AP Syllabus focus: ‘For accurate titrations, choose an acid–base indicator whose pKa is close to the pH at the equivalence point.’
Selecting an indicator is a matching problem: you want the indicator’s visible colour change to occur during the steep pH jump near the equivalence point, not before or after it.
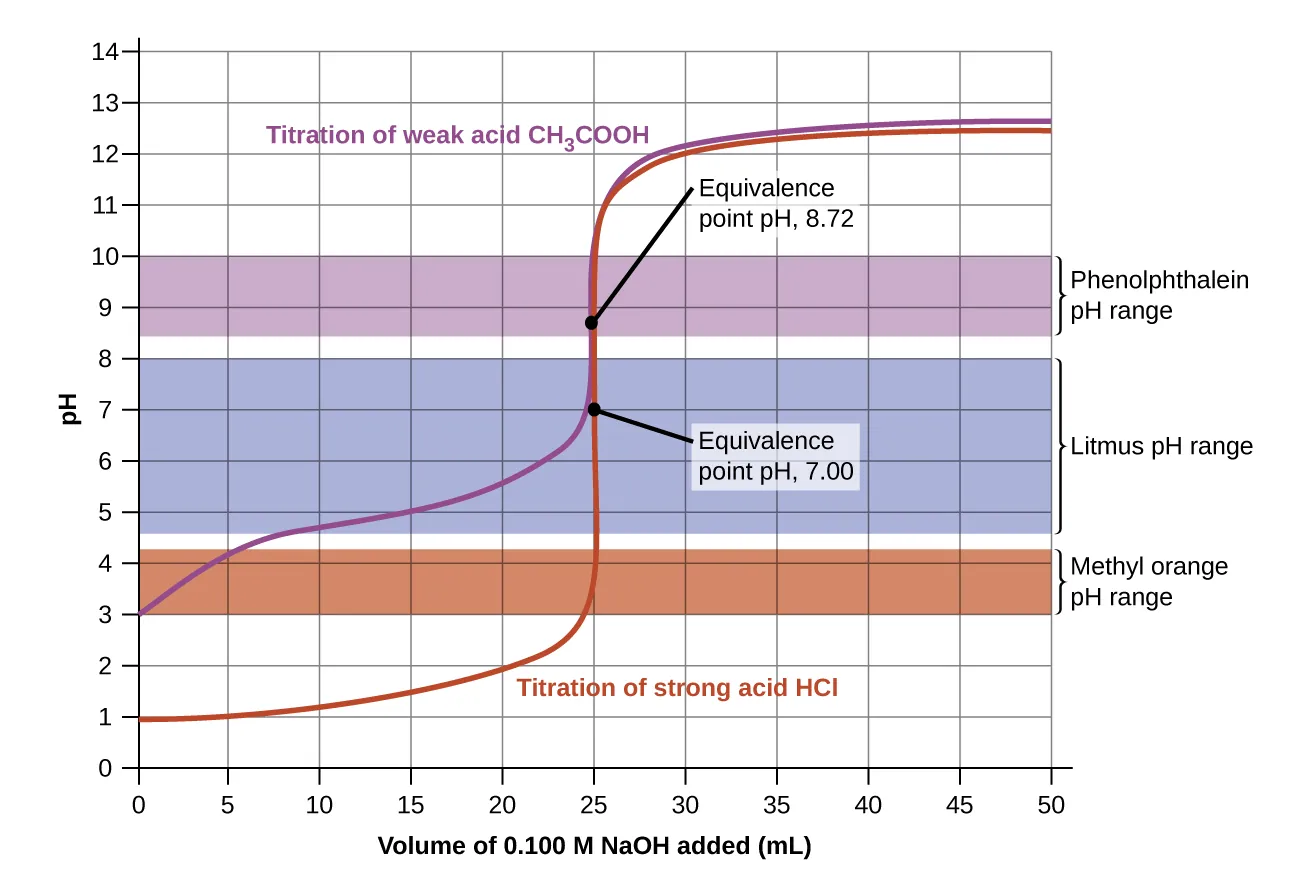
Titration curve with indicator transition ranges overlaid, illustrating how indicator choice must align with the steep pH change near the equivalence point. The horizontal bars represent each indicator’s color-change interval, making it clear why a well-matched indicator yields a sharper, more accurate endpoint for a given titration system. Source
Key ideas for indicator choice
What an indicator does
Acid–base indicator: a weak acid/base dye whose protonated and deprotonated forms have different colours; the observed colour depends on solution pH.
