AQA Specification focus:
'- Exploring the concept of entropy (ΔS) and its impact on physical and chemical changes.
- Understanding the Gibbs free-energy change (ΔG) equation: ΔG = ΔH – TΔS.
- Skills: Calculating entropy changes, applying the Gibbs equation to assess reaction feasibility, analysing how ΔG varies with temperature.'
Entropy (ΔS)
Entropy, symbolized as ΔS, serves as a quantifiable measure of disorder or randomness within a system. This concept is instrumental in understanding the spontaneous nature of processes in thermodynamics.
Fundamentals of Entropy
Definition: Entropy is a thermodynamic property that signifies the degree of disorder or randomness in a system. It is a measure of the system's thermal energy per unit temperature that is not available for doing useful work.
Physical Context: The concept of entropy is vividly illustrated through phase transitions. For instance, the melting of ice into water signifies an increase in entropy, as the structured arrangement of water molecules in ice gives way to a less ordered liquid state.
Chemical Perspective: In chemical reactions, particularly those involving gases, entropy plays a crucial role. The change in the number of gas molecules during a reaction often leads to significant changes in the system's entropy.
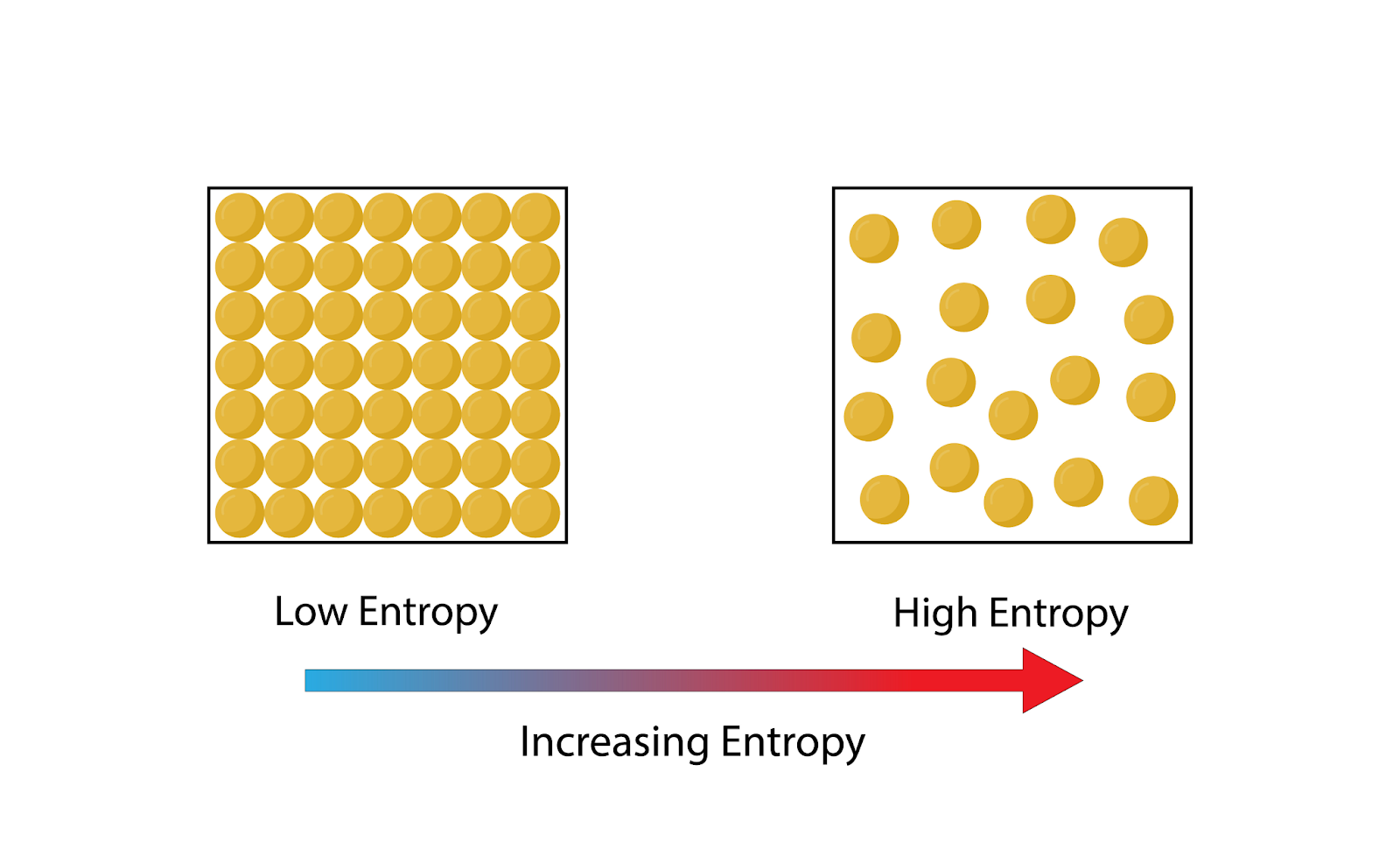
Image courtesy of PH-HY
Influencing Factors on Entropy
Temperature Influence: As temperature escalates, so does entropy, due to the enhanced kinetic energy of particles, which fosters increased movement and disorder.
