AQA Specification focus:
'- Definition and derivation of the equilibrium constant Kp for reversible gas-phase reactions.
- The relationship between Kp and the partial pressures of reactants and products.
- Skills: Deriving Kp expressions for homogeneous systems, understanding the concept and calculation of partial pressure.'
Definition of Equilibrium Constant ( Kp )
The equilibrium constant ( Kp ) plays a fundamental role in the analysis of gas-phase chemical reactions at equilibrium. It is defined as the ratio of the product of the partial pressures of the products to the product of the partial pressures of the reactants, with each term raised to the power of its stoichiometric coefficient in the balanced chemical equation.
Equilibrium State: A reaction reaches equilibrium when the rates of the forward and reverse reactions equalize, resulting in no net change in the concentrations of reactants and products over time.
Partial Pressure: The pressure exerted by a single gas in a mixture, as if it were the only gas present in the entire volume of the mixture.
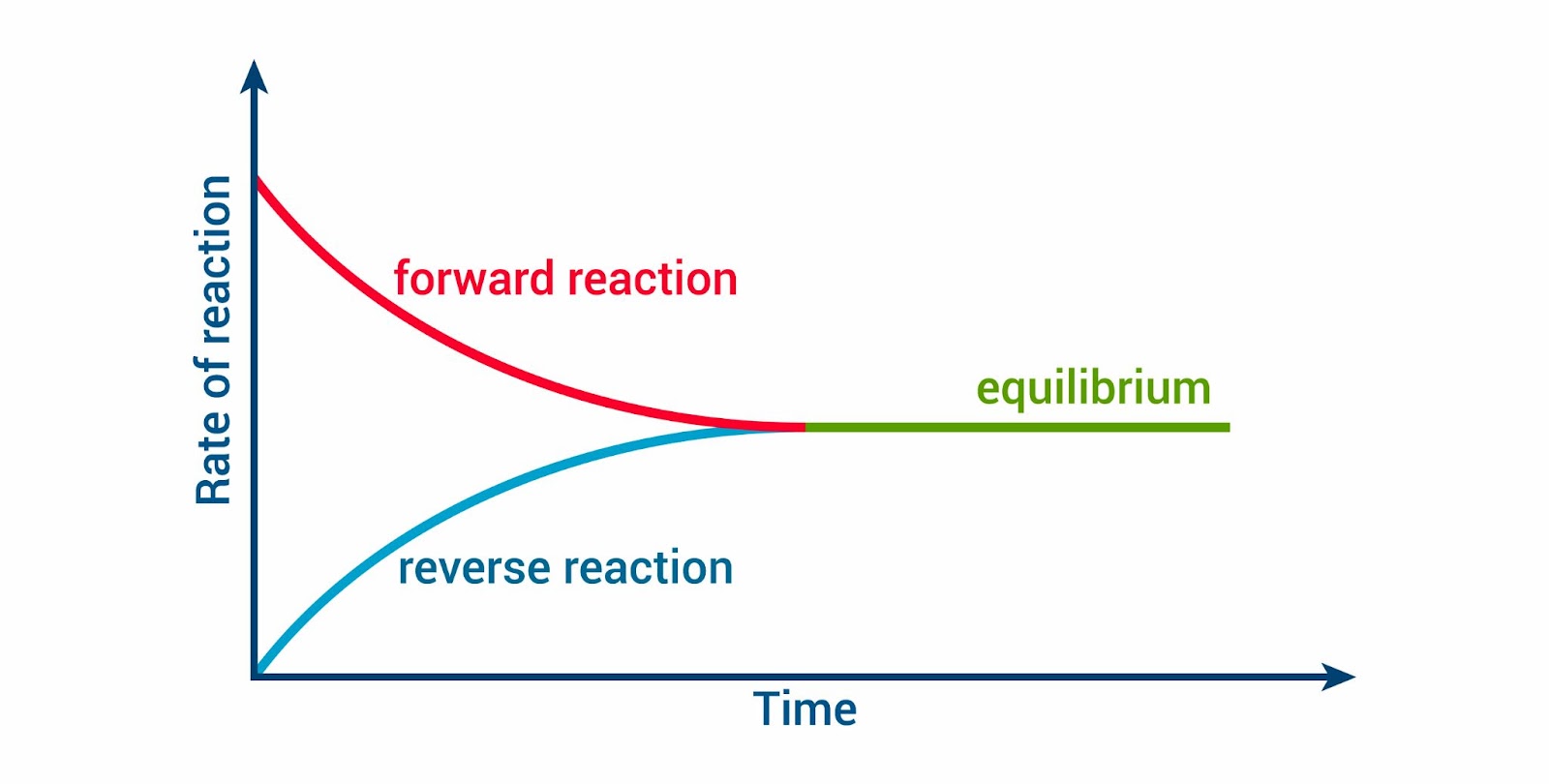
Image courtesy of Zizo
Derivation of ( Kp )
Deriving ( Kp ) involves translating the balanced chemical equation into an expression that relates the partial pressures of the reactants and products at equilibrium. Consider a general gas-phase reaction:
[ aA(g) + bB(g) \rightleftharpoons cC(g) + dD(g) ]
where ( a ), ( b ), ( c ), and ( d ) are the stoichiometric coefficients for reactants ( A ) and ( B ), and products ( C ) and ( D ) respectively. The equilibrium constant ( Kp ) for this reaction is expressed as:
[ Kp = \frac{{PCc \times PDd}}{{PAa \times PBb}} ]
where ( PX ) denotes the partial pressure of substance ( X ) at equilibrium.
