AP Syllabus focus: ‘Very small equilibrium constants indicate reactions that barely proceed, so reactants dominate the equilibrium mixture.’
Reactions that reach equilibrium can do so with very different final compositions. This page explains how to interpret a small equilibrium constant, , what it implies about equilibrium amounts, and common misconceptions.
What a “small ” means at equilibrium
When chemists say an equilibrium is reactant-favoured, they mean the equilibrium mixture contains mostly reactants rather than products.
Equilibrium constant (): A temperature-dependent ratio, built from equilibrium concentrations (or partial pressures), that quantifies the equilibrium composition for a balanced reversible reaction.
A very small (much less than 1) means the equilibrium ratio is dominated by reactant terms. In words, the system forms only a small amount of products before the reverse process becomes competitive enough to halt any net change.
Interpreting “barely proceed”
The syllabus statement “reactions that barely proceed” refers to extent of reaction at equilibrium, not whether products ever form.
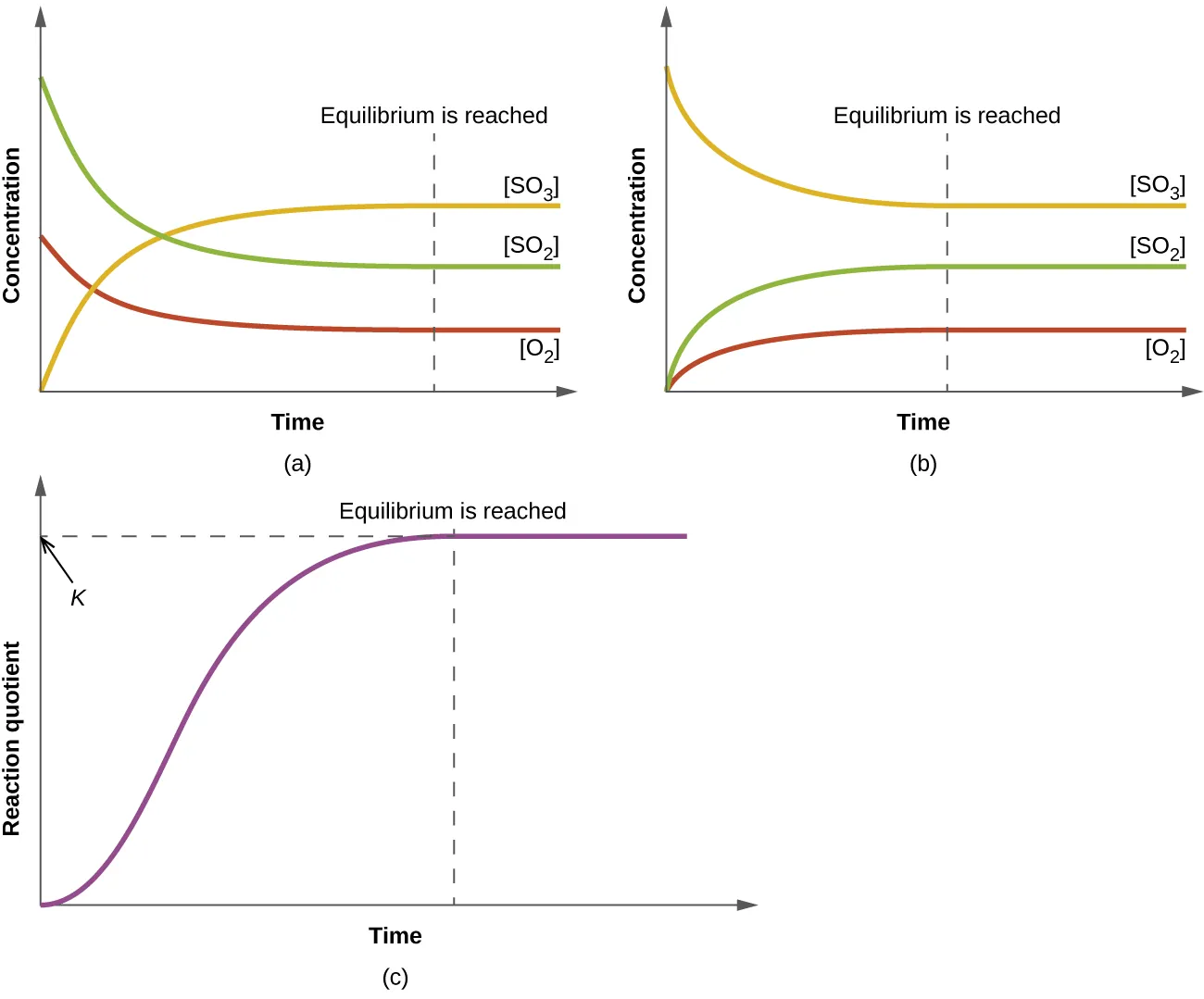
These plots show concentrations changing with time as a reversible reaction approaches equilibrium, leveling off once equilibrium is reached. The bottom panel shows the reaction quotient rising and then becoming constant at , emphasizing that equilibrium corresponds to a stable composition (not the absence of reaction). Source
Products are present at equilibrium, but in comparatively tiny amounts.
