AP Syllabus focus: ‘Given a balanced reaction, initial concentrations or partial pressures, and the appropriate equilibrium constant, the equilibrium composition of a system can be predicted.’
Predicting equilibrium composition connects measurable starting conditions to the final, constant mixture at equilibrium.
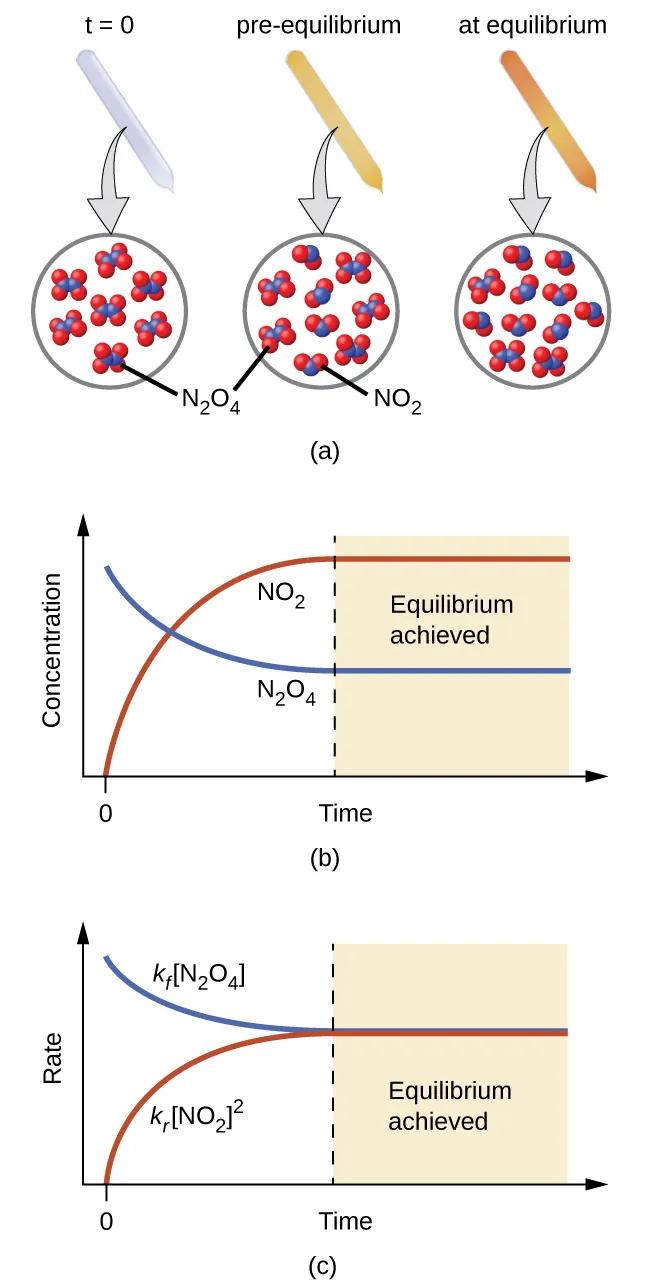
This figure shows how reactant and product concentrations evolve with time and then level off once equilibrium is reached. The flat (constant) regions of the curves represent the equilibrium composition, even though forward and reverse reactions continue to occur at equal rates. It provides a visual bridge between the qualitative idea of “constant mixture” and the quantitative ICE-table method used to compute equilibrium amounts. Source
You will translate a balanced equation into an algebraic model, then use the equilibrium constant to determine equilibrium concentrations or partial pressures.
Core idea: model equilibrium with a single variable
For a reversible reaction, stoichiometry constrains how species change as the system relaxes to equilibrium. You represent the change using one unknown (often ) and solve for the equilibrium amounts that satisfy the equilibrium constant.
ICE table: A bookkeeping setup that tracks Initial amounts, Change based on stoichiometry, and Equilibrium amounts in terms of a single variable.
An ICE table can be written using molar concentrations (for ) or partial pressures (for ), matching the form of the equilibrium constant provided.
Step-by-step procedure (no shortcuts)
1) Start from the balanced chemical equation
Confirm the equation is balanced and identify stoichiometric coefficients.
Decide whether the problem uses (concentrations) or (partial pressures); use the one given.
2) Write equilibrium amounts in terms of a reaction progress variable
Let a general reaction be: .
Choose a variable (commonly ) to represent the extent of the forward reaction.
Build change expressions using coefficients:
: decreases by
: decreases by
: increases by
: increases by
3) Convert given initial information into the right form
For problems, express initial conditions as initial concentrations.
For problems, express initial conditions as initial partial pressures.
If initial information is given as moles in a fixed volume, convert to concentration before building the ICE table.
If initial information is given as total pressure plus mole information, obtain partial pressures before building the ICE table.
4) Substitute equilibrium expressions into the equilibrium-constant expression
= equilibrium molar concentration,
= initial molar concentration of ,
= change variable (extent in concentration units),
After substitution, you will have one equation with one unknown (). The same substitution approach applies to using partial pressures (with terms instead of ).
Solving for equilibrium composition
Recognise the algebra you may need
If powers are 1 and the expression simplifies neatly, you may get a linear equation in .
Many common equilibria produce a quadratic (or higher-order) equation; solving may require the quadratic formula or numerical methods.
Apply chemical validity checks (required)
Once you obtain , compute each equilibrium amount and check:
Non-negativity: no equilibrium concentration/pressure can be negative.
Stoichiometric limits: cannot exceed what would consume a limiting reactant below zero (e.g., ).
Reasonableness with powers: when coefficients create squared/cubed terms, verify the final equilibrium amounts satisfy the constant’s expression when substituted back in.
Optional approximation (only if justified)
If equilibrium shifts only slightly from initial conditions, you may test an approximation such as “ is small compared with an initial amount” to simplify algebra, but you must confirm the approximation is consistent with the final value of .
FAQ
Reject any root that makes an equilibrium concentration/pressure negative.
Also reject roots that violate a stoichiometric bound, e.g. $[A]_0-ax<0$.
It is typically justified only when $x$ is a small fraction of the relevant initial amount (often a few per cent).
After solving, check the ratio $\dfrac{x}{[,]_0}$ (or $\dfrac{x}{P_0}$) to confirm.
Nothing about the method changes: include the nonzero product amount in the ICE table.
This can reduce the magnitude of the net change needed to satisfy the equilibrium constant.
The multiplier comes directly from stoichiometric coefficients.
If the balanced equation forms $2$ moles (or pressure units) of a species per “reaction event,” its change term must be $+2x$.
Keep units consistent with the form used: $\mathrm{mol,L^{-1}}$ for concentrations and pressure units (e.g. atm) for $K_p$ work.
Carry extra digits during algebra, then round final equilibrium amounts appropriately (typically to the given data’s significant figures).
Practice Questions
Question 1 (2 marks) For , at a fixed temperature. Initially, and . Write expressions for and in terms of , and write the equation you would solve for .
and (1)
(1)
Question 2 (5 marks) For , at a fixed temperature. Initially, and . Calculate the equilibrium partial pressures.
Let and (1)
Correct setup: (1)
Rearrangement to solvable form, e.g. (1)
Correct physically valid root: (1)
and (1)

