AP Syllabus focus: ‘The reaction quotient describes the relative amounts of species at any time and tends toward the equilibrium constant; at equilibrium, Q equals K (Qc = Kc and Qp = Kp).’
Chemical systems rarely start at equilibrium.
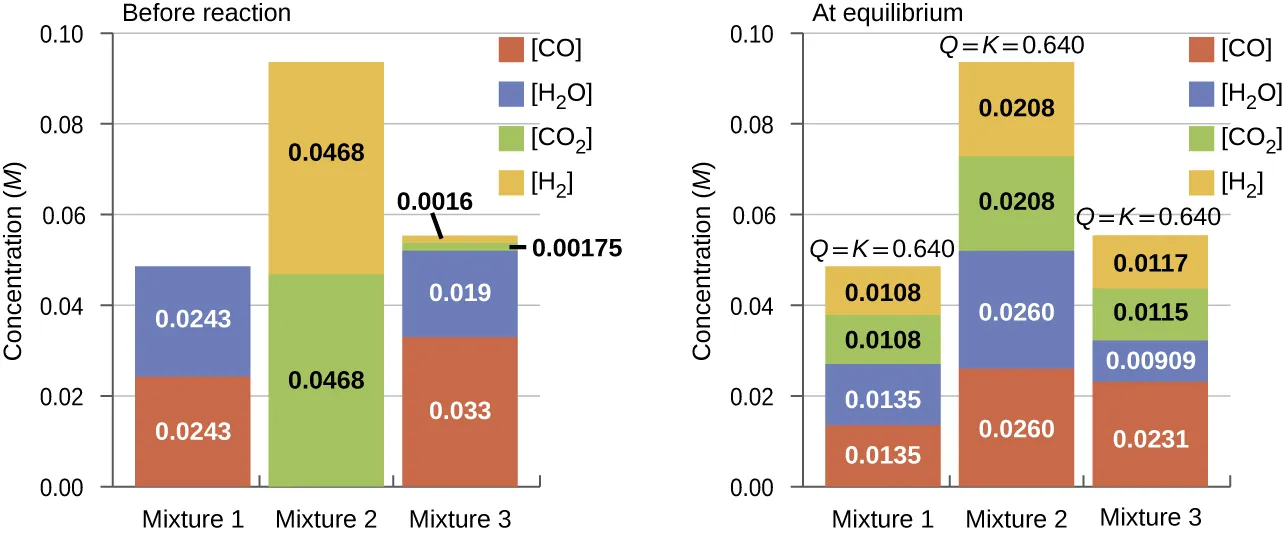
Bar charts compare multiple initial mixtures to their final equilibrium compositions for the same reaction at a fixed temperature. Even though the systems start with different amounts of reactants and products, each evolves until the reaction quotient reaches the same equilibrium value (i.e., becomes ). Source
The reaction quotient, Q, provides a snapshot of composition at any moment and allows you to judge whether the system has reached equilibrium, where Q matches K.
What Q and K represent
Reaction quotient (Q): a composition snapshot
Reaction quotient (Q): A unitless ratio constructed like an equilibrium-constant expression, using the current (not necessarily equilibrium) concentrations or partial pressures.
Q is calculated from measured or stated amounts “right now.” Because it mirrors the equilibrium expression, Q translates the macroscopic mixture into a single number that can be compared to the equilibrium benchmark.
= reaction quotient (unitless)
= stoichiometric coefficient (unitless)
“products/reactants” = the appropriate current quantities (typically concentrations for or partial pressures for )
In practice, you compute using molar concentrations and using gas partial pressures, matching the way K is reported for that reaction.
Equilibrium constant (K): the target ratio at equilibrium
Equilibrium constant (K): A unitless ratio (with the same form as Q) that equals the value of the reaction quotient only when the system is at equilibrium at a specific temperature.
K is not “how fast” a reaction occurs. It encodes the equilibrium composition constraint for a given balanced equation and temperature. For the same reaction at the same temperature, K is fixed, while Q can vary as the mixture changes.
