AP Syllabus focus: ‘Weak base and strong acid react quantitatively; excess weak base forms a buffer (use Henderson–Hasselbalch), while excess H3O+ sets pH when strong acid is in excess.’
When a weak base is mixed with a strong acid, the initial reaction is a fast, essentially complete neutralisation. The pH then depends on which reactant is left over, or whether only the conjugate acid remains.
Core reaction idea (quantitative step)
A weak base (symbol B) accepts a proton from strong acid (present as in water). Because the acid is strong, treat this first step as going to completion before any equilibrium calculations.
Net ionic neutralisation:
Use moles (not concentrations) to decide what remains after the reaction.
Only after limiting-reactant stoichiometry do you consider buffer logic or acid–base equilibria.
Case 1: Excess weak base (buffer forms)
If, after the quantitative reaction, B remains and is produced, the solution contains a conjugate pair (B / ), so it behaves as a buffer.
Buffer — a solution containing substantial amounts of a conjugate acid–base pair that resists large pH changes when small amounts of acid or base are added.

Side-by-side comparison of buffered vs unbuffered solutions before and after a small addition of strong acid, using an indicator color change to show pH response. The buffered beaker stays essentially the same pH because the conjugate pair consumes added (or ), whereas the unbuffered solution’s pH shifts dramatically. Source
In this situation:
Remaining B can consume added .
Formed can consume added (indirectly, by donating protons to neutralise base).
The pH is controlled primarily by the ratio of conjugate base to conjugate acid, not by their absolute amounts (as long as both are present in meaningful quantities).
Use the Henderson–Hasselbalch equation with the conjugate acid and conjugate base B. Concentrations can be replaced by mole ratios if the solution volume is the same for both species.
= acidity measure of the buffer solution (unitless)
= for the weak acid (unitless)
= concentration of the weak base form (mol L)
= concentration of the conjugate acid form (mol L)
Key setup points (no arithmetic required here):
Determine post-reaction amounts of B and by stoichiometry.
Convert to concentrations if needed (divide by total volume).
The buffer assumption is strongest when neither component is extremely small.
Case 2: Excess strong acid (excess sets pH)
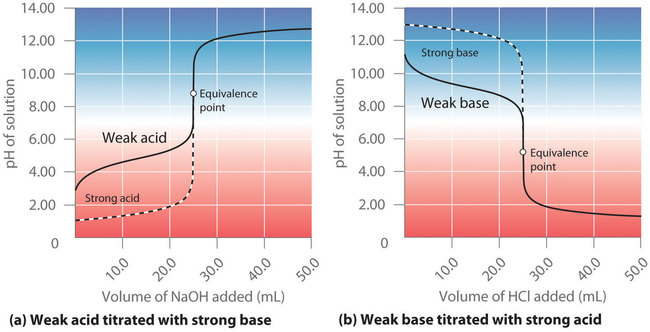
Titration curves comparing (a) weak acid + strong base and (b) weak base + strong acid, with the equivalence point labeled. The weak base/strong acid curve highlights a buffer region before equivalence (mixture of and ), an acidic equivalence point dominated by , and a low-pH region after equivalence where excess dictates pH. Source
If the strong acid provides more than the weak base can consume, then after neutralisation:
All B is consumed.
Excess remains, and this dominates pH.
In this case, treat the solution like a strong acid solution:
The contribution to from any weak acid chemistry of is negligible compared with the leftover strong acid.
Conceptually, pH depends on the remaining moles of divided by total volume.
Practical reasoning cues:
If the leftover strong acid is nonzero, do not use Henderson–Hasselbalch.
The mixture is not a buffer because there is no significant amount of conjugate base B remaining to neutralise added acid.
Case 3: Equimolar (no excess strong acid or weak base)
If moles of strong acid equal moles of weak base, the quantitative reaction consumes both completely:
No B remains and no excess remains.
The major solute is , the conjugate acid of the weak base.
The resulting solution is acidic because is typically a weak acid that can donate a proton to water (establishing an equilibrium). Conceptually:
pH is determined by the acid dissociation tendency of (its or , if provided).
This is not a buffer (only one member of the conjugate pair is present in significant amount).
Common pitfalls to avoid
Mixing up the order: do stoichiometry first, then decide whether the remaining system is buffer-controlled (excess base) or strong-acid-controlled (excess acid), or a conjugate-acid-only solution (equimolar).
Using Henderson–Hasselbalch when one component is essentially gone; without both B and , the buffer model does not apply.
Forgetting that “strong acid” means the reacting species in water is effectively , not undissociated acid molecules.
FAQ
Because the strong acid supplies effectively free $H_3O^+$, proton transfer to $B$ is highly favourable.
The equilibrium that makes $B$ “weak” concerns its reaction with water, not with a strong acid.
Check that both $B$ and $HB^+$ are present in non-negligible amounts after stoichiometry.
If one is tiny compared with the other, the buffer approximation becomes unreliable.
In very dilute solutions, water autoionisation can become comparatively important.
Activity effects (non-ideality) can also cause deviations from concentration-based predictions.
Count moles of transferable protons actually delivered as $H_3O^+$ equivalents.
For example, $1$ mole of a diprotic strong acid can supply up to $2$ moles of $H_3O^+$ if both protons fully dissociate.
Equimolar mixing leaves primarily $HB^+$, which can act as a weak acid in water.
Neutrality would require $HB^+$ to be negligibly acidic (very small $K_a$), which is not generally true.
Practice Questions
(2 marks) A solution contains a weak base and its conjugate acid . State the condition under which this mixture forms after adding a strong acid, and name the equation used to determine the pH.
Excess weak base remains after reaction with the strong acid / both and present (1)
Henderson–Hasselbalch equation (1)
(5 marks) of a weak base is mixed with of a strong acid. After mixing, there are more moles of than moles of . Explain, in terms of species present and appropriate method, how the pH is determined. You may assume for is given.
States the neutralisation is quantitative: (1)
Identifies limiting reactant is and that some remains; is formed (1)
Concludes a buffer is produced containing the conjugate pair (1)
States pH is found using Henderson–Hasselbalch with (1)
Notes that post-reaction amounts (then concentrations using total volume) supply the ratio in the equation (1)

