AP Syllabus focus: ‘Weak acid and strong base react quantitatively; if weak acid is in excess a buffer forms and pH can be found with Henderson–Hasselbalch, while excess OH− sets pH when base is in excess.’
Mixing a weak acid with a strong base combines stoichiometry and equilibrium thinking. First determine which reactant is left over after neutralization, then choose the correct pH method: buffer logic, leftover strong base, or conjugate-base hydrolysis.
Core idea: reaction goes to completion first
A weak acid (HA) only partially ionizes in water by itself, but with a strong base (like NaOH), the acid–base reaction is effectively quantitative (driven to products). Treat the initial step as a limiting-reactant problem using moles.
Buffer: a solution containing significant amounts of a weak acid and its conjugate base (or a weak base and its conjugate acid) that resists pH change when small amounts of acid or base are added.
After the quantitative reaction, the remaining major species determine the pH.
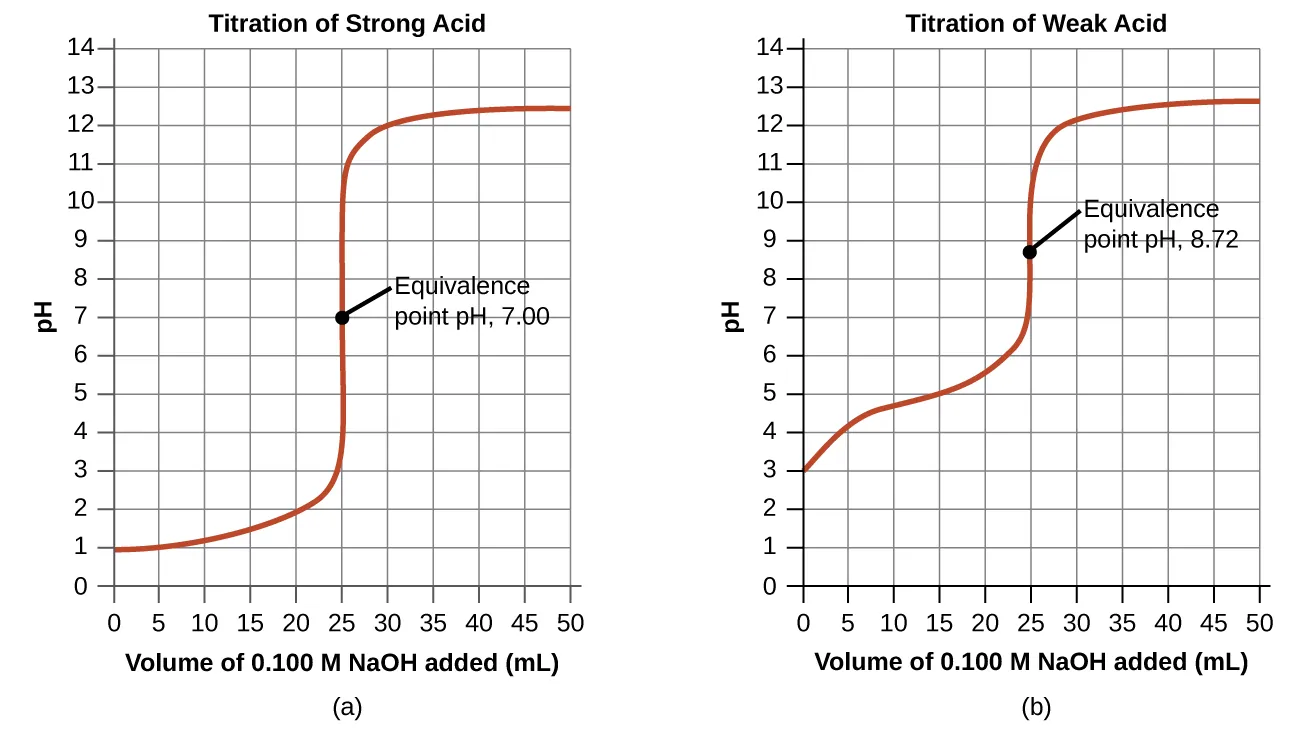
Titration curve for a weak acid–strong base system, showing how pH changes with added base. The buffer region occurs before the equivalence point, and the half-equivalence point highlights the relationship (a key justification for Henderson–Hasselbalch). The equivalence point is basic because the solution contains predominantly the conjugate base, which hydrolyzes water to form . Source
= weak acid (acid form)
= hydroxide from the strong base
= conjugate base produced
= (unitless)
= equilibrium concentrations (often taken from post-reaction amounts), in
How to decide the pH method (three cases)
Step 1: do stoichiometry with moles
Convert each reactant to moles (not molarity) because mixing changes volume.