How Q compares to K
The central relationship: equilibrium means equality
When a system is at equilibrium, the measured composition already satisfies the equilibrium constraint, so Q and K match exactly.
= reaction quotient evaluated with current amounts (unitless)
= equilibrium constant for that reaction at that temperature (unitless)
This equality is what the syllabus emphasizes: at equilibrium, , including the specific matching pairs and .
If Q and K are not equal, the system is not at equilibrium
A mismatch between Q and K indicates the composition does not yet satisfy the equilibrium constraint.
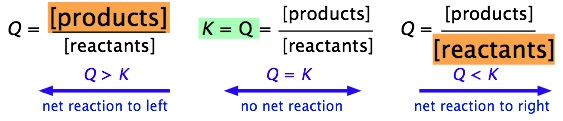
A schematic comparison diagram summarizes how the sign of the difference between and predicts the net direction of reaction. It visually encodes the rules (shift toward products), (equilibrium), and (shift toward reactants), linking the numerical comparison to Le Châtelier-style “shift” language. Source
In a closed system at constant temperature, the reaction proceeds in whatever net way is necessary for Q to move toward K. Conceptually:
Q changes because concentrations/partial pressures change as reactants and products interconvert.
K does not change during that adjustment (as long as temperature is unchanged).
This is the meaning of “Q tends toward K”: as the reaction mixture evolves over time, the quotient computed from the momentary composition approaches the constant value required at equilibrium.
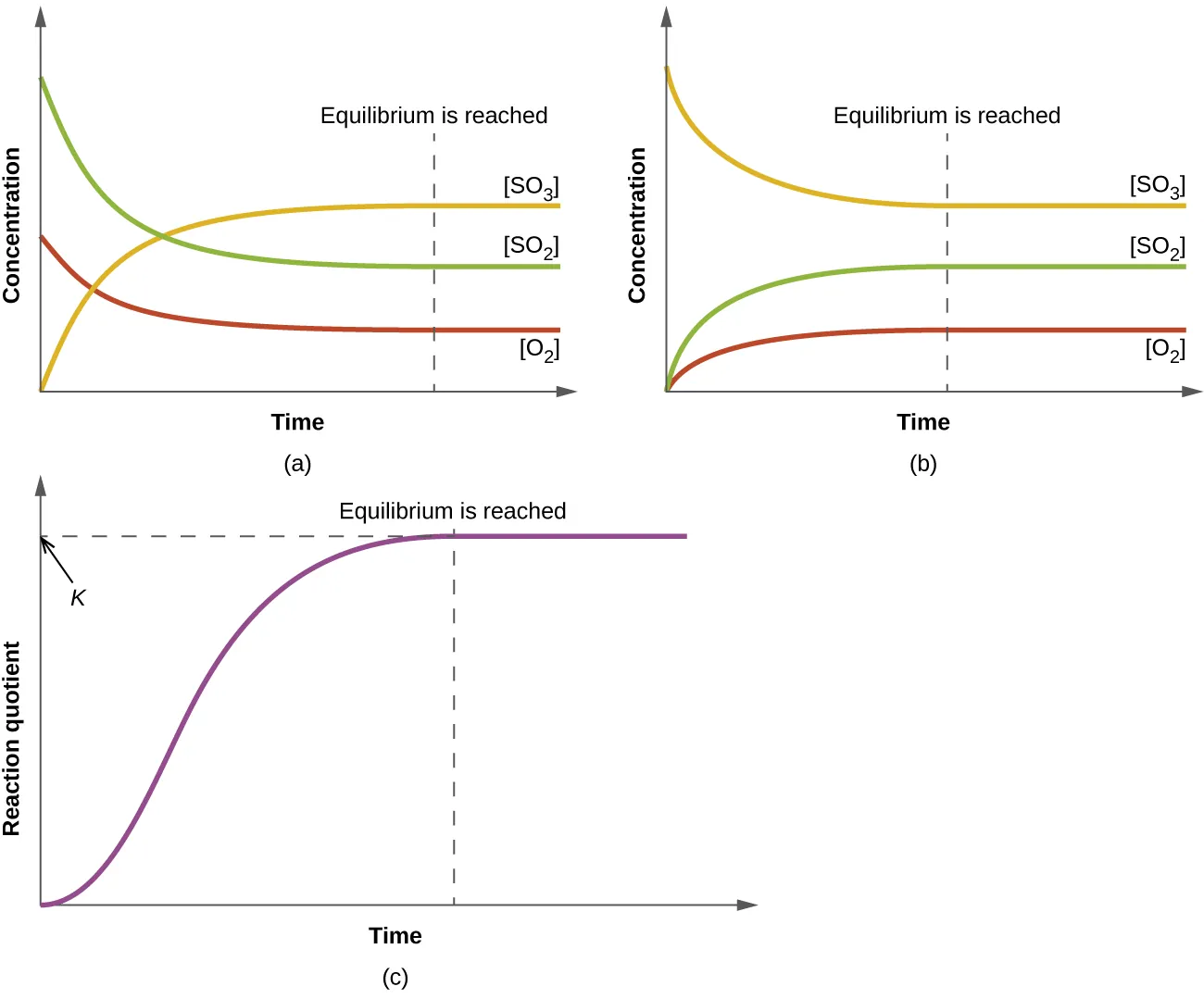
Time-course plots for a gas-phase equilibrium illustrate how concentrations change until they become constant at equilibrium. The third panel shows the reaction quotient rising and then leveling off at the constant value , emphasizing that changes during the approach to equilibrium while is the equilibrium benchmark at a fixed temperature. Source
Matching the correct forms: Qc with Kc, Qp with Kp
Because and are built from different measurable quantities, the comparison must be consistent:
Use with when the expression is based on molar concentrations.
Use with when the expression is based on gas partial pressures.
A correct comparison is not about which is “better,” but about whether Q and K were constructed from the same type of data for the reaction as written.
