AP Syllabus focus: ‘For a conjugate acid–base pair, Ka and Kb are related by Kw: Kw = Ka × Kb and pKw = pKa + pKb.’
Conjugate acid–base pairs are tightly linked by water’s autoionisation. This relationship lets you convert between and (and between and ) without solving a new equilibrium from scratch.
Core idea: conjugates are connected through water
Conjugate acid–base pair
Conjugate acid–base pair: Two species that differ by exactly one proton, such as or .
In water, acids ultimately increase and bases ultimately increase .
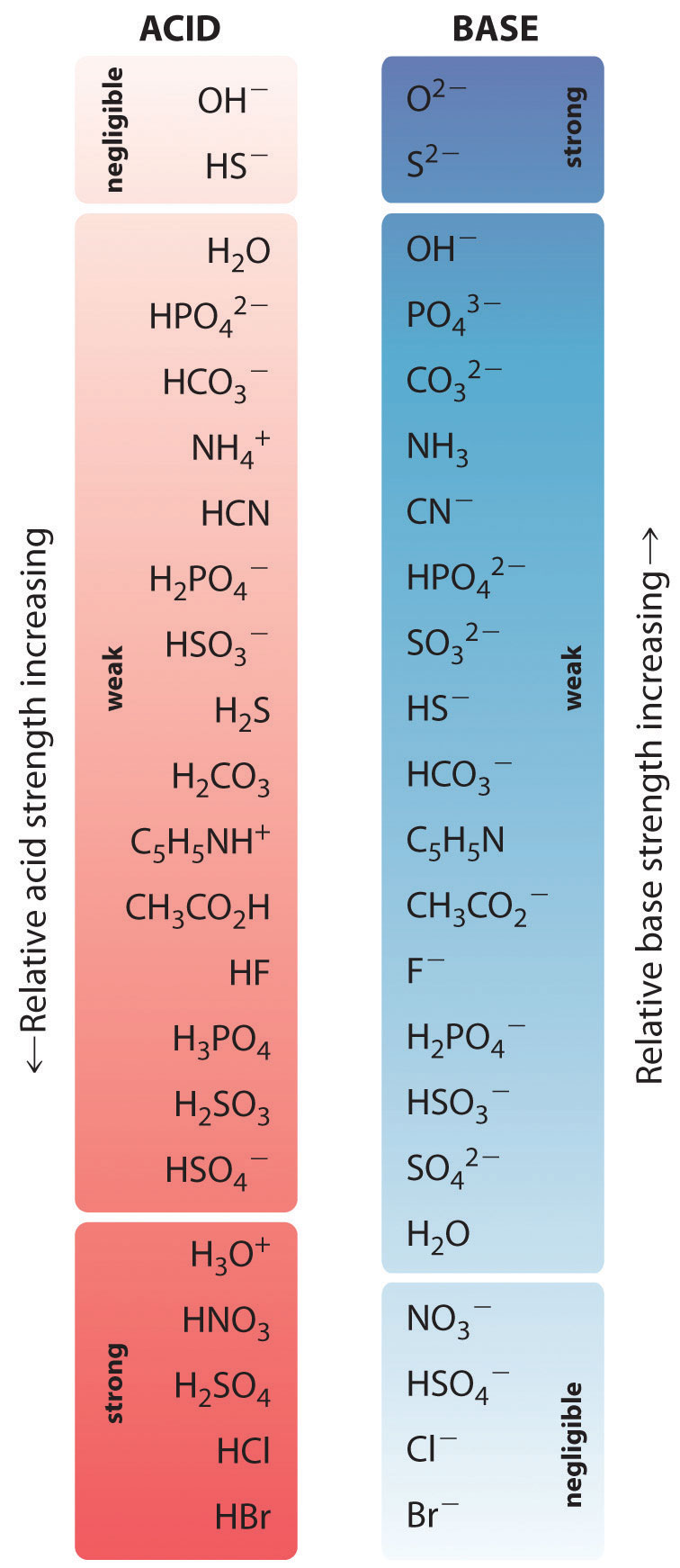
A labeled diagram showing how conjugate acid–base pairs are created by adding or removing exactly one proton. It visually connects the “parent” acid/base to its conjugate partner, reinforcing that conjugates differ by one . Source
Because and are themselves linked by water’s equilibrium, the strength of an acid determines the weakness of its conjugate base (and vice versa).
