AP Syllabus focus: ‘Standard Gibbs free energy change is proportional to the negative of cell potential: ΔG° = −nFE°. Positive E° implies a favored reaction; negative E° implies unfavored.’
Electrochemical cells connect thermodynamics to measurable voltage. The key idea is that the maximum electrical work a redox reaction can deliver is directly related to its Gibbs free energy change under standard conditions.
Linking voltage to thermodynamic driving force
A cell potential (voltage) reflects how strongly electrons are pushed from the species being oxidized to the species being reduced.
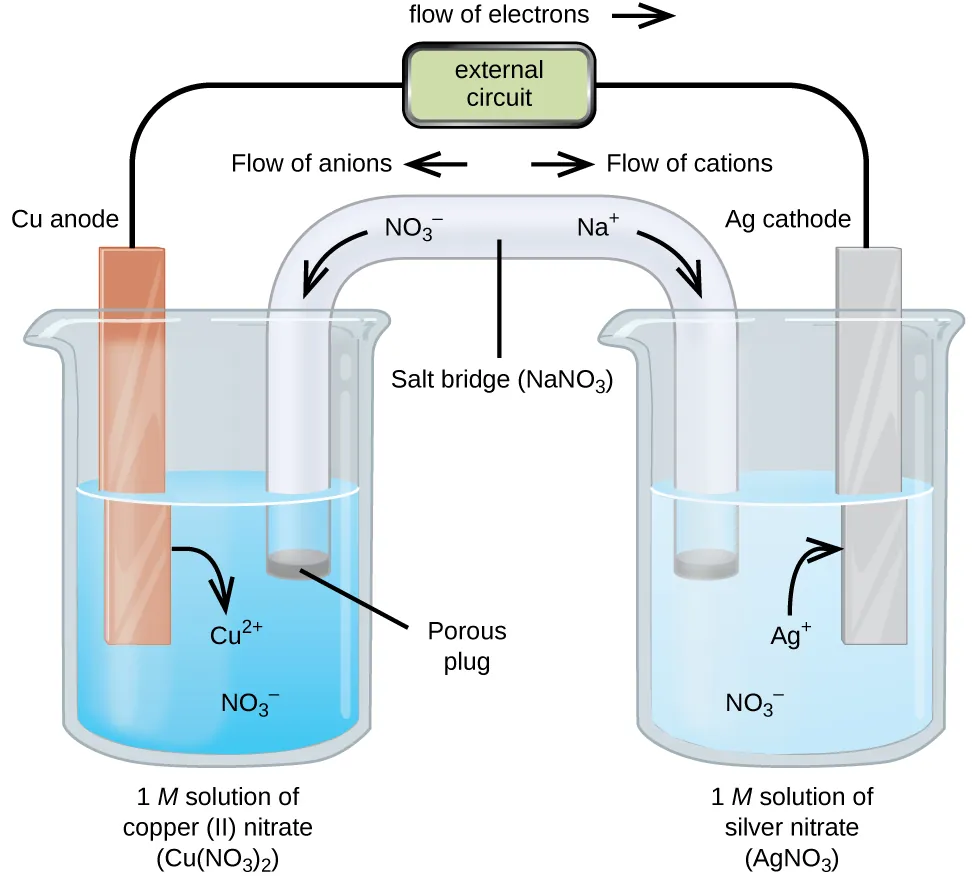
Labeled galvanic (voltaic) cell diagram showing the anode/cathode, the direction of electron flow in the external circuit, and compensating ion flow through the salt bridge. This directly connects the sign of to the direction a spontaneous redox process drives charge movement. Source
Thermodynamics describes that “push” as a change in Gibbs free energy, which predicts whether a process is thermodynamically favored under specified conditions.
In an electrochemical cell operating reversibly, the system can convert chemical energy into electrical energy. The thermodynamic quantity that tracks the maximum possible useful (non-expansion) work is the Gibbs free energy change. For an electrochemical cell, the useful work is electrical work, so voltage and free energy are linked.
Key quantities you must identify correctly
E°cell is the standard cell potential: the potential difference for the overall balanced cell reaction when all species are in their standard states.
n is the number of moles of electrons transferred in the balanced overall redox reaction (electrons cancel in the final overall equation, but n is taken from the half-reactions).
F is the Faraday constant: charge per mole of electrons, which bridges chemistry (mol e−) and electricity (coulombs).
A common source of errors is mixing “per mole of reaction as written” with “per mole of electrons.” The value of n ensures the free energy change corresponds to the balanced reaction stoichiometry you are using.
