AP Syllabus focus: ‘Compute standard cell potential by identifying oxidation and reduction half-reactions and using their standard reduction potentials.’
Electrochemical tables list standard reduction potentials for half-reactions. To calculate , you decide which half-reaction is oxidised and which is reduced, then combine the tabulated values with correct sign conventions.
Standard reduction potentials (what the table gives you)
Standard reduction potential (): the potential for a half-reaction written as a reduction, measured relative to the standard hydrogen electrode under standard conditions (solutes 1.0 M, gases 1.0 bar, pure solids/liquids).
Standard reduction potential tables are always written as reductions (electrons on the reactant side). A more positive indicates a greater tendency for that reduction to occur.
Steps to calculate from a reduction potential table
1) Identify the two half-reactions actually occurring
Determine which species is oxidised (loses electrons) and which is reduced (gains electrons).
Choose the corresponding half-reactions from the table (they may need to be matched to the species present, including phases like (s), (aq), (g)).
2) Assign anode and cathode by process (not by sign labels)
Oxidation occurs at the anode.
Reduction occurs at the cathode. When using a reduction table, you will use one listed reduction as-is (at the cathode) and reverse the other to represent oxidation (at the anode).
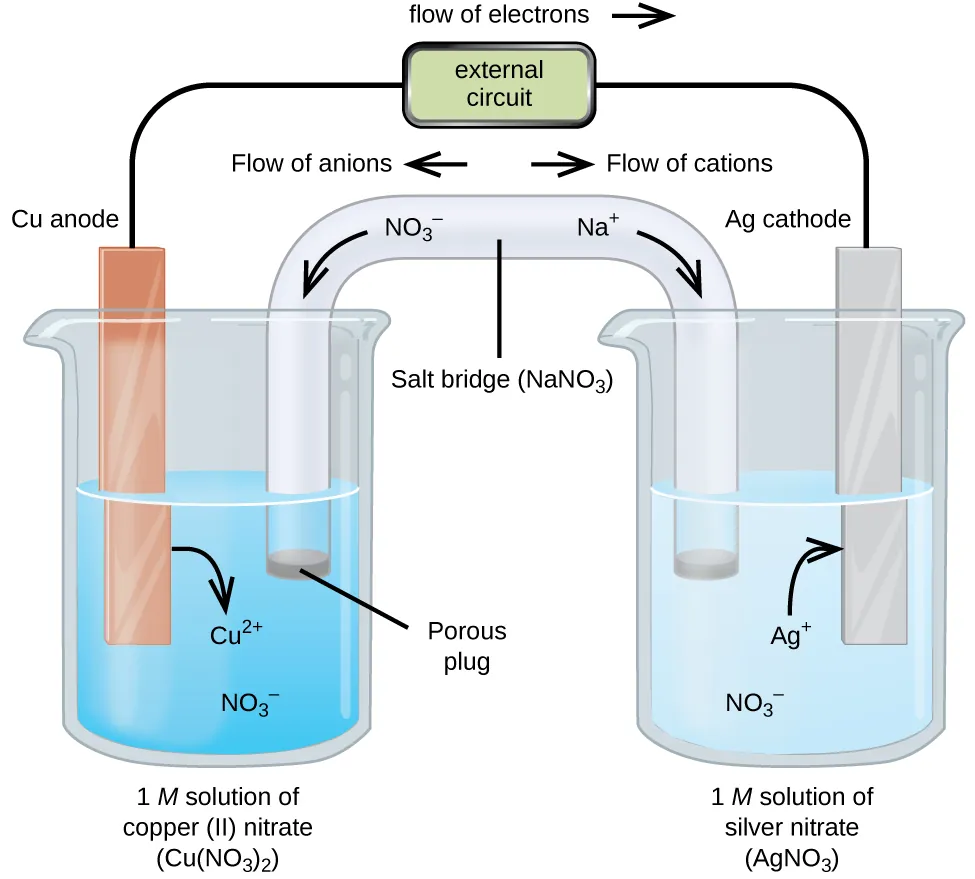
A labeled galvanic cell diagram showing that oxidation occurs at the anode, reduction occurs at the cathode, and electrons flow through the external wire from anode to cathode. It also depicts ion migration in the salt bridge that maintains charge neutrality in each half-cell during operation. Source
3) Use reduction potentials with the correct arithmetic
= standard cell potential (V)
= reduction potential for the cathode half-reaction as written in the table (V)
= reduction potential for the anode half-reaction as written in the table (V)
This subtraction method works because the anode process is oxidation, but the table value you looked up for that half-cell is still a reduction potential.
4) Balance electrons without changing values
You must balance electrons when combining half-reactions to write the overall redox reaction.
Do not multiply values by stoichiometric coefficients; potentials are intensive (they do not scale with amount).
Common reasoning checks (to avoid sign mistakes)
Choosing which half-reaction is reduced
The cathode is typically the half-reaction with the more positive (greater reduction tendency).
The anode corresponds to the other half-reaction; it runs in reverse as oxidation, but you still plug in its tabulated reduction value into the equation.
Interpreting the computed sign
A correctly assembled standard (galvanic) cell should give when the reaction direction matches the spontaneous direction under standard conditions.
If you obtain a negative value, it usually means you swapped anode/cathode for the intended reaction direction.
State matters
Make sure you use the correct half-reaction for the correct phase and ion charges (for example, vs are different couples with different values).
FAQ
Potentials measure energy change per unit charge (driving force), so they are intensive.
Multiplying a half-reaction changes moles of electrons, not the “per charge” tendency reflected by $E^\circ$.
Look up the corresponding reduction (reverse direction) to obtain $E^\circ_{\text{red}}$.
Then use subtraction: $E^\circ_{\text{cell}} = E^\circ_{\text{cathode}} - E^\circ_{\text{anode}}$ (both from the reduction table).
Check for the correct oxidation state, phase, and whether the table lists an equivalent couple.
If multiple couples exist (e.g., $Fe^{3+}/Fe^{2+}$ vs $Fe^{2+}/Fe$), choose the one matching the actual species present.
No. Inert electrodes provide a surface for electron transfer but do not set the redox potential.
$E^\circ_{\text{cell}}$ depends on the redox couples and their standard reduction potentials.
By pairing the half-cell with the standard hydrogen electrode ($2H^+ + 2e^- \rightleftharpoons H_2$) defined as $0.00\text{ V}$ under standard conditions.
The measured voltage vs SHE gives the half-cell’s $E^\circ_{\text{red}}$.
Practice Questions
(2 marks) Using a table of standard reduction potentials, state how you would calculate once you have identified the anode and cathode half-reactions.
Uses the relationship with values taken as reduction potentials from the table. (1)
Makes clear that the anode value used is the tabulated reduction potential (even though oxidation occurs at the anode). (1)
(5 marks) Given the half-cells and and a standard reduction potential table, describe the method to obtain for the standard cell, including identification of oxidation/reduction and correct use of tabulated values. Do not perform any numerical calculation.
Identifies that one half-reaction must be oxidation (anode) and the other reduction (cathode). (1)
States that the table provides reduction half-reactions and values. (1)
Selects as the cathode reduction and as the anode (oxidised) based on relative values (more positive is reduced). (1)
Uses . (1)
States that values are not multiplied when balancing electrons/stoichiometry. (1)

