AP Syllabus focus: ‘When Q equals K, the system is at dynamic equilibrium: forward and reverse reactions occur at the same rate and the reactant-to-product ratio remains constant.’
Chemical equilibrium is identified by comparing the system’s current composition to its equilibrium condition. The most direct test uses the reaction quotient Q and the equilibrium constant K at the same temperature.
What it means to “be at equilibrium”
A system is at equilibrium when its composition matches the equilibrium condition for that reaction at that temperature. Practically, that means a calculation based on the current amounts of reactants and products gives .
Dynamic equilibrium (what is happening microscopically)
Dynamic equilibrium: A state in which forward and reverse reactions continue simultaneously at equal rates, so the macroscopic amounts of reactants and products do not change.
Even though particles are still reacting, there is no net change in measurable quantities (concentration or partial pressure) because the forward and reverse processes balance.
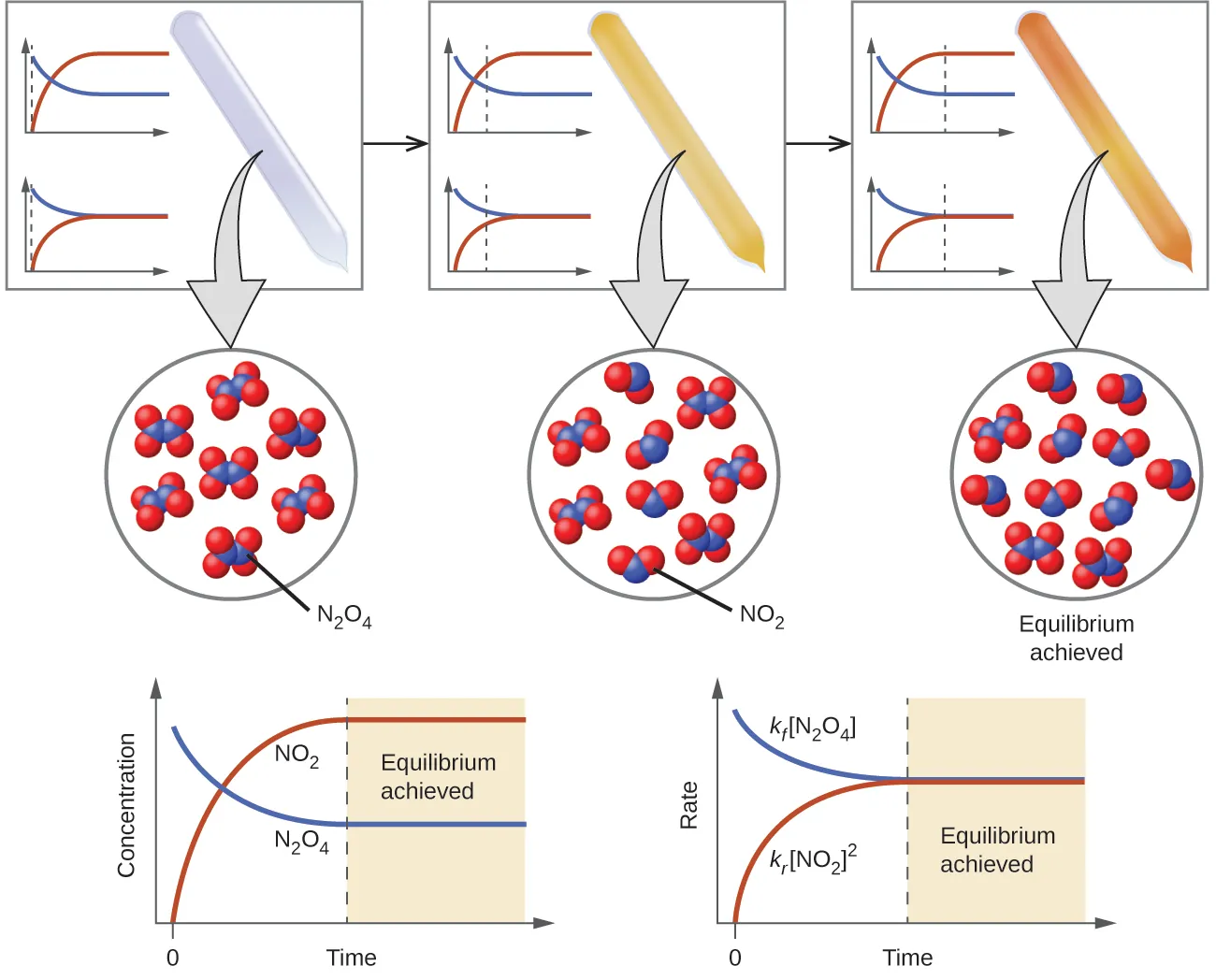
Concentration–time and rate–time plots for the reversible system. The concentration curves flatten when equilibrium is reached, while the forward and reverse rate curves converge to the same value—illustrating dynamic equilibrium (no macroscopic change despite ongoing microscopic reaction). Source
The two quantities you compare: Q and K
To recognise equilibrium, you need both:
Q, calculated from the current (not necessarily equilibrium) composition
K, known for that reaction at the given temperature
Reaction quotient, Q
Reaction quotient (Q): A number computed from the current concentrations (or partial pressures) of species in a reaction mixture, using the same algebraic form as the equilibrium constant.