Use the 1:1 stoichiometric relationship for a monoprotic weak acid: moles HA react with equal moles OH⁻.
Subtract to find what remains after neutralization:
leftover HA, or leftover OH⁻, or neither (equimolar).
Case A: weak acid in excess (buffer forms)
If moles HA > moles OH⁻:
All OH⁻ is consumed, producing some A⁻.
The final mixture contains both HA and A⁻ in appreciable amounts → this is a buffer.
pH is found using Henderson–Hasselbalch with the ratio (often computed from post-reaction amounts divided by total volume). Key AP point: the buffer pH depends on pKₐ and the conjugate pair ratio, not on the initial acid concentration alone.
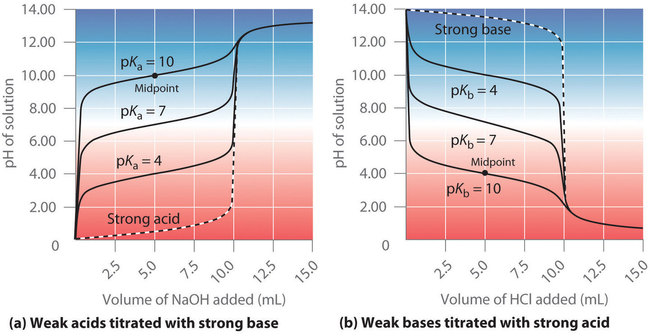
Family of titration curves illustrating how changing weak-acid strength (different values) alters the curve shape and the pH at key points. The midpoint (half-equivalence) occurs where the conjugate pair amounts are comparable, making the buffer behavior most evident and giving . This helps connect the Henderson–Hasselbalch relationship to a concrete, graphical feature of the titration. Source
Case B: strong base in excess (leftover OH⁻ sets pH)
If moles OH⁻ > moles HA:
All HA is consumed; extra OH⁻ remains in solution.
The pH is dominated by the excess strong base, so treat it like a strong-base solution:
find from leftover moles and total volume
then convert to pH using pOH and as needed. Important: in this case, the weak-acid equilibrium is negligible compared with the large, direct contribution of remaining OH⁻.
Case C: equimolar amounts (no excess; conjugate base only)
If moles OH⁻ = moles HA:
All HA is converted to A⁻; there is no HA left to pair with A⁻, so it is not a buffer.
The pH is controlled by A⁻ reacting with water to produce some OH⁻ (basic hydrolysis).
Setting up the pH requires an equilibrium expression for A⁻ + H₂O and an appropriate base strength value for A⁻ (commonly handled via a approach). Practically, the major conceptual outcome is that the solution is basic because the conjugate base is present without its acid partner.
FAQ
Because mixing changes the total volume, initial molarities are not directly comparable.
Use $n=C\times V$ for each reactant, then compare stoichiometric amounts.
When one buffer component is extremely small after neutralisation.
If $[A^-]/[HA]$ is very large or very small, the “buffer” approximation breaks down and full equilibrium may be needed.
Dilution changes $[A^-]$ and $[HA]$ but often leaves their ratio nearly unchanged.
Therefore pH changes little, even though buffer capacity decreases.
Neutralisation can occur stepwise.
Depending on moles of $OH^-$ added, you may form intermediate conjugate species (e.g., $H_2A \rightarrow HA^-$), changing which conjugate pair is present.
Because the product $A^-$ is a base.
It reacts with water to generate some $OH^-$, shifting pH above 7 even without excess strong base.
Practice Questions
(2 marks) A solution of weak acid is mixed with aqueous . After reaction, there are more moles of than . State (i) what type of solution results and (ii) which equation should be used to determine the pH.
(1) Identifies a buffer / buffer solution forms.
(1) States Henderson–Hasselbalch: .
(5 marks) Equal volumes of weak acid and are mixed. The acid has . Explain how you would determine the pH of the final mixture. No numerical calculation is required.
(1) Converts to moles and compares moles and (limiting reagent approach).
(1) States reaction is quantitative: .
(1) Concludes is limiting and is in excess; both and present (buffer).
(1) Determines post-reaction amounts: remaining moles and formed moles .
(1) Uses Henderson–Hasselbalch with concentrations (or mole ratio with common volume): .

