AP Syllabus focus: ‘The law of mass action relates reaction stoichiometry to Q and K. Q can be written using concentrations (Qc) or gas partial pressures (Qp), with matching equilibrium forms Kc and Kp.’
Chemical equilibria are quantified by writing algebraic expressions that represent reactant and product amounts. In AP Chemistry, you must correctly build Q and K expressions from a balanced equation using concentrations or partial pressures.
Law of mass action: the blueprint for Q and K
For a general reversible reaction:
the law of mass action states that the reaction’s composition can be summarized by a ratio:
Products in the numerator
Reactants in the denominator
Each term raised to a power equal to its stoichiometric coefficient in the balanced equation
This same structure is used for both the reaction quotient (Q) and the equilibrium constant (K); only when you evaluate it differs.
Reaction quotient Q (any time) vs equilibrium constant K (at equilibrium)
Reaction quotient (Q): A mass-action expression evaluated using the system’s current concentrations or partial pressures (not necessarily at equilibrium).
A system’s Q value changes as the reaction proceeds, because the amounts of reactants and products change.
Equilibrium constant (K): The value of the same mass-action expression specifically when the system is at equilibrium at a given temperature.
K is fixed for a given reaction at a fixed temperature, but Q can take on many values as conditions change.
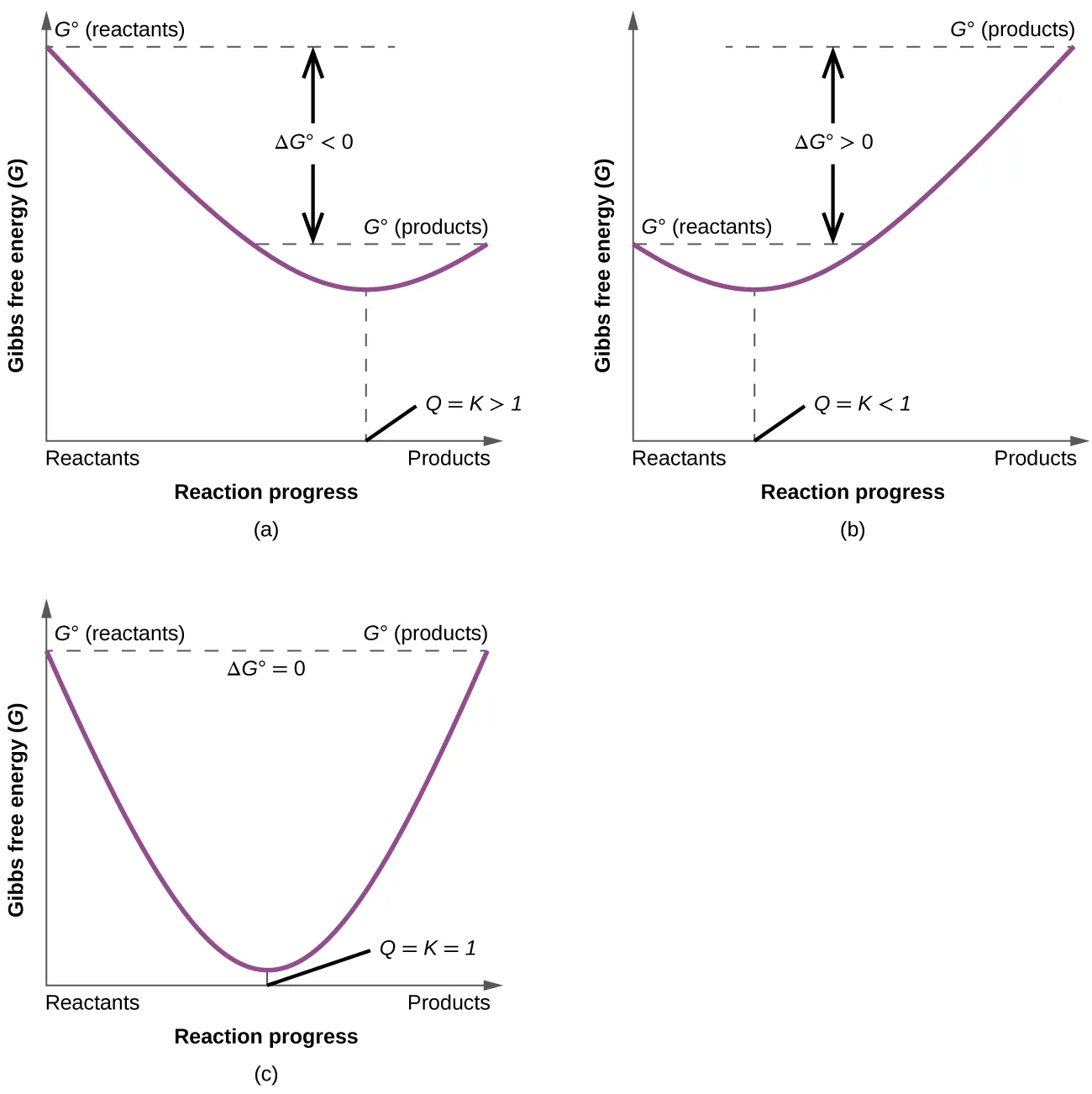
These plots show Gibbs free energy versus reaction progress, with the minimum corresponding to equilibrium where . If the mixture starts with nonequilibrium amounts (), the reaction proceeds in the direction that decreases until it reaches that minimum. The figure reinforces that the “drive to equilibrium” is fundamentally an energetic tendency toward minimum . Source
Writing and (concentration-based)
Use or when amounts are expressed as molar concentrations (typical for dissolved species, and sometimes gases when concentration data are provided).
= molar concentration of species in
= equilibrium concentration of species in
The exponents (like and ) come directly from the balanced equation coefficients, so balancing must happen before writing any Q or K expression.
Checklist for writing or
Balance the chemical equation.
Write brackets for each species: .
Multiply product terms together in the numerator.
