AP Syllabus focus: ‘When a reaction is written in reverse, the equilibrium constant is inverted (K becomes 1/K).’
Reversible reactions can be written in either direction, but the equilibrium constant must match the written equation. This page explains the single most important rule: reversing a reaction inverts its equilibrium constant.
Core idea: K depends on how the reaction is written
Equilibrium constant (K) is tied to a specific balanced chemical equation and temperature. If you rewrite the same equilibrium with products and reactants swapped, the algebra of the equilibrium expression swaps numerator and denominator, so the numerical value must invert.
Equilibrium constant (K): A temperature-dependent constant equal to the ratio of product activities to reactant activities, each raised to their stoichiometric coefficients, for a particular balanced reaction.
This dependence on the written reaction is why it is essential to keep careful track of direction when using a given K value.
General rule for reversing
If the forward reaction has equilibrium constant K, then the reverse reaction has equilibrium constant 1/K. This is true regardless of reaction type, as long as it is the exact same chemical change written backwards.
= equilibrium constant for the reaction written in reverse (dimensionless)
= equilibrium constant for the reaction written in the forward direction (dimensionless)
Why inversion works (law-of-mass-action logic)
Consider a generic reaction written in the forward direction:
Reactants appear in the denominator of K.
Products appear in the numerator of K.
Stoichiometric coefficients become exponents in K.
When you reverse the reaction:
Every species that was a product becomes a reactant, and vice versa.
Therefore, everything that was in the numerator moves to the denominator, and everything that was in the denominator moves to the numerator.
Swapping numerator and denominator is exactly taking the reciprocal.
What this means conceptually
Reversing does not change the underlying equilibrium state at a given temperature; it changes only how you describe it.
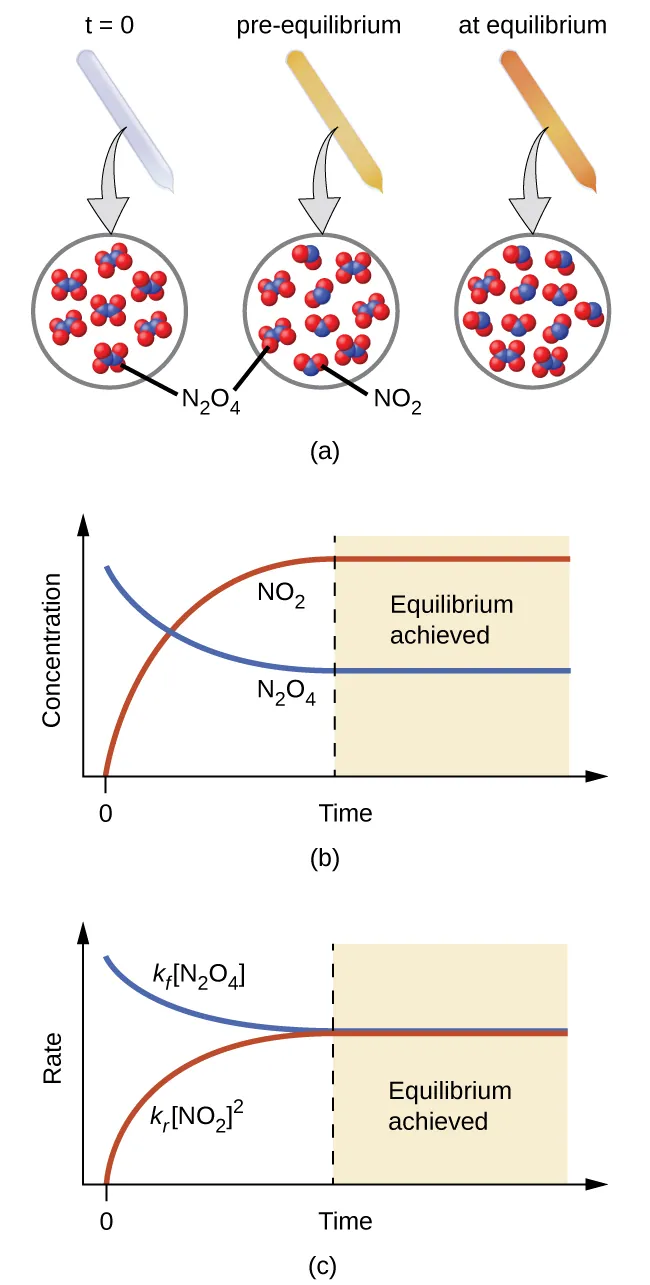
A classic concentration-vs-time (and rate-vs-time) depiction for the \ce{N2O4(g) <=> 2 NO2(g)} system. It shows reactant and product concentrations approaching constant plateau values while the forward and reverse rates converge to the same value at equilibrium, emphasizing equilibrium’s dynamic (not static) nature. Source
The “forward” constant and the “reverse” constant are just two reciprocals describing the same equilibrium mixture.