Impact of Phase Changes: Transitions from solid to liquid to gas are accompanied by marked increases in entropy, attributable to the progressive loosening of molecular arrangements.
Complexity and Size of Molecules: Molecules of greater complexity and size possess a higher number of possible microstates, thereby exhibiting higher entropy.
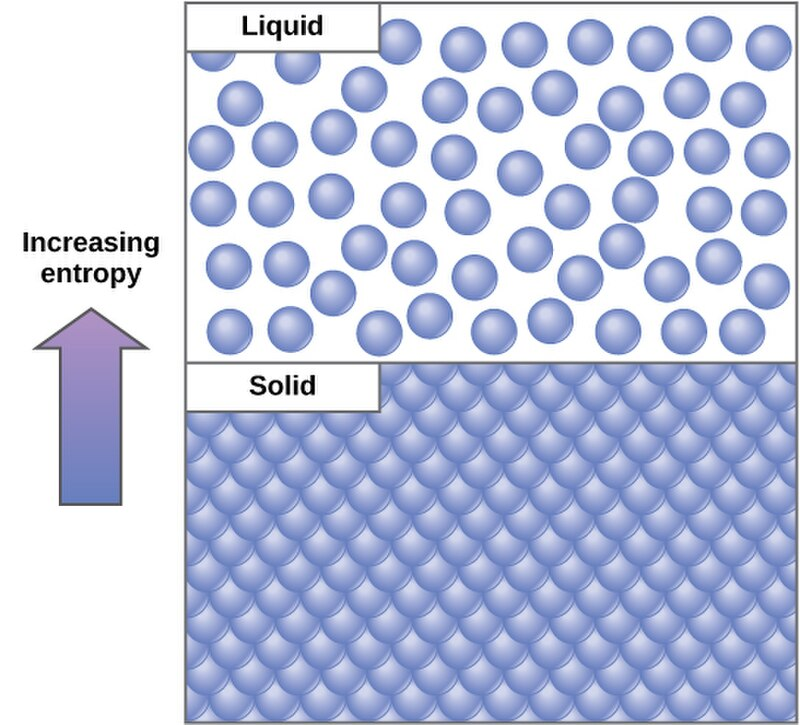
Image courtesy of CNX OpenStax
Entropy Change Calculations
Standard Entropy Values: The calculation of entropy changes in reactions often involves the use of standard entropy values (S°) for substances, which are readily available in thermodynamic data tables.
Calculation Method: The entropy change for a chemical reaction can be determined using the formula ΔS = ΣS°(products) - ΣS°(reactants), where Σ denotes summation over all reactants and products.
Gibbs Free Energy (ΔG)
The Gibbs free energy, denoted as ΔG, is a thermodynamic function that predicts the direction of chemical reactions under constant pressure and temperature conditions.
Understanding Gibbs Free Energy
Definition and Equation: Gibbs free energy is defined as the maximum reversible work that a thermodynamic system can perform at constant temperature and pressure. It is calculated using the equation ΔG = ΔH - TΔS, where ΔH is the enthalpy change, T is the absolute temperature, and ΔS is the entropy change.