Q is a snapshot: it describes the system’s current reactant-to-product ratio using stoichiometric exponents.
A key point is that Q is only meaningful when written for the balanced chemical equation.
Equilibrium constant, K
Equilibrium constant (K): The value of Q specifically when the system is at equilibrium for a particular reaction at a particular temperature.
K sets the target ratio required for equilibrium at that temperature; recognising equilibrium is checking whether the current ratio (Q) has reached that target (K).
Shared mathematical form (how Q and K are built)
= reaction quotient (unitless)
= molar concentrations,
= stoichiometric coefficients from the balanced equation
= equilibrium constant (unitless)
Because the algebra is the same, recognising equilibrium is a direct equality check: compute Q from the current mixture and see whether it matches K.
Recognising equilibrium (Q = K) in practice
What you can conclude when Q = K
If Q equals K, then:
The system is at dynamic equilibrium
The forward and reverse reaction rates are equal
The reactant-to-product ratio remains constant (even though individual molecules keep reacting)
Importantly, equilibrium does not require equal concentrations of reactants and products; it requires the ratio described by K.
A reliable checklist
To justify a claim that a system is at equilibrium, ensure:
You used the correct balanced equation to write Q
Q and K are expressed in a consistent way for the data type you used (concentrations with concentration-form expressions)
The value of K corresponds to the same temperature as the system
Your comparison accounts for measurement uncertainty (experimental data rarely give a perfect match)
Common interpretation pitfalls
“No observable change” is consistent with equilibrium, but not proof by itself; a system can change very slowly. The quantitative criterion is .
Equal forward/reverse rates does not mean equal amounts present; it means equal rates of conversion in opposite directions.
If Q is not equal to K, the system is not at equilibrium (regardless of whether concentrations look steady over a short time interval).
FAQ
It depends on uncertainty in concentration/pressure measurements and propagation of error in the Q expression.
A practical approach is to compare the difference using significant figures and estimated percentage uncertainties rather than expecting an exact match.
Yes. If conditions are changing (e.g., temperature drifting, leaks, side reactions), a momentary match can occur.
True equilibrium requires the system’s conditions to be constant so the match persists.
At higher ionic strength or non-ideal gas conditions, “effective concentration” differs from measured concentration/pressure.
Activities correct for non-ideality so the equilibrium criterion remains $Q=K$ in terms of activities.
You still use the same idea, but the “constant” components are not included in the expression.
Then $Q=K$ is checked using only the variable species (typically gases and aqueous ions).
Each independent equilibrium has its own Q and K condition.
A mixture is fully at equilibrium only when every relevant equilibrium expression satisfies $Q=K$ simultaneously under the same conditions.
Practice Questions
(3 marks) For at a fixed temperature, . A reaction mixture has , , and . Determine whether the system is at equilibrium.
Writes/uses (1)
Calculates (1)
Compares and concludes not at equilibrium because (1)
(5 marks) For at a given temperature, explain how the statement “” is used to recognise equilibrium. Your answer must refer to reaction rates and to what happens to the reactant-to-product ratio over time.
States that is calculated from the current composition using the balanced equation (1)
States that is the value of that same expression at equilibrium for that temperature (1)
States that equilibrium is recognised when (1)
Links to dynamic equilibrium: forward and reverse reactions continue (1)
States that at equilibrium forward and reverse rates are equal and the reactant-to-product ratio remains constant (1)