Common interpretation and reporting pitfalls
Mixing forms: Comparing to (or to ) is not meaningful without additional relationships; treat them as distinct equilibrium descriptions.
Using the wrong reaction: Q and K depend on the balanced equation as written; if the written reaction changes, the expression changes, so the numerical comparison must match that exact form.
Forgetting Q is time-dependent: Q can be computed at any instant (initial, intermediate, or equilibrium), so a single system can have many Q values but only one K at a fixed temperature.
Assuming equality from “no visible change”: A lack of observable change suggests equilibrium, but the definitive chemical criterion is based on composition data.
FAQ
Strictly, they are written using activities (dimensionless). In AP contexts, concentrations/pressures approximate activities, and the standard-state scaling makes the overall ratio treated as unitless.
Collisions continue in both directions. As particles interconvert, the measured ratio in $Q$ changes until the composition stabilises at the value required by $K$.
Yes. At equilibrium, reactions still occur in both directions, but the composition no longer changes, so $Q$ stays equal to $K$ over time.
Because of measurement uncertainty, you judge whether $Q$ agrees with $K$ within experimental error. Small discrepancies may reflect instrument limits rather than genuine non-equilibrium.
Not automatically, but it must match the constant you compare to. Concentration-based data should be compared with $K_c$ (not $K_p$) for the reaction as written.
Practice Questions
Q1 (3 marks) For a reaction at a fixed temperature, . A sample is analysed and found to have . (a) State whether the system is at equilibrium.
(b) Give a reason based on the relationship between and .
(a) At equilibrium (1)
(b) Because (1)
Explicitly links equality to equilibrium composition constraint (1)
Q2 (6 marks) For at a certain temperature, . A mixture has and . (a) Write an expression for .
(b) Calculate .
(c) Use your result to state whether the mixture is at equilibrium.
(a) (2: correct form; correct powers)
(b) Substitution shown and (2)
(c) Not at equilibrium because (or ) (2)

