that any change (or lack of change) in amounts reflects the reaction dynamics rather than physical loss or gain of substances.
No. Individual molecules continue to convert between reactant and product forms.
Equilibrium means the population-level conversions balance: as many molecules go forward per unit time as go backward per unit time.
Practice Questions
(2 marks) In a sealed container, a reversible reaction reaches a state where the amounts of reactants and products remain constant over time. State the condition on reaction rates that must be true at this point.
States that the forward and reverse reaction rates are equal (1)
Uses correct comparative language/symbolism (e.g., ) (1)
(5 marks) A student claims that at equilibrium “the reaction has stopped and the amounts of reactants and products are equal.”
(a) Explain, in terms of forward and reverse reaction rates, what it means for equilibrium to be established. (3 marks)
(b) State whether reactant and product amounts must be equal at equilibrium, and justify your answer. (2 marks)
Mentions the reaction is reversible with both forward and reverse processes occurring (1)
States that at equilibrium the forward and reverse rates are equal (1)
Links equal rates to no net change in amounts over time (1) (b)
States amounts (or concentrations) do not have to be equal (1)
Justifies that equilibrium requires equal rates, not equal amounts; equilibrium composition can be reactant- or product-favoured (1)

