AP Syllabus focus: ‘A buffer contains large concentrations of both members of a conjugate acid–base pair.’
Buffers are defined by what is present in solution, not by a procedure. This page focuses on the required composition of a buffer and how to recognise whether a mixture qualifies as one.
Core idea: what a buffer is made of
A solution is a buffer when it contains both members of a conjugate acid–base pair in large concentrations (relative to the amount of acid or base you might add).
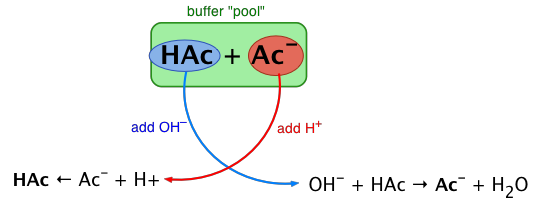
Diagram of a buffer “reservoir” built from a conjugate acid–base pair (e.g., weak acid and its conjugate base) that consumes added or . The visual emphasizes why buffering requires substantial amounts of both partners present simultaneously, so additions shift the balance without a large pH change. Source
Buffer: a solution that contains substantial amounts of a conjugate acid–base pair and therefore has the capacity to resist large pH changes upon small additions of acid or base.
The syllabus emphasis is compositional: you must be able to identify whether both conjugate partners are present together in appreciable amounts.
Conjugate acid–base pairs (the required “two components”)
Conjugate acid–base pair: two species that differ by exactly one proton, (for example, or ).
In a buffer, both species must be present at the same time in solution:
Weak acid buffer: (weak acid) and (its conjugate base)
Weak base buffer: (weak base) and (its conjugate acid)
Recognising buffer components in common mixtures
Typical buffer recipe patterns (composition only)
A buffer is commonly prepared by combining:
A weak acid with a soluble salt containing its conjugate base (source of ), or
A weak base with a soluble salt containing its conjugate acid (source of )
The salt is used because it provides the conjugate partner directly and in significant concentration.
= weak acid component (aqueous)
= conjugate base component (aqueous)
= hydronium formed by acid ionisation (aqueous)
This equilibrium is relevant because it shows the paired species ( and ) associated with a weak acid system; a buffer requires that both are already present in large concentrations, not just produced in trace amounts.
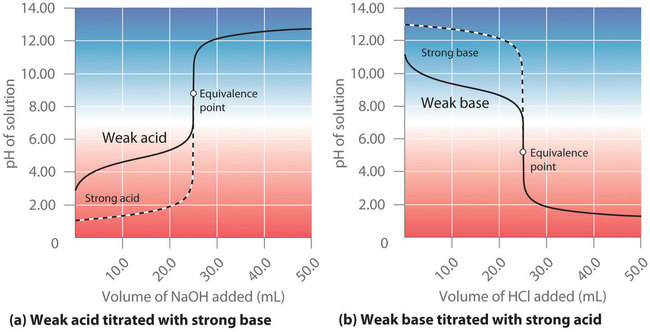
Titration-curve plot comparing a weak acid (acetic acid) titrated with strong base to a strong acid titration. The weak-acid curve shows a broad, gently sloped region where both and are present together—corresponding to effective buffering before the equivalence point. Source
What “large concentrations” means in practice
To function as a buffer (by the syllabus criterion), the solution must contain:
Non-negligible amounts of both conjugate partners
Often comparable order of magnitude amounts (neither component should be essentially absent)
If one component is present only in a tiny amount, the mixture is better described as a weak acid (or weak base) solution rather than a buffer.
Common non-buffers (mixtures that fail the definition)
A mixture does not qualify as a buffer if it lacks one member of the conjugate pair in significant concentration. Examples of why a solution may fail:
Only a weak acid present (mostly , very little ): not a buffer by composition.
Only a weak base present (mostly , very little ): not a buffer by composition.
Strong acid with its conjugate base (e.g., ): is an extremely weak base and does not create a meaningful conjugate pair buffer system.
Strong base with its conjugate acid (e.g., ): there is no suitable weak conjugate acid partner present as a stable component.
Two species that are not conjugates (do not differ by exactly one ): cannot form the required pair.
Checklist: does this solution meet the syllabus definition?
Use this rapid identification checklist:
Identify a candidate pair that differs by one proton: or .
Confirm both species are present in solution (not just theoretically possible).
Confirm both are present in large concentrations (neither is essentially zero).
FAQ
Yes. Choose two adjacent species that differ by one proton (e.g., $H_2A/HA^-$ or $HA^-/A^{2-}$).
Only one conjugate pair is considered at a time for identifying a buffer.
Weak species ionise only partially, so the conjugate partner formed may be too small in concentration.
A soluble salt supplies the conjugate partner directly and substantially.
Dilution lowers concentrations of both components.
If both remain “large” relative to expected additions, it still qualifies; if too dilute, it may no longer meet the “large concentrations” criterion.
Not unless one is the conjugate base of the other (i.e., they differ by one $H^+$).
Two unrelated weak acids do not constitute a conjugate pair.
Compare formulas and charge:
Conjugates differ by exactly one $H$ atom and the charge differs by $+1$ (acid form) versus base form.
Examples: $HCO_3^-/CO_3^{2-}$, $H_2PO_4^-/HPO_4^{2-}$.
Practice Questions
A student mixes with . State whether the resulting solution is a buffer and justify your answer.
States it is a buffer (1)
Justifies: contains large concentrations of a conjugate acid–base pair, (1)
For each mixture below, state whether it forms a buffer. If it does, identify the conjugate acid–base pair present. If it does not, give one reason based on composition.
(i) and
(ii) and
(iii) only
(i) Buffer (1); identifies pair (1)
(ii) Not a buffer (1); reason: involves strong acid, conjugate base is too weak / not a weak acid–base conjugate pair buffer system (1)
(iii) Not a buffer (1); reason: lacks large concentration of conjugate base (or only one member present) (1)
(Max 5; award best-supported points.)