Multiply reactant terms together in the denominator.
Raise every term to its coefficient power.
Writing and (partial-pressure-based)
Use or for gaseous equilibria when amounts are expressed as partial pressures.
= partial pressure of gas (commonly in atm or kPa)
= equilibrium partial pressure of gas (same pressure unit throughout)
When writing or , pressures must be partial pressures for each gas (not total pressure), and you must be consistent with units within the expression.
Choosing between concentration and pressure forms
Use / when the problem gives concentrations ().
Use / when the problem gives partial pressures ().
The structure (products over reactants with coefficient exponents) stays the same in all cases.
FAQ
Because the expression is based on how the reaction rate depends on repeated “units” of reactants/products in the balanced chemical equation.
In thermodynamic terms, the exponent reflects how each species’ chemical potential contributes to the overall equilibrium condition.
In rigorous treatments, $K$ is unitless because it is defined using activities relative to a standard state.
Intro courses sometimes attach units by treating $[,]$ or $P$ as numerical values with units; AP questions typically focus on correct expression form, not unit analysis.
Yes. If the equation is correctly balanced with fractional coefficients, the corresponding $Q$ or $K$ expression uses fractional exponents.
Many students prefer multiplying all coefficients by a common factor to avoid fractions before writing the expression.
You can write the symbolic form of $Q_p$, but to evaluate it numerically you need each gas’s partial pressure.
Partial pressures can be found from mole fractions: $P_i=x_iP_\text{total}$.
They can differ because concentration and pressure scale differently with temperature and volume.
That is why $Q_c$ should be compared to $K_c$, and $Q_p$ should be compared to $K_p$, not mixed across forms.
Practice Questions
(1–3 marks) For , write an expression for .
1 mark: Correct form with partial pressures:
1 mark: Correct exponent of 2 on matching the coefficient
(4–6 marks) Consider . (a) Write an expression for .
(b) Write an expression for .
(c) Using , , , calculate .
(a) 2 marks: (1 mark for correct numerator species and exponent; 1 mark for correct denominator species and exponent)
(b) 1 mark:
(c) 2 marks: Substitution shown (1 mark) and correct value (1 mark)