Indicators establish an equilibrium between two coloured forms, so their behaviour is governed by an acid dissociation constant and a pKa value (for the indicator’s acidic form).
Endpoint vs equivalence point
Endpoint: the point in a titration when the indicator’s colour change is observed; it is used to estimate the equivalence point, where stoichiometric amounts of acid and base have reacted.
The goal is a small indicator error, meaning the endpoint pH is as close as possible to the equivalence-point pH.
How pKa controls the colour-change window
An indicator changes colour when both forms are present in comparable amounts; practically, the human eye detects a transition over a pH range of about two units.
= solution pH (unitless)
= indicator’s acid dissociation pKa (unitless)
= concentration of deprotonated indicator form (mol L)
= concentration of protonated indicator form (mol L)
From this relationship:
When , (midpoint colour).
A noticeable colour shift typically occurs as the ratio moves from about 1:10 to 10:1, corresponding roughly to .
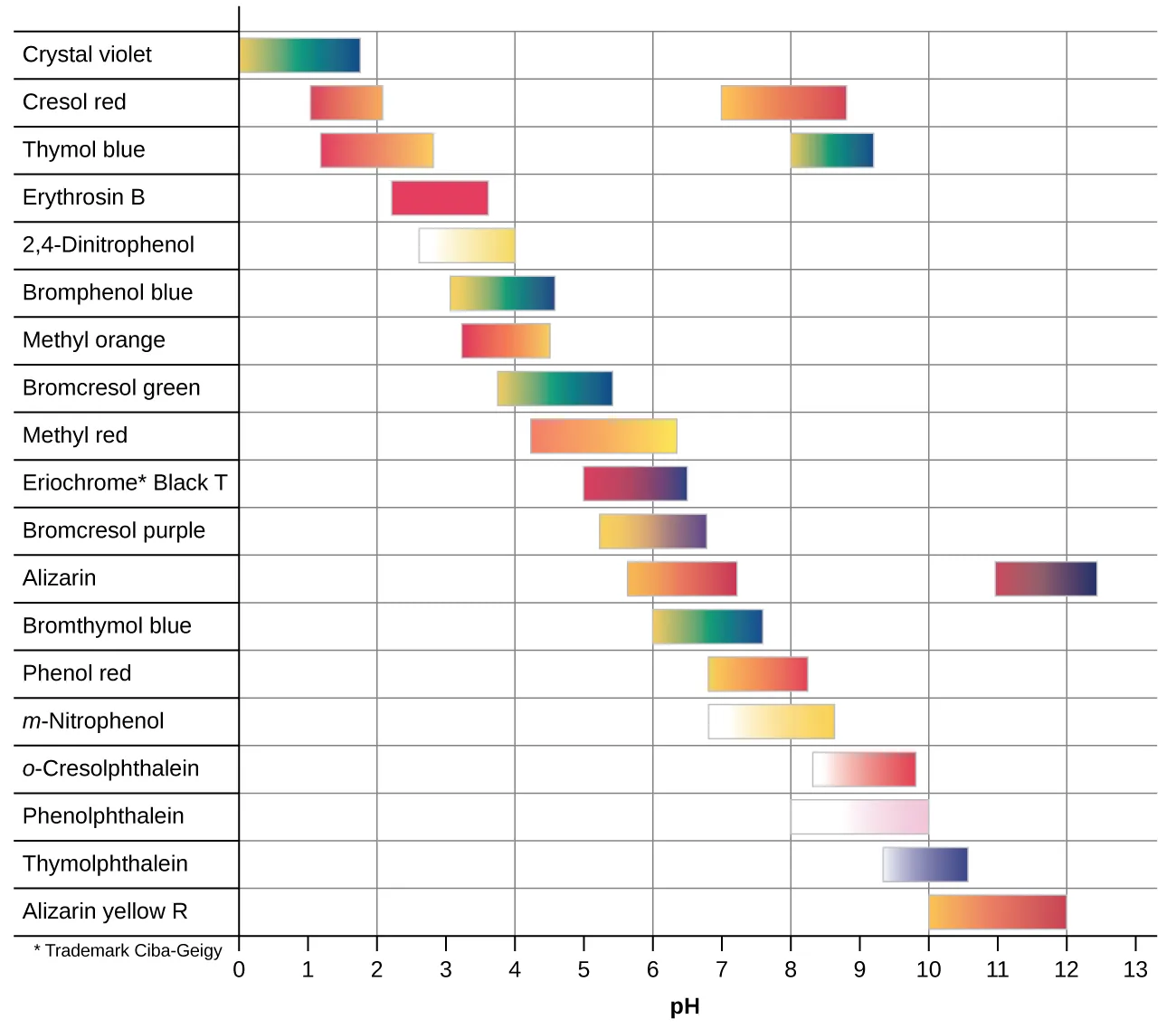
Color-change interval chart for common acid–base indicators, showing the approximate pH ranges over which each indicator transitions between its acidic and basic colors. This visual emphasizes that an indicator does not change color at a single pH value, but across a window that is typically about . Source
Practical rule for choosing an indicator
Match pKa to the equivalence-point pH
The syllabus requirement is the central selection rule:
Choose an indicator whose pKa is close to the pH at the equivalence point.
More precisely, aim for the equivalence-point pH to fall within the indicator’s transition range (approximately ), so the colour change happens during the steepest part of the titration curve.
Why “close” matters experimentally
An indicator is most accurate when a very small added volume of titrant carries the solution through the indicator’s transition range. If the indicator’s pKa is mismatched:
The colour change can occur too early (systematic under-titration).
Or too late (systematic over-titration). Either case produces a larger difference between endpoint and equivalence point, increasing uncertainty in calculated concentrations.
Additional selection considerations (secondary to pKa matching)