Significance of ΔG: The sign of ΔG serves as an indicator of a process's spontaneity. A negative ΔG indicates a spontaneous process, ΔG equal to zero denotes equilibrium, and a positive ΔG signifies non-spontaneity.

Image courtesy of SAMYA
Application in Chemical Reactions
Feasibility of Reactions: The Gibbs free energy change provides a criterion for the spontaneity of chemical reactions, with negative ΔG values indicating thermodynamically favorable reactions.
Temperature Dependency: The term TΔS in the Gibbs equation highlights the temperature dependency of reaction spontaneity, allowing for the prediction of how reaction feasibility varies with temperature changes.
Temperature Dependence of ΔG
Exothermic Reactions: In exothermic reactions (ΔH < 0), an increase in temperature can render the reaction less spontaneous due to the larger TΔS term, which may offset the negative ΔH.
Endothermic Reactions: For endothermic reactions (ΔH > 0), raising the temperature can enhance the reaction's spontaneity, as the positive TΔS term contributes favorably to a more negative ΔG.
Skills Development
Mastering Entropy Calculations
Engagement with Practice Problems: To consolidate their understanding, students should tackle a variety of problems that involve calculating the entropy changes associated with different chemical reactions, using standard entropy values.
Real-World Contextualization: Applying the concept of entropy to real-life scenarios, such as the dissolution of salts or the combustion of hydrocarbons, can help students appreciate its relevance beyond the theoretical framework.
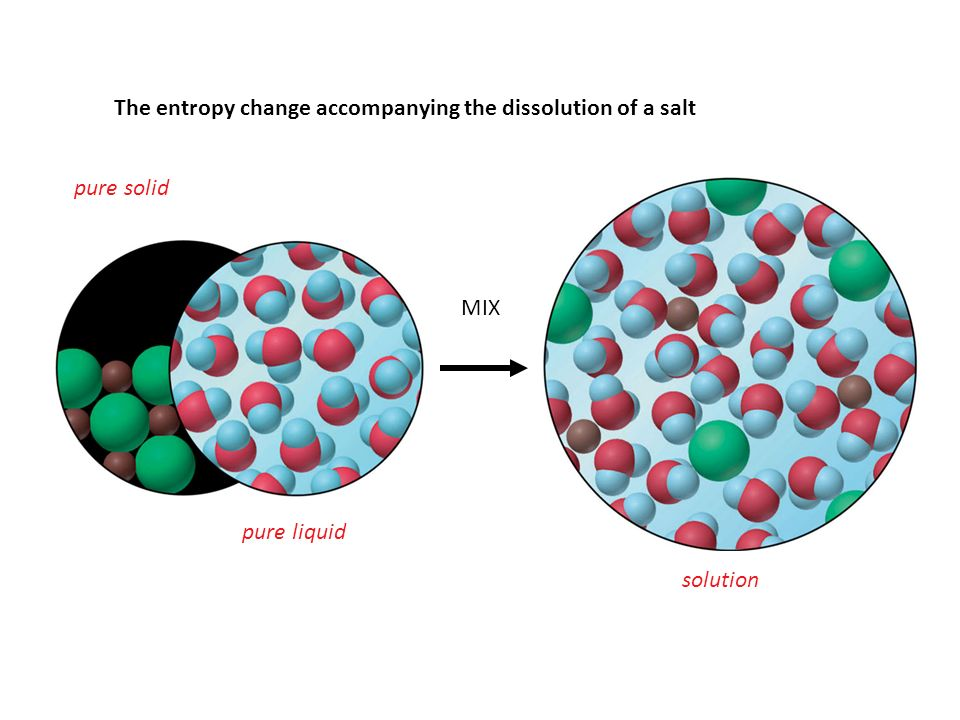
Image courtesy of SlidePlayer
Proficiency in Gibbs Free Energy
Problem-Solving Exercises: Students should be encouraged to solve problems that require the application of the Gibbs free energy equation to assess the spontaneity of chemical reactions under various conditions.
