AP Syllabus focus: ‘Calculate standard entropy change with ΔS°rxn = ΣS°(products) − ΣS°(reactants), including coefficients from the balanced equation.’
Entropy changes in reactions are calculated from tabulated standard molar entropy values and the balanced equation.
Stoichiometry matters: every coefficient scales the entropy contribution of that species in the overall reaction.
What you are calculating: ΔS°rxn from tabulated S°
To find the standard entropy change of reaction, you combine the absolute entropies of all products and reactants, weighted by stoichiometry.
Standard reaction entropy change, : the entropy change when the reaction occurs as written, with all species in their standard states, using tabulated values.
Tabulated values are typically given per mole of a substance, so a reaction producing 2 moles of a product counts that product’s entropy twice.
Core relationship (products minus reactants, with coefficients)
The calculation is a sum of “how much entropy is present in the products” minus “how much entropy is present in the reactants,” using the balanced chemical equation as the map for mole ratios.
= standard reaction entropy change,
= standard molar entropy of a species (from a table),
= stoichiometric coefficient from the balanced equation (dimensionless)
This relationship is purely additive: you do not apply exponents, equilibrium ideas, or rate concepts—only stoichiometric scaling and subtraction.
Step-by-step stoichiometric procedure
1) Balance the chemical equation first
Ensure atoms and charge (if ionic) are balanced.
The coefficients you obtain are the only multipliers used in the entropy sum.
2) Collect the correct values for every species
Use the value that matches the species’ chemical identity and physical state (e.g., H₂O(l) vs H₂O(g)).
Include all reactants and products shown in the net equation, including pure solids and liquids (they still have nonzero ).
3) Multiply each by its coefficient
Treat each term as .
If a coefficient is 1, it is still conceptually included.
4) Add products and add reactants, then subtract
Compute .
Compute .
Subtract: products minus reactants, exactly as written in the equation.
What must match the balanced equation (common stoichiometric logic)
Coefficients scale entropy contributions
If you double an entire reaction equation, doubles (because all values double).
If you reverse a reaction, changes sign (because products and reactants swap).
Do not confuse coefficients with subscripts
A subscript is part of a substance’s formula (e.g., O₂); it does not represent “2 moles” unless the balanced equation coefficient is 2.
Only the balanced-equation coefficients multiply .
Species, phases, and omission rules
Physical states are part of the species
Using the wrong phase gives the wrong and therefore the wrong .
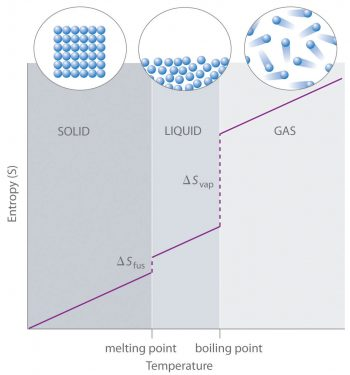
Generalized plot of entropy versus temperature showing gradual increases within a phase and abrupt jumps at melting and boiling. The step changes ( and ) visually explain why gases typically have much larger molar entropies than liquids and solids, making phase labels essential in table lookups. Source
For aqueous ions, use the tabulated entropy for that aqueous ion, not the solid salt.
“Spectator” ions are not automatically removed unless you write a net ionic equation
If you are given a molecular equation, calculate using that equation as written.
If you are instructed to use a net ionic equation, then only include species present in that net reaction.
Units follow from the table
Because values are typically in , will be in the same units.
Keep unit consistency if a table uses (convert if needed).
FAQ
$S^\circ$ is an absolute entropy on a scale set by the Third Law, not a relative “formation” quantity.
Only a perfect crystal at $0\ \rm K$ has $S=0$ ideally; at $298\ \rm K$, all substances have accessible microstates, so $S^\circ>0$.
Only when the question explicitly asks for the net ionic reaction or when it clearly describes the reaction occurring at the ionic level.
Otherwise, use the equation provided, because it defines which species’ entropies are included.
Not for typical thermodynamics tables used in AP Chemistry.
Electrons appear in half-reactions for bookkeeping, but standard molar entropies are ordinarily tabulated for chemical species (atoms, molecules, ions), not free electrons.
Treat the hydrate as its own species and use the tabulated $S^\circ$ for that exact formula and phase when available.
Do not split it into “salt + water” unless the problem explicitly gives a decomposition reaction and relevant $S^\circ$ values.
Keep guard digits through the sum/subtraction, then round at the end to match the least precise decimal place in the given $S^\circ$ data.
Report units explicitly as $\rm J,mol^{-1},K^{-1}$ (or converted units if required).
Practice Questions
(2 marks) For , write the expression for in terms of values, including coefficients.
1 mark: Correct structure “products − reactants”.
1 mark: Correct coefficients: .
(5 marks) Calculate for given: , , in .
1 mark: Uses .
1 mark: Multiplies by coefficients (2 for NH, 3 for H).
1 mark: Correct products total: .
1 mark: Correct reactants total: .
1 mark: Correct subtraction and units: .