The role of
(the ion-product constant of water) ties hydronium and hydroxide together, so any equilibrium that produces one tends to suppress the other. For a conjugate pair, this creates an exact quantitative link between their equilibrium constants.
Required relationships: and
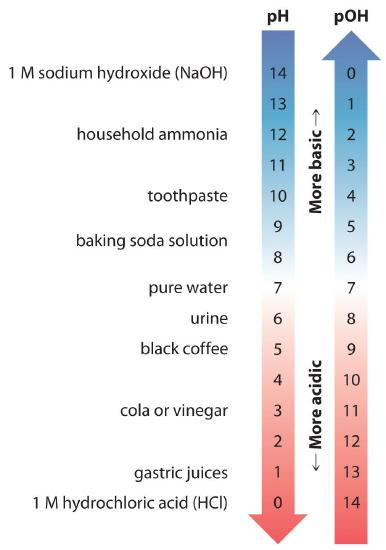
A pH/pOH scale diagram illustrating the inverse relationship between acidity (higher , lower pH) and basicity (higher , lower pOH). This provides a visual rationale for why changes in one ion concentration suppress the other in water, consistent with . Source
= ion-product constant of water (dimensionless; commonly treated as at )
= acid dissociation constant for the acid in the conjugate pair (dimensionless)
= base dissociation constant for the conjugate base in the conjugate pair (dimensionless)
= (dimensionless; commonly at )
These equations apply only when the two equilibria correspond to true conjugates in the same solvent system (water).
Why the relationship is true (conceptual, equilibrium-based)
Consider the standard acid reaction and the conjugate base reaction in water:
Acid reaction (defines ):
Conjugate base reaction (defines ):
If you conceptually add these reactions, and cancel, leaving the net process:
That net process is exactly water autoionisation, so the product of the two equilibrium constants must equal . This is the chemical reason a stronger acid (larger , smaller ) must have a weaker conjugate base (smaller , larger ).
How AP Chemistry expects you to use it
Converting between conjugate strengths
Common uses that stay within this subsubtopic:
Given , find using .
Given , find using .
Given (or ), reverse the process to get (or ).
Interpreting magnitude without calculating pH
You should be able to reason that:
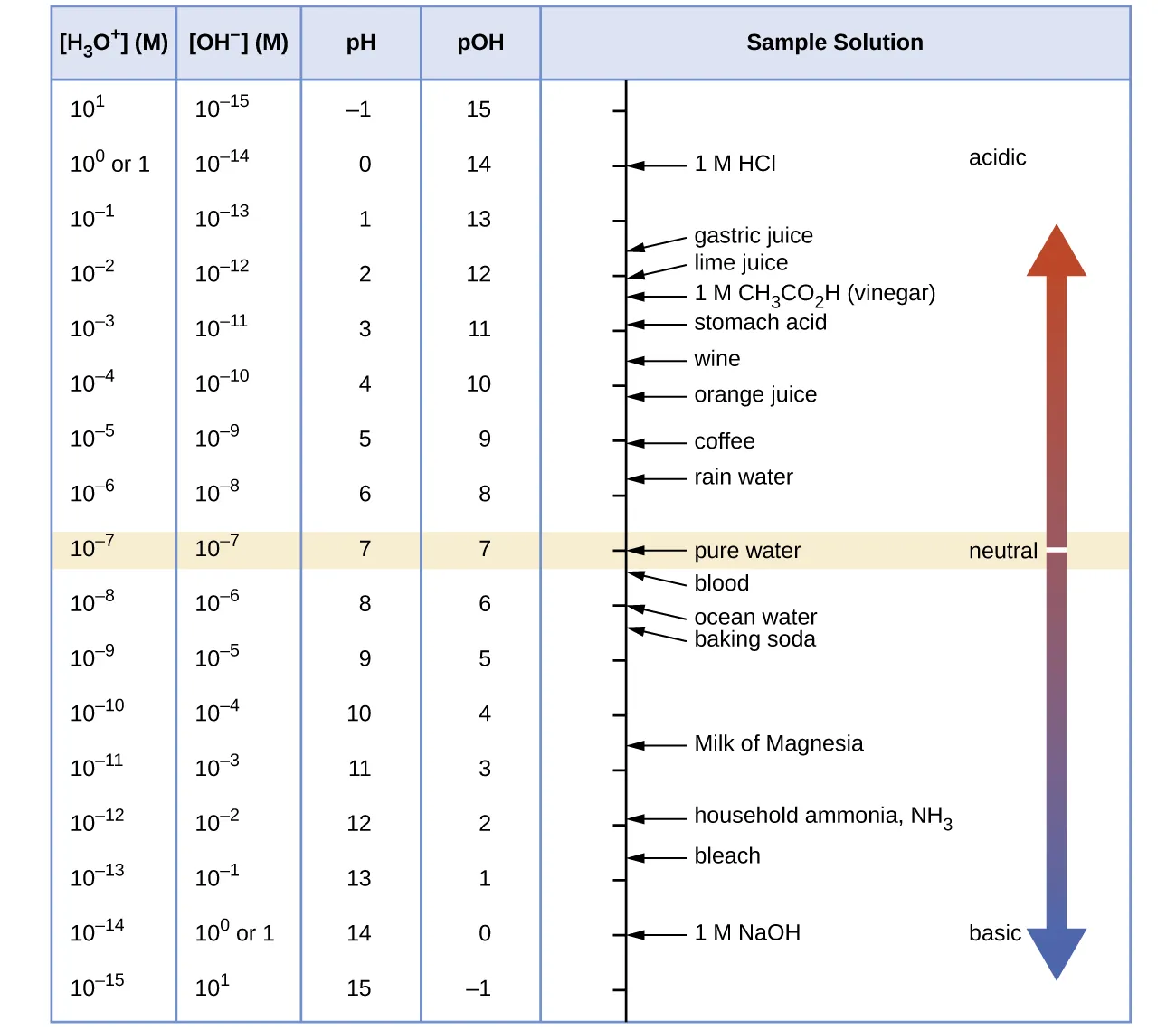
A reference chart linking , , pH, and pOH across a wide concentration range, making the logarithmic relationships concrete. It supports quick magnitude-based reasoning (e.g., when pH decreases by 1, increases by a factor of 10). Source