Prefer indicators with a sharp, distinct colour contrast in the relevant pH region.
Avoid indicators whose transition colour is hard to detect in the sample matrix (e.g., strongly coloured analytes).
Use only a small amount of indicator; although usually negligible, excessive indicator can slightly perturb pH because it is itself a weak acid/base.
FAQ
The indicator exists as a mixture of two coloured forms whose ratio varies continuously with pH.
Perception depends on human vision and lighting, so a “single pH” change is seen as a range.
The analyte’s colour can mask the indicator’s intermediate hues.
Choose an indicator with a high-contrast colour pair, or one whose colours differ strongly in intensity against the sample background.
Yes. Indicator $K_a$ (and therefore $pK_a$) can shift with temperature.
If the titration is performed far from room temperature, the indicator’s effective transition range may move, so a different indicator may be preferable.
Indicators are weak acids/bases; adding too much can slightly change the solution’s pH and broaden the endpoint.
Small volumes minimise any chemical disturbance and make the endpoint sharper.
If the solution is opaque, highly coloured, or the endpoint colour change is ambiguous, a pH meter avoids subjective judgement.
It also helps when very high precision is required or when the endpoint is difficult to observe consistently.
Practice Questions
(2 marks) Explain which property of an acid–base indicator you use to choose a suitable indicator for a titration, and state the criterion for an accurate endpoint.
Mentions indicator (or transition range linked to ). (1)
States indicator should be close to the pH at the equivalence point (or equivalence pH lies within transition range). (1)
(5 marks) An indicator HIn has , where HIn is one colour and is a different colour.
(a) Using , determine the pH at which . (2 marks)
(b) Hence state the approximate pH interval over which this indicator changes colour. (1 mark)
(c) For best accuracy, where should the equivalence-point pH lie relative to this interval? (2 marks)
(a) Substitutes ratio 10: . (1)
(a) Calculates . (1)
(b) States transition range approximately : about to . (1)
(c) Equivalence-point pH should fall within the transition interval. (1)
(c) Best when equivalence-point pH is near the midpoint (close to ) so endpoint aligns closely. (1)

