AP Syllabus focus: ‘Percent ionization can be calculated using pKa (or pKb) and initial concentration, or using initial concentration and an equilibrium concentration of any species in the expression.’
Percent ionisation connects equilibrium thinking to how much of a weak acid or base actually forms ions in water. It is a ratio-based quantity that can be obtained from information or measured equilibrium concentrations.
What percent ionisation means
Weak acids and bases partially ionise, so the equilibrium amount of ions formed is typically small compared with the initial amount dissolved.
Percent ionisation: the percentage of the initial weak acid/base that has reacted with water to form ions at equilibrium.
Percent ionisation is a practical way to compare “how weak” two weak acids/bases are under stated conditions, but it always depends on the initial concentration.
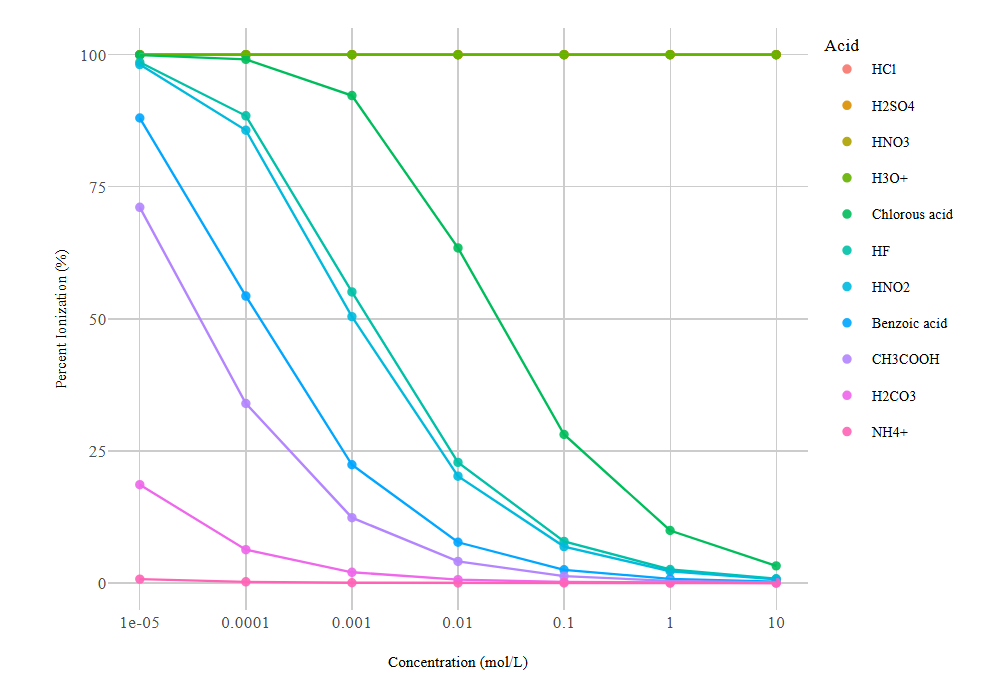
Percent ionisation plotted against molar concentration for several acids (including strong vs. weak). The graph highlights the concentration-dependence of weak-acid ionisation: as the analytical concentration decreases (dilution), the fraction ionised increases, whereas strong acids remain near 100% across concentrations. Source
Core setup: relate “ionised amount” to an equilibrium concentration
For a monoprotic weak acid, . The amount that ionises is the equilibrium increase in (and equals formed). For a weak base, , and the ionised amount tracks the equilibrium increase in (and equals formed).
In typical AP problems, you treat water’s contribution to or as negligible compared with the weak acid/base contribution (unless explicitly told otherwise).
= percent ionisation, %
= equilibrium concentration produced by ionisation (e.g., for a weak acid, for a weak base), M
= initial concentration of or , M
The “ionised amount” can come directly from an equilibrium concentration you are given (for any species tied stoichiometrically to ion formation) or indirectly from data.
Method 1: Use (or ) and initial concentration
This method matches the syllabus statement: you can compute percent ionisation using (or ) plus the initial concentration.
Key idea:
Convert to (or to ).
Use an ICE-style equilibrium setup to relate or to the unknown “ionised amount” (often called ).
Once is found (it represents for a weak acid or for a weak base), plug into the percent ionisation equation.
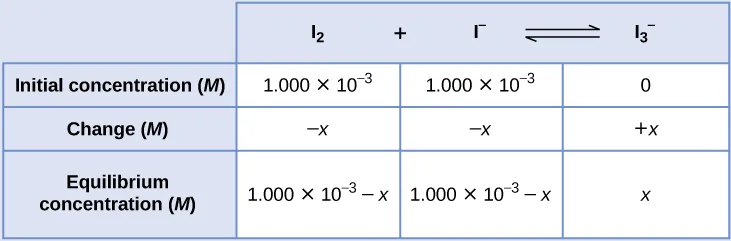
Example ICE table showing the “Initial–Change–Equilibrium” structure with an unknown extent of reaction . This format makes the stoichiometric link between ion formation and equilibrium concentrations explicit, which is the step needed before computing . Source
Important interpretation points:
At fixed , decreasing the initial concentration generally increases percent ionisation (because the denominator shrinks faster than the equilibrium shift reduces ion formation).
A smaller (larger ) generally means a larger percent ionisation at the same initial concentration.
Method 2: Use initial concentration and an equilibrium concentration from the equilibrium expression
This is the second syllabus route: use the initial concentration and an equilibrium concentration of any species in the expression.
Common ways this appears:
You are given (from pH) for a weak acid solution.
You are given (from pOH) for a weak base solution.
You are given or at equilibrium, which equals the ionised amount by stoichiometry for monoprotic systems.
Then:
Identify the ionised amount (the equilibrium concentration created by ionisation).
Divide by the initial concentration of the weak acid/base and multiply by 100.
Common pitfalls (conceptual, not computational)
Using for a weak base (you need for percent ionisation of bases).
Confusing percent ionisation with “percent dissociation” of strong electrolytes; for weak acids/bases it is an equilibrium-dependent quantity.
Forgetting that percent ionisation is tied to the initial concentration, not the equilibrium concentration remaining.
FAQ
Yes. Because $K_a$ and $K_b$ vary with temperature, the equilibrium position changes, so the ionised amount (and therefore percent ionisation) can change.
Percent ionisation depends on initial concentration. Dilution typically increases percent ionisation even though the solution becomes less acidic overall.
Unless stated otherwise, it commonly refers to the first ionisation step (formation of the first $H_3O^+$) because later steps are typically much smaller.
When the solution is extremely dilute or explicitly includes other significant acid/base sources; then background water autoionisation or added ions may not be negligible.
Measured pH (or pOH) gives $[H_3O^+]{eq}$ (or $[OH^-]{eq}$). Combined with the prepared initial concentration, this directly yields percent ionisation.
Practice Questions
(2 marks) A solution of a monoprotic weak acid has . Calculate the percent ionisation.
Uses (1)
Correct substitution and result (1)
(5 marks) A weak base has . A student prepares . Determine the expression you would use to find percent ionisation and state the quantity that represents the ionised amount. (No numerical solving required.)
Converts to via (1)
Writes equilibrium: (1)
Identifies ionised amount as for a monoprotic base (1)
Gives percent ionisation setup: with (1)
States how would be related to using equilibrium concentrations (e.g., with ICE form) (1)