Steps in Derivation
Equilibrium Expression from the Balanced Equation: Begin by writing the equilibrium expression in terms of concentration for the reaction.
Conversion to Partial Pressures: Utilize the ideal gas law (( P = \frac{nRT}{V} )), where ( P ) is pressure, ( n ) is moles, ( R ) is the universal gas constant, ( T ) is temperature, and ( V ) is volume, to convert concentrations to partial pressures.
Formulation of ( Kp ): Rearrange the terms to express the equilibrium constant in terms of the partial pressures of the reactants and products.
Relationship Between ( Kp ) and Partial Pressures
The equilibrium constant ( Kp ) is intrinsically linked to the partial pressures of the reactants and products in a gas-phase reaction. This relationship is crucial for understanding how changes in conditions affect the position of equilibrium.
Proportionality with Product Pressures: An increase in the partial pressures of the products, with reactant pressures held constant, leads to an increase in ( Kp ).
Inverse Relation with Reactant Pressures: Conversely, increasing the partial pressures of the reactants, while keeping product pressures constant, results in a decrease in ( Kp ).
Deriving ( Kp ) Expressions for Homogeneous Systems
In homogeneous systems, where all reactants and products are in the same phase (gas phase in this context), deriving ( Kp ) expressions involves several key steps:
Balanced Chemical Equation: Start with a correctly balanced equation to reflect accurate stoichiometry.
Identification of Partial Pressures: Assign symbols to represent the partial pressures of all gases involved.
Equilibrium Constant Expression: Use the general form ( Kp = \frac{{PCc \times PDd}}{{PAa \times PBb}} ), substituting the respective partial pressures and stoichiometric coefficients.
Example
Consider the synthesis of ammonia:
[ N2(g) + 3H2(g) \rightleftharpoons 2NH3(g) ]
The ( Kp ) expression for this reaction is:
[ Kp = \frac{{P{NH3}2}}{{P{N2} \times P{H2}3}} ]
This example illustrates how ( Kp ) is influenced by the stoichiometry of the reaction, highlighting the direct relationship between ( Kp ) and the partial pressures of products and the inverse relationship with those of the reactants.
Understanding Partial Pressure
Partial pressure is a concept that underpins the discussion of gas-phase equilibria. It is defined as the pressure a gas component of a mixture would exert if it occupied the entire volume of the container on its own.
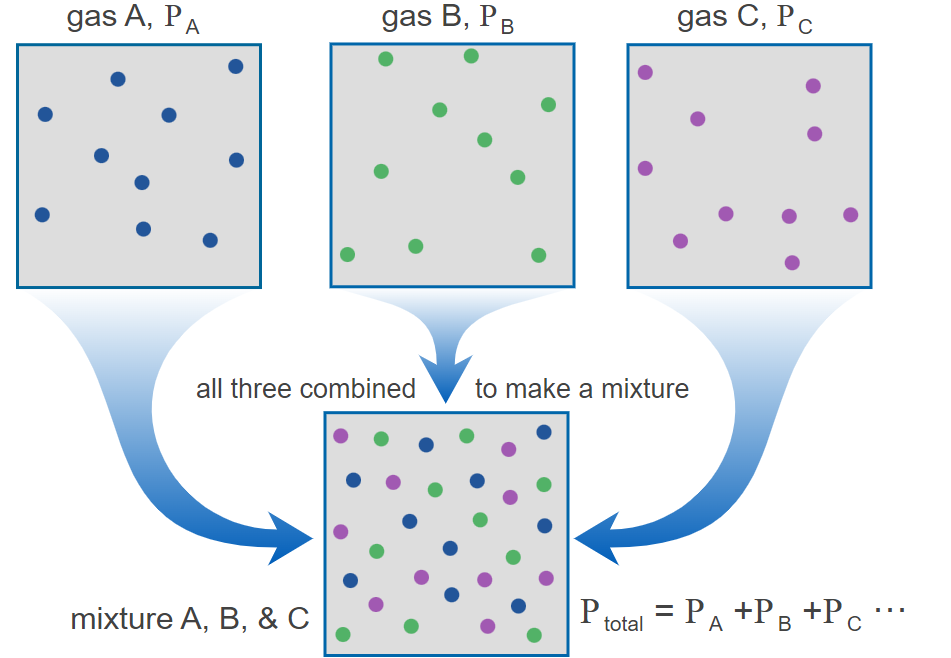
Image courtesy of Chemistry 301
Calculation of Partial Pressure
The partial pressure of a gas in a mixture can be determined by multiplying its mole fraction by the total pressure of the mixture. The mole fraction is calculated as the number of moles of the gas divided by the total number of moles in the mixture.