Practical implications for AP Chemistry
Using a tabulated K with your written equation
Before substituting into any equilibrium expression:
Confirm whether the given K corresponds to the reaction as written in the problem.
If your equation is the reverse of the tabulated one, replace K with 1/K.
Common pitfalls to avoid
Forgetting the inversion: Using K as-is after flipping the arrow gives an answer inconsistent with the reaction you wrote.
Mixing up direction words: “Formation” versus “decomposition” or “products” versus “reactants” often signals that the reaction has been reversed relative to a standard reference.
Assuming K stays the same: K changes when you rewrite the equation, even though the equilibrium composition at that temperature is unchanged.
Interpreting magnitude after reversal
Inverting K flips whether the reaction appears product-favoured or reactant-favoured for the written direction:
If K >> 1 for the forward direction, products are favoured for that written equation; the reverse direction will have K << 1, meaning reactants (of the reverse equation) are favoured.
If K = 1, the forward and reverse constants are the same (since ), indicating neither side is favoured for the written reaction.
Quick checklist (direction ↔ K consistency)
Did you reverse the chemical equation? Then invert K.
Is your K expression consistent with the species on each side of your written equation? Products must be on top, reactants on bottom.
Are you treating K as dimensionless (as used in AP Chemistry problem setups)?
FAQ
At equilibrium, the same mixture must satisfy either written direction.
Reversal changes which species you label “products” in the expression, so the ratio is flipped, even though the particle-level equilibrium composition is unchanged.
It implies the reverse reaction, as written, is strongly unfavoured under standard equilibrium conditions at that temperature.
In practice, it means the reverse direction produces very little “product” before equilibrium is reached.
Because $\log(1/K) = -\log K$, reversing a reaction changes the sign of $\log K$.
So a positive $\log K$ becomes negative for the reverse reaction, and vice versa.
In rigorous thermodynamics, K is defined using activities and is dimensionless; inversion keeps it dimensionless.
If you see apparent units from concentration forms, treat them as artefacts of the simplified model; the reciprocal rule still applies.
Compare reactants/products carefully, including physical states and coefficients.
If every species appears on the opposite side (with the same coefficients), it is the exact reverse, so you must use $1/K$ for consistency.
Practice Questions
A reaction is reported as: with at a fixed temperature.
Write the equilibrium constant for .
States that reversing the reaction inverts the constant: (1)
Gives (1)
For , at a given temperature.
A student instead writes the equilibrium as .
(a) State the relationship between the two equilibrium constants. (1)
(b) Calculate the value of the equilibrium constant for the student’s equation. (2)
(c) Explain, in terms of the numerator and denominator of the equilibrium expression, why the value changes. (2)
(a) (1)
(b) Correct reciprocal set-up (1); correct value (accept 0.14–0.15) (1)
(c) Identifies products in numerator/reactants in denominator for K (1); explains reversal swaps numerator and denominator so K becomes the reciprocal (1)