The forward and reverse reactions still occur; equilibrium composition is simply weighted toward reactants.
The equilibrium position is such that only a small fraction of reactant molecules are converted into product molecules.
Linking the magnitude of to the equilibrium composition
For a general reaction, the mathematical structure of shows why small values correspond to reactant-heavy mixtures.
= equilibrium constant (unitless in AP Chemistry treatment)
= equilibrium concentration in
Because product terms appear in the numerator, a very small implies the numerator is small compared with the denominator at equilibrium.
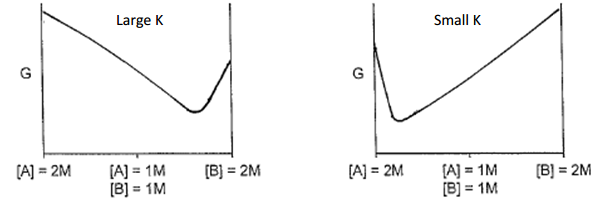
This paired diagram plots Gibbs energy versus composition for a simple system, contrasting a large- case with a small- case. When is small, the minimum in occurs at a reactant-rich composition, illustrating why equilibrium favors reactants even though some products are still present. Source
Conceptually:
Either product concentrations are small, or
reactant concentrations remain relatively large, or
both.
“Reactants dominate” depends on stoichiometry
“Reactants dominate” is a statement about the overall mixture, but the exact ratio depends on:
the balanced equation (exponents matter),
the form of used for the system (concentration-based versus pressure-based expressions, if applicable),
the specific species included (only species that appear in the balanced equilibrium expression).
Even when is very small, a species raised to a power (because of its coefficient) can make the ratio highly sensitive to modest concentration changes.
What small does and does not tell you
What it tells you (high-utility interpretations)
A very small equilibrium constant indicates:
the equilibrium composition is reactant-rich;
the net formation of products is thermodynamically unfavoured under those conditions (products are not strongly stabilised relative to reactants);
reaching equilibrium does not require large product buildup.
What it does not tell you (common misconceptions)
A small is often confused with kinetic ideas, but:
Small does not mean “slow reaction.” Speed is controlled by kinetics (activation energy, mechanism), not by the equilibrium constant.
Small does not mean “no products.” Equilibrium mixtures can still contain detectable products; the key is that reactants are much more abundant.
Small does not guarantee low yields in every situation. The equilibrium position limits yield for a closed system at that temperature; practical strategies that change conditions or remove products are separate ideas (beyond this subsubtopic’s focus).
Recognising “very small” in practice
On the AP Chemistry scale, “very small” is typically interpreted as orders of magnitude below 1 (for example, , , etc.). The smaller the value:
the more strongly the equilibrium mixture is weighted toward reactants;
the more the reaction “barely proceeds” before equilibrium is established.
This interpretation aligns directly with the syllabus: Very small equilibrium constants indicate reactions that barely proceed, so reactants dominate the equilibrium mixture.
FAQ
At a fixed temperature, $K$ and $\Delta G^\circ$ are linked: a very small $K$ corresponds to a positive $\Delta G^\circ$, meaning products are less thermodynamically favoured under standard conditions.
Yes. “Dominates” is relative. If initial amounts are large or detection is sensitive, even a tiny equilibrium fraction of product can be observable.
A more rigorous treatment uses activities (effective concentrations) referenced to a standard state, which makes $K$ dimensionless. AP Chemistry typically uses concentrations/pressures directly as an approximation.
No. Equilibrium means both sides are present. “Reactant-favoured” only means the left side is much more abundant at equilibrium than the right side.
When product concentrations are near instrument detection limits, measured $K$ can have large relative uncertainty. Side reactions, adsorption, or contamination can also bias very small equilibrium measurements.
Practice Questions
(2 marks) For the equilibrium , the value of at a given temperature is . State what this indicates about the equilibrium mixture.
States that equilibrium is reactant-favoured / mostly present (1)
States that only a very small amount of forms / reaction barely proceeds toward products (1)
(5 marks) Consider with a very small equilibrium constant at a particular temperature. Explain, using the equilibrium constant expression, why reactants dominate at equilibrium, and address one common misconception about what a small implies.
Correct expression: K=\dfrac{[NO]^2[Cl_2]}{[NOCl]^2} (1)
Explains that very small means numerator is much smaller than denominator at equilibrium (1)
Links to composition: and/or must be small and/or remains large (1)
Interprets as reactant-favoured equilibrium / reaction barely proceeds to products (1)
Correctly rejects misconception: small does not mean slow rate OR does not mean zero products (1)

