AP Syllabus focus: ‘In a buffer, the conjugate acid consumes added base and the conjugate base consumes added acid; these reactions are responsible for stabilizing pH.’
Buffers resist sharp pH changes because they contain chemical “sponges” for both added acid and added base. Their stabilizing action comes from fast, stoichiometric neutralisation reactions that preserve the solution’s overall acid–base balance.
What it means for a buffer to stabilise pH
A buffer works by converting a strong acid or strong base added to the solution into a much weaker species, so the hydronium/hydroxide levels change only slightly.
Buffer: A solution that contains substantial amounts of a weak acid and its conjugate base (or a weak base and its conjugate acid) and therefore resists changes in pH when small amounts of acid or base are added.
The key idea in the syllabus statement is role-sharing within a conjugate pair:
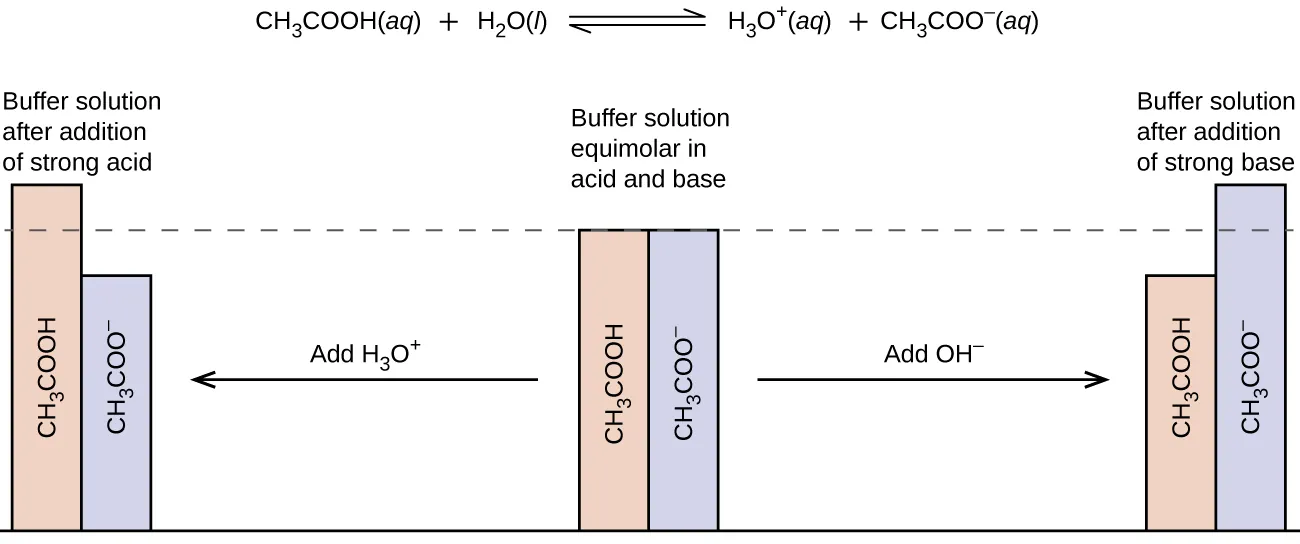
Bar-graph schematic of buffer action in an acetic acid/acetate system. It shows that adding strong acid () increases the weak-acid form while decreasing the conjugate base, and adding strong base () does the opposite—illustrating why buffers convert strong acid/base into weaker conjugate partners. Source
The conjugate acid component reacts with (consumes) added base.
The conjugate base component reacts with (consumes) added acid.
The conjugate pair as “reservoirs”
Because both members of the conjugate pair are present in large amounts relative to what is added (in typical buffer problems), the added acid/base is “used up” primarily by reaction with the buffer components rather than by directly changing the solution’s pH.
How a buffer responds when acid is added
When a strong acid is added, it increases . In a buffer containing a weak acid and its conjugate base , the conjugate base removes most of the added by forming .
This consumes added acid (removes ).
It increases the amount of and decreases the amount of .
The pH changes only modestly because the strong acid is converted into the weak acid form already present in the buffer system.
Chemically, the added is not left “free” in solution for long; it is incorporated into the weak-acid/conjugate-base system.
Why the pH change is limited (qualitative)
Even though some is converted to , both species remain present, so the solution still behaves like a buffer after the addition (unless too much acid is added). The buffer therefore resists a dramatic rise in .
How a buffer responds when base is added
When a strong base is added, it increases . In the same buffer, the conjugate acid neutralises most of the added to form water while producing more .
This consumes added base (removes ).
It decreases the amount of and increases the amount of .
The pH changes only modestly because the strong base is converted into the weak base form that belongs to the buffer pair.
This matches the syllabus statement directly: the conjugate acid is the component responsible for reacting with added base.
= weak acid component of the buffer
= conjugate base component of the buffer
= added base that is consumed by the buffer
= added acid that is consumed by the buffer
These reactions are typically fast and proceed essentially to completion compared with the slower equilibrium ionisation of the weak acid/base itself.
What is (and is not) changing during buffering
Buffering does not mean “nothing reacts.” Instead, the buffer channels the disturbance into changing the amounts of and rather than allowing a large change in or .
Key chemical consequences of each addition
Adding acid:
Major event: is converted to
Direct effect opposed: increase in
Adding base:
Major event: is converted to
Direct effect opposed: increase in
Limits of buffer stabilisation (conceptual)
A buffer can only stabilise pH while it still contains meaningful amounts of both conjugate partners.
If too much acid is added, most is consumed; the solution can no longer effectively remove additional .
If too much base is added, most is consumed; the solution can no longer effectively remove additional .
The stabilising mechanism is therefore inherently tied to the presence of both members of the conjugate acid–base pair, exactly as described in the syllabus focus statement.
FAQ
The buffer components are already present at appreciable concentrations, so reactions like $\mathrm{A^- + H_3O^+ \rightarrow HA + H_2O}$ proceed rapidly via frequent collisions.
Equilibrium re-establishes afterwards, but the initial consumption of the added strong acid/base is typically the dominant immediate effect.
Most added $\mathrm{H_3O^+}$ is transferred to the conjugate base (a proton-transfer event).
Water remains the solvent and mediator, but the net result is protonation of the base component rather than a persistent rise in free $\mathrm{H_3O^+}$.
Yes. Dilution lowers the concentrations of both conjugate partners, so a given amount of added acid/base represents a larger fraction of what is available to react.
As a result, the same addition causes a larger relative change in the buffer composition.
Because the conjugate base is finite. Once most $\mathrm{A^-}$ has been converted into $\mathrm{HA}$, there is little $\mathrm{A^-}$ left to consume further $\mathrm{H_3O^+}$.
At that point, added acid accumulates and pH drops sharply.
They usually do not participate directly in the neutralisation reactions, but they can affect ionic strength.
Changes in ionic strength can slightly alter activity (effective concentration), causing small real-world deviations from ideal buffer behaviour.
Practice Questions
(2 marks) A buffer contains and . State which component reacts when a small amount of strong acid is added, and state what it is converted into.
Identifies (conjugate base) reacts with added acid (1)
States is converted to (1)
(5 marks) A buffer is made from a weak acid and its conjugate base . Describe, using chemical equations, how the buffer minimises the pH change when (i) is added and (ii) is added. Explain in words why these reactions stabilise pH.
Equation showing consuming added (or added ) to form , e.g. (1)
States conjugate base removes most added acid (1)
Equation showing consuming added to form , e.g. (1)
States conjugate acid removes most added base (1)
Explains that converting strong acid/base into the weak-conjugate partner limits the change in or , hence limiting pH change (1)

