AP Syllabus focus: ‘Particulate representations can show the relative numbers of reactant and product particles present before equilibrium is reached and once the system is at equilibrium.’
Equilibrium is often introduced with macroscopic observations, but AP Chemistry also expects you to interpret particle-level (particulate) models. These diagrams communicate composition changes over time and what “no observable change” means microscopically.
What a particulate model is showing
Purpose and conventions
Particulate model: a drawing that represents individual particles (atoms, molecules, or ions) as symbols to show relative amounts, identity, and sometimes arrangement within a system.
A particulate model is a snapshot of a system at a particular time.
When multiple snapshots are shown (e.g., “before” and “at equilibrium”), the goal is to compare relative numbers of reactant and product particles.
Common conventions you should read carefully:
Each symbol corresponds to one particle (unless a key states otherwise).
Different shapes/colours represent different species.
The container boundary implies a fixed volume for that snapshot.
Particles are usually drawn as randomly distributed to suggest constant motion and mixing.
“Before equilibrium” in particulate terms
Before equilibrium is established, particle counts change in the net direction of the reaction:
If reactant particles are being converted overall, the diagram sequence shows:
reactant counts decreasing
product counts increasing
Both reactants and products may be present even early on; the key idea is that the ratio is still changing from snapshot to snapshot.
Recognising “at equilibrium” from particles
The equilibrium snapshot
Dynamic equilibrium: a state in which reactant and product particles continue to interconvert, but the amounts of each remain constant over time in a closed system.
A particulate model labelled “at equilibrium” should imply constant composition. In practice, that means if you compare two “late-time” snapshots:
the number of each type of particle is the same (or effectively unchanged within the diagram’s resolution)
both reactants and products are present together
A crucial interpretation skill: “constant” does not mean “equal numbers,” and it does not mean “reaction stopped.”
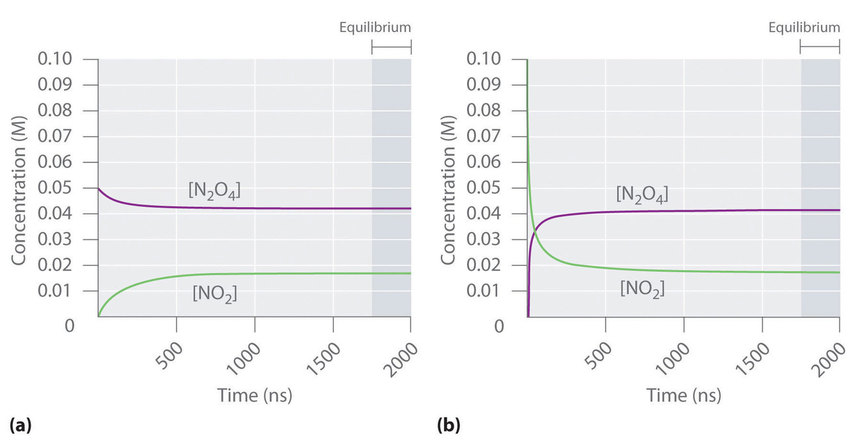
Concentration–time curves for the \ce{N2O4(g) <=> 2NO2(g)} system illustrate how reactant and product concentrations change initially and then become constant once equilibrium is reached. The shaded/marked equilibrium region highlights that “no observable change” corresponds to concentrations (and thus particle counts in a fixed volume) no longer changing with time. This helps link particle-level constancy to a standard macroscopic graph used in equilibrium problems. Source
It means the counts are no longer changing with time in the sequence of particulate frames.
How to justify equilibrium using particle counts
When deciding whether a system has reached equilibrium from particulate diagrams, focus on:
Time comparison: Are the numbers of each species unchanged between the last two snapshots?
Coexistence: Are both reactants and products present simultaneously?
Net change: Is there any continued overall trend in counts (increase/decrease), or has the trend stopped?
Use precise language tied to what is shown:
“The system appears at equilibrium because the reactant and product particle counts are constant between the two final frames.” Avoid unsupported claims like “it’s at equilibrium because there are lots of products.”