Standard Gibbs free energy change (ΔG°): the free energy change for a reaction as written when all reactants and products are in their standard states; it indicates thermodynamic favorability under those conditions.
The sign relationship is the high-utility takeaway: a positive standard cell potential corresponds to a negative standard Gibbs free energy change.
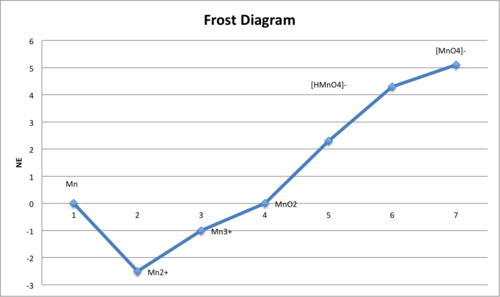
Example Frost diagram (for manganese) plotting oxidation state vs. , a vertical scale proportional to . The slopes between points correspond to standard potentials, making the connection between electrode potentials and Gibbs free-energy changes visually explicit. Source
= standard Gibbs free energy change per mole of reaction as written (J mol)
= moles of electrons transferred per mole of reaction (mol e)
= Faraday constant, C mol e
= standard cell potential (V = J C)
Interpreting sign: favored vs. unfavored under standard conditions
The equation contains a built-in negative sign, so ΔG° and E°cell always have opposite signs (when n and F are positive, which they are by definition).
If E°cell > 0, then ΔG°rxn < 0
The reaction is thermodynamically favored under standard conditions.
The cell can, in principle, do electrical work on the surroundings without an external power source.
If E°cell < 0, then ΔG°rxn > 0
The reaction is thermodynamically unfavored under standard conditions.
Driving it as written would require an externally applied potential (consistent with an electrolytic direction).
If E°cell = 0, then ΔG°rxn = 0
There is no thermodynamic driving force under standard conditions.
When writing explanations, be explicit that “favored/unfavored” here means thermodynamically favored/unfavored under standard conditions, matching the meaning of ΔG° and E°.
Interpreting magnitude: what a “bigger voltage” means
Because ΔG°rxn = −nFE°cell, the magnitude of ΔG° depends on both:
E°cell (intensive): does not scale with how you multiply the balanced equation.
n (extensive with stoichiometry): scales when you change the reaction stoichiometry.
Therefore:
A larger |E°cell| implies a larger free energy change per mole of reaction (greater driving force), provided n is fixed.
For reactions with different n values, ΔG° cannot be compared using E° alone; you must account for n.
Unit and setup checkpoints (high-frequency pitfalls)
Use a balanced overall redox reaction before identifying n.
Keep E°cell in volts (J C−1) and F in C mol−1 e− so ΔG° comes out in J mol−1.
If you want ΔG° in kJ mol−1, convert at the end (do not change F).
Do not multiply tabulated potentials by coefficients when balancing; instead, determine E°cell from half-reaction potentials, then use n to scale ΔG° appropriately.
FAQ
$F$ is a physical constant: the charge carried by one mole of electrons. It does not depend on the reaction, only on the electron’s charge and Avogadro’s constant.
It means $\Delta G^\circ$ corresponds to the balanced overall equation exactly as stated. If you double the equation, you double $\Delta G^\circ$ because you have doubled the amount of reaction.
Yes. Half-reactions are not independent processes in a functioning cell; only the combined overall reaction determines $E^\circ_{\text{cell}}$ and thus $\Delta G^\circ$.
The equation uses the reversible (ideal) cell potential. Real cells under load can show a lower measured voltage due to internal resistance and overpotentials, so the measured value may not reflect the thermodynamic maximum.
Modern standard state for gases is often 1 bar. The difference from 1 atm is small, but it can slightly change reported standard potentials and derived $\Delta G^\circ$ values; use a consistent data set.
Practice Questions
Question 1 (3 marks) A redox reaction has and transfers moles of electrons per mole of reaction. State whether the reaction is thermodynamically favourable under standard conditions and determine the sign of .
States implies reaction is thermodynamically favourable under standard conditions. (1)
Uses relationship to link signs. (1)
Correctly states is negative. (1)
Question 2 (6 marks) Explain how the equation links measurable voltage to thermodynamic driving force. In your answer, include the meaning of , why the negative sign matters, and why is not multiplied when the overall equation is scaled.
Identifies as moles of electrons transferred for the balanced overall reaction. (1)
Explains converts between moles of electrons and charge (C mol e). (1)
Links voltage (V = J C) with energy per charge, so gives energy per mole of reaction. (1)
Correctly interprets negative sign: (favourable) and (unfavourable). (2)
Explains is intensive (does not scale with stoichiometric multiplication), whereas scales via . (1)

