AP Syllabus focus: ‘Compare solution pH to pKa (or pKb) to predict protonation state: if pH < pKa the acid form dominates, and if pH > pKa the base form dominates.’
Predicting the predominant acid–base form of a species is a fast, conceptual skill that links equilibrium to structure and reactivity. It relies on comparing solution pH to tabulated (or ) values.
Core comparison: pH vs
For a conjugate acid–base pair written as , the solution pH tells you whether the species is mostly protonated () or deprotonated ().
Meaning of and “protonation state”
: A logarithmic measure of acid strength, defined as ; smaller means a stronger acid (more products at equilibrium).
In water, “protonated” generally means “has the extra ” relative to its conjugate partner (e.g., is protonated relative to ). The predominant form is the form present at the higher concentration under the stated conditions.
The central rule (from the syllabus)
If pH < , the acid form dominates: .
If pH > , the base form dominates: .
If pH = , the two forms are present in equal amounts: .
This comparison is powerful because it works without solving a full equilibrium problem when you only need the dominant species.
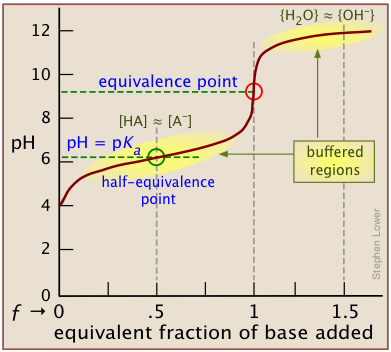
This titration-curve schematic for a weak acid titrated with strong base highlights the buffer region and explicitly marks the half-equivalence point where and therefore . It provides a graph-based way to see why the pH– comparison predicts which conjugate form predominates. Source
Quantifying “dominates” (ratio idea)
= concentration ratio (unitless)
= solution pH (unitless)
= acidity constant on the pH scale for (unitless)
A one-unit difference between pH and implies about a tenfold dominance; a two-unit difference implies about a hundredfold dominance. This helps justify calling one form “predominant” rather than merely “larger.”
Predominant form: The member of a conjugate pair present at greater concentration under the stated pH (often overwhelmingly greater when pH and differ by 1–2 units or more).
Using information (bases)
Some species are described as bases () with a conjugate acid (). The same “compare pH” logic applies if you have the of :
If pH < , the protonated base dominates: .
If pH > , the unprotonated base dominates: .
If you are given instead, interpret it as describing how strongly accepts a proton (stronger bases have smaller ). For predominance questions, you still make the core comparison by relating the given basicity information to whether the environment is relatively acidic (favors ) or basic (favors ).
Polyprotic species: multiple transitions
Molecules with more than one ionisable proton have multiple values and therefore multiple “switch points” for predominant form.
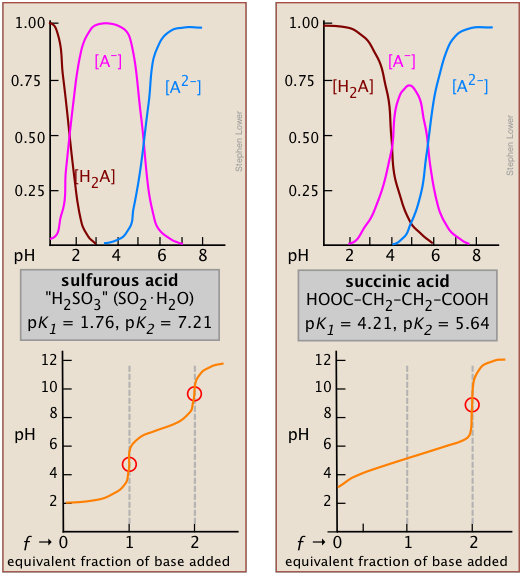
These species-distribution curves for diprotic acids show how the fractional amounts of , , and vary with pH, making the “switch points” associated with each visually explicit. The intermediate species is maximal between the two values, illustrating why polyprotic systems have distinct predominant forms across different pH ranges. Source
Each marks a pH region where one proton is mostly lost.
Between two successive values, an intermediate protonation state is typically predominant.
Below the lowest relevant , the most protonated form is favored; above the highest relevant , the most deprotonated form is favored.
FAQ
At $pH=pK_a$, the equilibrium condition corresponds to equal tendencies to donate and accept a proton for that conjugate pair.
Thermodynamically, this is the point where the chemical potentials balance so neither form is favoured over the other.
A common practical guideline is based on ratios:
1 pH unit away gives about a 10:1 ratio
2 pH units away gives about a 100:1 ratio
Depending on the context (analytical vs biological vs industrial), “effectively predominant” may require different ratio thresholds.
Yes. Tabulated $pK_a$ values typically assume dilute aqueous conditions. In higher ionic strength solutions, activities differ from concentrations, shifting the apparent balance.
The pH comparison still guides intuition, but boundaries can move slightly.
Yes. $pK_a$ values are solvent-dependent because solvent stabilisation of charged vs neutral forms changes.
A “given pH” also depends on solvent definition and measurement method, so aqueous-based intuition may not transfer directly to non-aqueous media.
$K_a$ (and thus $pK_a$) can vary with temperature because acid dissociation has an enthalpy change.
If temperature differs substantially from the conditions under which $pK_a$ was measured, the pH at which forms are equally present can shift.
Practice Questions
(2 marks) A weak acid has . In a solution of pH 3.8, which form is predominant, or ? Justify using the pH vs comparison.
Identifies as predominant (1)
Justification: pH < so acid/protonated form dominates (1)
(5 marks) Consider a diprotic acid with and . (a) At pH 1.0, state the predominant form: , , or . (1) (b) At pH 4.0, state the predominant form. (1) (c) At pH 9.0, state the predominant form. (1) (d) Explain, using comparisons to values, why the form in (b) is predominant. (2)
(a) (1)
(b) (1)
(c) (1)
(d) pH is above so first deprotonation favoured, but below so second deprotonation not favoured; therefore intermediate form dominates (2: one point for each comparison)