Graphical Interpretation: Learning to interpret and construct ΔG versus temperature plots can greatly aid in visualizing how the spontaneity of reactions changes with temperature, offering deeper insights into thermodynamic principles.
Experimental Techniques
Conducting Relevant Experiments: Engaging in laboratory experiments designed to measure entropy and Gibbs free energy changes, such as those involving phase changes or reactions at different temperatures, can provide valuable hands-on experience.
Data Analysis Skills: Developing the ability to interpret experimental results in the context of theoretical concepts like entropy and Gibbs free energy is crucial for bridging the gap between theory and practice.
In summary, a profound comprehension of entropy and Gibbs free energy is indispensable for A-level chemistry students. These concepts not only elucidate the fundamental principles governing chemical and physical processes but also equip students with the analytical tools necessary to predict the behavior of chemical systems. Through diligent study, practical application, and problem-solving, students can attain a robust understanding of these pivotal thermodynamic concepts, laying a solid foundation for their future academic and professional endeavors in the field of chemistry.
FAQ
The concept of microstates is fundamental to understanding entropy, as it directly relates to the disorder or randomness within a system. A microstate is a specific way in which a system's components (e.g., atoms, molecules) can be arranged, consistent with the system's macroscopic properties. Entropy is a measure of the number of microstates available to a system at a given energy level. The more microstates a system has, the higher its entropy.
In chemical reactions, changes in the number of microstates play a crucial role in determining entropy changes. For instance, when a solid dissolves in a solvent, the solute particles become more dispersed in the solution, leading to a significant increase in the number of possible microstates and, consequently, an increase in entropy. Similarly, a gas expanding into a vacuum sees a dramatic increase in the number of accessible microstates, resulting in a substantial rise in entropy. Understanding microstates allows chemists to predict how changes in system conditions (like phase changes, mixing, or chemical reactions) will affect the system's entropy. It provides a microscopic perspective on the macroscopic observation that processes tend to move towards more disordered states, aligning with the second law of thermodynamics.
A reaction with a positive ΔG, indicating non-spontaneity under initial conditions, can indeed become spontaneous if certain conditions change. This shift primarily hinges on the factors influencing ΔG, which include the enthalpy change (ΔH), the entropy change (ΔS), and the temperature (T), as outlined in the Gibbs free energy equation: ΔG = ΔH - TΔS.
Temperature is a critical factor that can alter the spontaneity of a reaction. For a reaction with a positive ΔG due to a high positive ΔH (endothermic process) and a positive ΔS (increase in disorder), increasing the temperature can make the reaction spontaneous. This is because the TΔS term becomes more significant with rising temperature, potentially outweighing the positive ΔH, leading to a negative ΔG.
Pressure and concentration changes can also affect the spontaneity of reactions, especially in gas-phase reactions or reactions involving solutes. Altering the pressure or concentration can shift the equilibrium position, thereby changing the system's enthalpy and entropy and potentially the sign of ΔG.
In summary, by manipulating temperature, pressure, concentration, or other relevant conditions, it is possible to change the sign of ΔG from positive to negative, making a previously non-spontaneous reaction spontaneous.
Exothermic reactions typically have a negative ΔG because they release energy to the surroundings, resulting in a negative enthalpy change (ΔH). According to the Gibbs free energy equation, ΔG = ΔH - TΔS, a negative ΔH contributes to making ΔG negative, implying spontaneity under standard conditions.
However, the sign of ΔG also depends on the entropy change (ΔS) and the temperature (T). Even in an exothermic reaction, if the entropy change is negative (indicating a decrease in disorder) and significant enough, it could, at certain temperatures, counteract the effect of the negative ΔH, leading to a positive ΔG. This scenario would make the reaction non-spontaneous under those specific conditions.
An example is the freezing of water. Despite being exothermic, it only occurs spontaneously at temperatures below 0°C. Above this temperature, the positive TΔS term (due to the decrease in entropy as liquid water becomes solid ice) outweighs the negative ΔH, resulting in a positive ΔG, and the process is non-spontaneous. Thus, while exothermic reactions often have a negative ΔG, entropy changes and temperature conditions can create exceptions to this general trend.