Influencing Factors
Several factors can affect the partial pressures of gases in a mixture:
Total Pressure: Variations in the total pressure of the system can alter the partial pressures of the individual gases.
Temperature: Changes in temperature affect the kinetic energy of gas molecules, which in turn influences their partial pressures.
Volume Changes: Changes in the volume of the gas container can lead to variations in partial pressures, as described by Boyle’s Law, which states that pressure and volume are inversely proportional at constant temperature and number of moles.
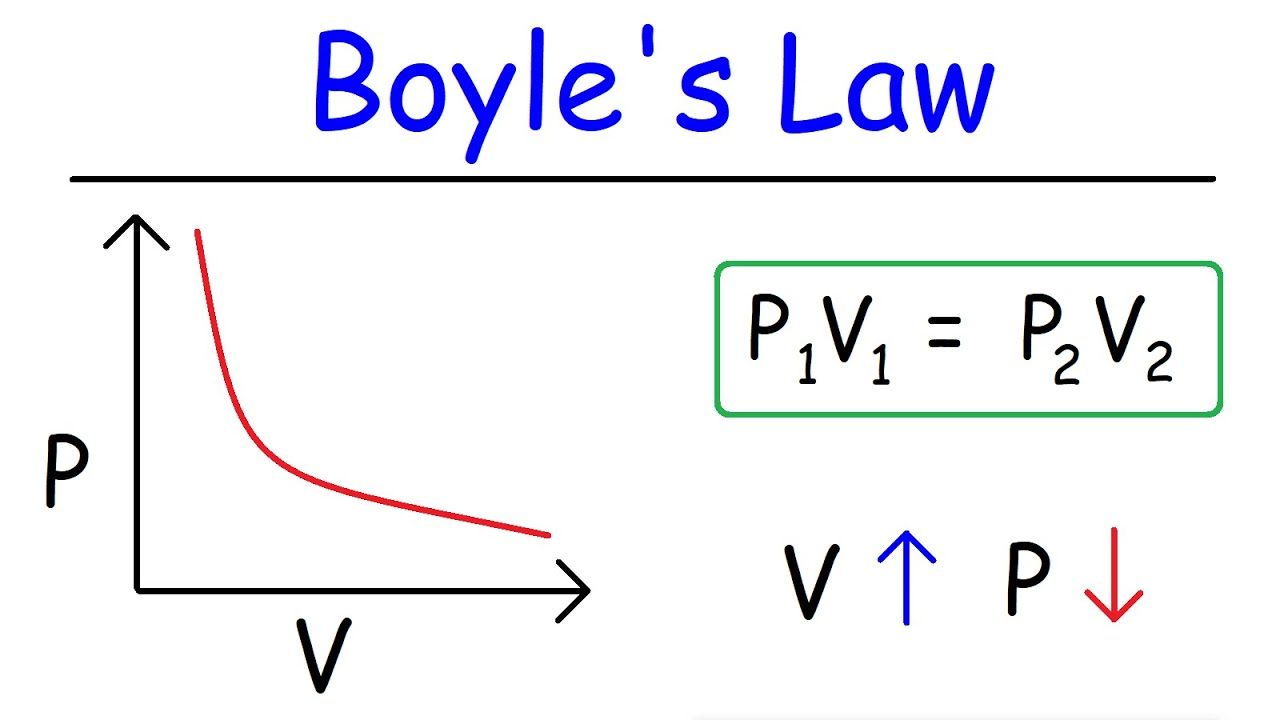
Image courtesy of The Organic Chemistry Tutor
Skills Development
Deriving ( Kp ) Expressions
To master equilibrium concepts, practice deriving ( Kp ) expressions for various gas-phase reactions. Pay special attention to the stoichiometry of the reactions, as it directly influences the form of the ( Kp ) expression.
Calculating Partial Pressures
Develop proficiency in calculating the partial pressures of gases in a mixture using mole fractions and total pressure. This skill is essential for solving equilibrium problems involving gas-phase reactions.
Conceptual Understanding
Deepen your understanding of how changes in conditions, such as temperature, pressure, and volume, affect the partial pressures of gases and, consequently, the value of ( Kp ). This understanding is crucial for predicting the direction in which equilibrium will shift in response to external changes.