Connecting particle counts to the balanced reaction
Stoichiometric consistency in the model
Particulate models must be consistent with the balanced chemical equation, because particle numbers change in fixed reacting ratios. When reading a diagram sequence:
Identify the species (reactants/products) from the symbols.
Use the balanced equation to check whether changes in counts make sense.
Example logic (no calculation required): if , then a decrease of 2 A particles should correspond to an increase of 1 B particle in the net forward direction (and vice versa for net reverse).
This stoichiometric lens helps you detect common diagram traps:
showing products forming without the required reactant decrease
showing the wrong relative change in particle counts for a given equation
What particulate models do (and do not) encode
A particulate snapshot typically communicates:
relative amounts (counts) of each species
whether a species is present or absent
qualitative mixing and distribution
It usually does not communicate:
exact concentration values (unless a scale is given)
particle speeds or collision details
the actual forward/reverse rates (only their consequence: changing or constant amounts)
Practical reading checklist for AP-style diagrams
Step-by-step interpretation (no maths required)
Confirm what each symbol means using the key.
Decide whether each frame represents the same container/volume.
Count (or estimate) each species to compare relative numbers.
Determine whether the counts are still changing (pre-equilibrium) or constant (equilibrium).
State your reasoning using particle language: “numbers of particles remain constant over time” and “both reactants and products are present.”
Common misconceptions to avoid
Equilibrium is not 50/50. A diagram can show many more reactant particles than product particles (or the reverse) and still be at equilibrium, as long as counts are constant over time.
Equilibrium is not static. Even if the snapshot looks “unchanging,” equilibrium implies ongoing interconversion at the particle level; the diagram sequence signals this by showing no net change in counts at late times.
Do not infer completion. If reactant particles remain, the reaction has not “gone to completion”; it has reached a steady composition.
FAQ
Use relative comparisons rather than perfect counting.
If exact counting is impractical, look for:
clear increases/decreases between frames
whether the final two frames are visually identical in composition
dominance of one species (many vs few) without claiming exact percentages
Treat each ion as a separate particle if the diagram shows dissociation.
Focus on:
which ions change in number (involved in the reversible process)
which ions remain unchanged (often spectator ions in the drawing style)
charge is usually implied by labels; electroneutrality may not be explicitly enforced in simplified diagrams
They are emphasising a well-mixed system where particles are in constant motion.
Random placement communicates:
no macroscopic layering
frequent collisions throughout the container
that the diagram is a composition snapshot, not a structural model (unless stated otherwise)
Yes, if they represent different equilibrium conditions (e.g., different starting amounts or constraints) while still showing constant composition over time.
Equilibrium is defined by time-independence of amounts in that system, not by a unique universal ratio in all circumstances.
They compress many realities into a simple picture.
Limitations include:
rates are not shown directly
concentration is approximated by counts without an explicit scale
real systems have vastly more particles, so “small-number” drawings can exaggerate fluctuations
some drawings omit solvent or use non-literal particle sizes for clarity
Practice Questions
Q1 (1–3 marks) A particulate diagram sequence for a reversible reaction shows three snapshots at times , , and . From to , the numbers of each type of particle are unchanged. State what time the system first reaches equilibrium and give one particle-level reason.
Correct time identified: (1)
Reason: the numbers of reactant and product particles remain constant from onward / no net change in particle counts (1)
Additional correct statement: both reactants and products are present at that time (1)
Q2 (4–6 marks) A reversible reaction is represented by . A student draws particulate snapshots of a closed container:
At time : 10 particles of and 2 particles of
At time : 8 particles of and 6 particles of
At time : 8 particles of and 6 particles of
(a) Identify whether equilibrium has been reached by and by .
(b) Use the particulate information to justify your answers.
(c) Comment on whether the reaction has stopped at equilibrium.
(a) By : not at equilibrium (1)
(a) By : at equilibrium (1)
(b) Justification for : particle numbers are still changing compared with (1)
(b) Justification for : particle numbers are constant from to (1)
(b) Correct stoichiometric consistency statement: decrease of 2 from to corresponds to increase of 4 (1)
(c) Reaction has not stopped; forward and reverse processes continue but with no net change in amounts (1)