If (very strong acid), then (extremely weak conjugate base).
If is small (weak acid), the conjugate base has a larger (but often still small) .
Common pitfalls to avoid
Using the relationship for species that are not conjugates (do they differ by exactly one ?).
Mixing up which constant belongs to which species (acid gets ; its conjugate base gets ).
Forgetting that relationships are subtraction: .
FAQ
Yes, but you must use the correct $K_w$ (and therefore $pK_w$) for that temperature.
At different temperatures, $K_w$ changes, so the product $K_aK_b$ matches that new value.
The cancellation argument relies on the two equilibria summing to water autoionisation.
If two species differ by more than one proton, or are related by another reaction pathway, their constants won’t multiply to $K_w$.
An amphiprotic species has both a $K_a$ (as an acid) and a $K_b$ (as a base), but they refer to different conjugate partners.
You must pair each constant with the correct conjugate:
$HCO_3^-/CO_3^{2-}$ for $K_a$
$H_2CO_3/HCO_3^-$ for $K_b$
The exact thermodynamic relationship uses activities, not concentrations.
In high-ionic-strength solutions, concentration-based “$K$” values can shift, so the simple product may appear imperfect unless activity corrections are applied.
Often yes, because it avoids rounding from repeated log and antilog steps.
Using $pK$ values is typically cleaner when data are given in $pK_a$ form, provided you use a consistent $pK_w$ value.
Practice Questions
Q1 (2 marks) The acid has in water at . State the value of for .
Uses (or ). (1)
. (1)
Q2 (5 marks) In water at , for is .
(a) Write the expression linking , , and . (1)
(b) Calculate for . (2)
(c) Hence calculate for . (2)
(a) . (1)
(b) . (2: subtraction set-up; correct value)
(c) (allow ). (2: correct conversion; correct value)