The relationship between Gibbs free energy change (ΔG) and the equilibrium constant (K) is a pivotal concept in thermodynamics, encapsulated by the equation ΔG° = -RT ln K, where ΔG° is the standard Gibbs free energy change, R is the universal gas constant, and T is the temperature in Kelvin. This equation establishes a direct link between the thermodynamic spontaneity of a reaction (as indicated by ΔG°) and its equilibrium position (as represented by K).
At equilibrium, ΔG for the reaction is zero, indicating no net change; however, ΔG° provides insight into the position of equilibrium. A negative ΔG° implies a large positive value of K, indicating that the reaction favors the formation of products at equilibrium. Conversely, a positive ΔG° corresponds to a small value of K (less than 1), suggesting that reactants are favored at equilibrium.
This relationship implies that the spontaneity of a reaction (as determined by the sign of ΔG°) directly influences the extent to which the reaction proceeds before reaching equilibrium. A highly spontaneous reaction (large negative ΔG°) will proceed almost to completion, yielding a high concentration of products, whereas a non-spontaneous reaction (positive ΔG°) will have limited product formation at equilibrium. Understanding this correlation is crucial for predicting the direction and extent of chemical reactions under standard conditions.
Coupling reactions with positive ΔG (non-spontaneous) to those with negative ΔG (spontaneous) is a common strategy in both chemistry and biochemistry to drive unfavorable reactions forward. The overall spontaneity of coupled reactions is determined by the sum of the individual ΔG values. If the sum of the Gibbs free energy changes for the coupled reactions is negative, the overall process becomes spontaneous.
This strategy is particularly significant in biological systems, where it is essential for synthesizing complex biomolecules and carrying out vital processes that would otherwise be non-spontaneous. For example, the synthesis of ATP (adenosine triphosphate) in cells involves the coupling of reactions. The hydrolysis of ATP releases a significant amount of energy (negative ΔG), which can be used to power other reactions that require an input of energy (positive ΔG). This coupling ensures that essential biological functions, such as muscle contraction, nerve impulse propagation, and biosynthesis of vital compounds, can occur despite their unfavorable thermodynamics.
The ability to couple reactions allows biological systems to maintain homeostasis and carry out a wide range of functions efficiently. It exemplifies the principle of energy conservation and the interconnectedness of metabolic pathways, highlighting the elegance and complexity of biochemical processes in living organisms.
Practice Questions
A reaction has a ΔH of -120 kJ/mol and a ΔS of -200 J/K·mol. Calculate the Gibbs free energy change (ΔG) at 298 K and determine whether the reaction is spontaneous under these conditions.
The Gibbs free energy change (ΔG) is calculated using the equation ΔG = ΔH - TΔS. Here, ΔH = -120 kJ/mol (which is -120,000 J/mol for consistency with ΔS units), ΔS = -200 J/K·mol, and T = 298 K. Substituting these values gives ΔG = -120,000 J/mol - (298 K × -200 J/K·mol) = -120,000 J/mol + 59,600 J/mol = -60,400 J/mol. Since ΔG is negative, the reaction is spontaneous at 298 K. This result indicates that the exothermic nature of the reaction (negative ΔH) outweighs the decrease in entropy (negative ΔS), leading to spontaneity.
Explain how the sign of ΔG changes for an endothermic reaction (positive ΔH) with an increase in entropy (positive ΔS) as the temperature is increased.
For an endothermic reaction with a positive ΔH, the increase in entropy (positive ΔS) plays a key role in determining the spontaneity as the temperature increases. As the temperature rises, the TΔS term in the Gibbs free energy equation (ΔG = ΔH - TΔS) becomes more significant. Since ΔS is positive, the TΔS term will increase with temperature, thus reducing the overall value of ΔG. At lower temperatures, the positive ΔH may dominate, leading to a positive ΔG (non-spontaneous reaction). However, at higher temperatures, the increasing TΔS term can offset the positive ΔH, potentially making ΔG negative and the reaction spontaneous. This demonstrates how temperature can critically influence the spontaneity of endothermic reactions with positive entropy changes.

