AP Syllabus focus: ‘Report ΔS° with correct units and sign; ensure you multiply S° values by coefficients and compare products to reactants.’
Entropy change calculations are often straightforward, but interpretation is where many errors occur. This page focuses on reporting correctly, reading its sign meaningfully, and avoiding the most common scoring pitfalls.
What you are reporting: sign and units
Standard entropy change, : the entropy change for a reaction as written (per “mole of reaction”) when all species are in their standard states.
Because entropies measure energy/matter dispersal, the sign of communicates the direction of dispersal in the reaction as written, not whether the reaction “happens fast” or “goes to completion.”
= standard entropy change for the reaction as written,
= stoichiometric coefficient from the balanced equation, unitless
= standard molar entropy of a species,
Units: what to write and what not to write
Correct unit conventions
Standard molar entropy values, , are typically tabulated in .
Therefore, is also reported in .
Use Kelvin in the unit: writing is not acceptable.
Common unit pitfalls
Mixing kJ and J: entropy tables are usually in J, so don’t report in kJ/K unless you explicitly convert and label it consistently.
Dropping the “per mole” idea: is per mole of reaction (based on the balanced equation), so the must remain in the unit.
Sign: how to interpret positive vs negative
Meaning of the sign (interpretation language)
: products have greater total entropy than reactants; dispersal/number of accessible microstates is higher for the reaction as written.
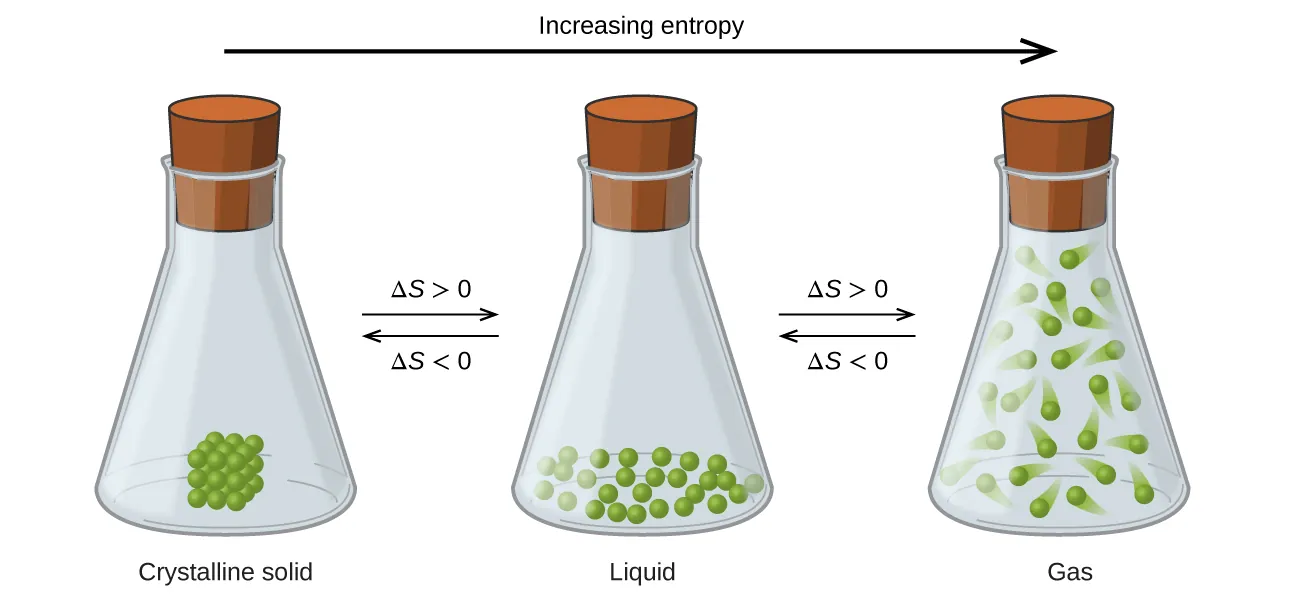
Phase-based entropy comparison showing increasing entropy from crystalline solid to liquid to gas, with arrows indicating when is positive versus negative. This is a quick conceptual check: reactions that produce gas or convert condensed phases into gas often have , while the reverse tends to yield . Source
: products have lower total entropy; the reaction as written results in less dispersal.
Sign pitfalls that lose points
Confusing the subtraction order: it must be products − reactants. Reversing it flips the sign.
Forgetting that reversing the chemical equation reverses the sign: if you write the reaction backward, changes sign.
“Multiply by coefficients” and other stoichiometry traps
Coefficients are not optional
Every tabulated must be multiplied by its stoichiometric coefficient before summing.
If you later scale the entire balanced equation (e.g., double it), scales by the same factor because it is extensive.
Physical-state mismatches
Use the correct species and phase that appears in the reaction: S^\circ$ by its coefficient?
Did you use the correct phases?
FAQ
$S^\circ$ values are absolute entropies and are positive, but $\Delta S^\circ_{\text{rxn}}$ is a difference between totals.
A decrease in total entropy is entirely possible, so the difference can be negative.
Report $\Delta S^\circ_{\text{rxn}}$ per the reaction as written, even if coefficients are fractional.
If you later multiply the equation to clear fractions, $\Delta S^\circ_{\text{rxn}}$ must be multiplied by the same factor.
Only if you clearly indicate you are reporting the entropy change for a specific stated amount of reaction, not per mole.
On AP-style reporting for tabulated standard data, include $\text{mol}^{-1}$.
Match the least precise decimal place implied by the tabulated $S^\circ$ data and your arithmetic.
Avoid over-rounding mid-calculation; round at the end to maintain the correct sign and magnitude.
Not by itself.
Spontaneity depends on Gibbs free energy, which involves both entropy and temperature; $\Delta S^\circ_{\text{rxn}}$ alone is not a standalone criterion.
Practice Questions
Question 1 (1–3 marks) A student reports for a reaction after using tabulated values given in . State two issues with how the result is presented.
1 mark: Identifies unit inconsistency (used in J but reported in kJ without stating/doing a conversion).
1 mark: Corrects/requests correct unit format for entropy (e.g., or consistent conversion to ).
1 mark: Notes appropriate significant figures/clarity (e.g., convert to or clearly show conversion).
Question 2 (4–6 marks) For the reaction , the tabulated values are: , , . (a) Calculate with units. (b) A classmate subtracts “reactants − products” and obtains the opposite sign. Explain why that is incorrect. (c) State one additional common mistake (other than subtraction order) that would change the numerical value.
(a) 1 mark: Uses coefficients: products , reactants .
(a) 1 mark: Correct subtraction products − reactants and correct arithmetic to obtain .
(a) 1 mark: Correct units .
(b) 1 mark: States definition requires products − reactants; reversing flips the sign and corresponds to the reverse reaction.
(c) 1–2 marks: One valid pitfall, e.g. forgetting to multiply by 3 for , using wrong species/phase, or mixing J and kJ.