By mastering the derivation of ( Kp ), its relationship with partial pressures, and the factors affecting these pressures, you will gain a robust foundation in chemical equilibrium. This knowledge is not only fundamental to academic success in chemistry but also critical for understanding the principles behind various industrial processes and natural phenomena.
FAQ
The value of the equilibrium constant ( Kp ) provides significant insight into the position of equilibrium and, thus, the direction of a gas-phase reaction. A high ( Kp ) value, significantly greater than 1, indicates that the equilibrium position is heavily skewed towards the products, suggesting that the reaction proceeds predominantly in the forward direction under given conditions. Conversely, a ( Kp ) value much less than 1 implies that the reactants are favoured, and the reaction tends to proceed in the reverse direction. When ( Kp ) is approximately 1, it suggests that neither reactants nor products are significantly favoured, and the reaction mixture contains appreciable amounts of both. This understanding is crucial for chemists, allowing them to predict reaction outcomes and devise conditions that favour the desired direction of a reaction. For instance, in industrial synthesis processes, knowing how to manipulate conditions to achieve a high yield of products can significantly impact efficiency and cost-effectiveness.
A catalyst accelerates both the forward and reverse reactions in a chemical system without being consumed, thus reaching equilibrium more quickly. However, it does not alter the thermodynamics of the reaction, including the relative energies of the reactants and products or the position of equilibrium. Since ( Kp ) is a reflection of the ratio of the concentrations (or partial pressures) of the products to the reactants at equilibrium, and a catalyst does not change these ratios, ( Kp ) remains unaffected. This principle is fundamental in understanding that while catalysts are invaluable for increasing the rate of chemical reactions and making processes more efficient, they cannot be used to alter the equilibrium constant or shift the position of equilibrium in favour of either the reactants or the products. The effectiveness of a catalyst in a particular reaction is a critical consideration in industrial chemistry, where reaction rates are as important as equilibrium positions.
The value of ( Kp ) is directly influenced by the stoichiometry of the reaction equation. If the balanced equation for a reaction is altered, for instance, by multiplying all the coefficients by a factor, the expression for ( Kp ) changes accordingly. For a reaction where the coefficients are multiplied by a factor ( n ), the partial pressures in the ( Kp ) expression are raised to the power of ( n ), significantly affecting the value of ( Kp ). This change is due to the exponential nature of the ( Kp ) expression, where the partial pressures are raised to the power of their respective stoichiometric coefficients. Therefore, a seemingly minor change in stoichiometry can lead to a substantial change in the equilibrium constant. This concept underscores the importance of accurate stoichiometric balance in predicting and understanding equilibrium positions and is a fundamental aspect of reaction stoichiometry in chemical equilibrium studies.
( Kp ) offers valuable information about the extent to which a reaction proceeds before reaching equilibrium. A high ( Kp ) value indicates a large extent of reaction, meaning that, at equilibrium, the concentration (or partial pressure) of the products is much greater than that of the reactants. This suggests that the reaction proceeds almost to completion in the forward direction under the given conditions. On the other hand, a low ( Kp ) value implies that the reactants are predominant at equilibrium, and the reaction does not proceed far in the forward direction. However, ( Kp ) alone does not provide the complete picture of the reaction's dynamics, such as how quickly equilibrium is reached or the exact concentrations of reactants and products at equilibrium. Still, it is a crucial parameter for understanding the general behaviour of chemical systems and designing processes and reactions to achieve desired outcomes.
Adding an inert gas to a gas-phase equilibrium system at constant volume does not affect the value of ( Kp ). This is because ( Kp ) is dependent only on the partial pressures of the reactive gases and is independent of the total pressure of the system. Inert gases do not react with the components of the mixture; thus, their addition does not change the partial pressures of the reactive components, assuming the volume remains constant. However, if the addition of an inert gas results in a change in volume, this could indirectly affect the partial pressures of the reactants and products, potentially shifting the position of equilibrium. But the intrinsic value of ( Kp ), which reflects the ratio of product to reactant partial pressures at equilibrium, remains unchanged. This principle is important in understanding how external factors influence chemical equilibria and is particularly relevant in industrial processes where reactions are carried out under varying conditions.
Practice Questions
Consider the reaction: ( N2(g) + O2(g) \rightleftharpoons 2NO(g) ) at equilibrium. The partial pressures are found to be: ( P{N2} = 2.0 \, \text{atm} ), ( P{O2} = 1.0 \, \text{atm} ), and ( P{NO} = 4.0 \, \text{atm} ). Calculate the equilibrium constant ( Kp ) for the reaction.
To calculate the equilibrium constant ( Kp ), we use the expression ( Kp = \frac{{P{NO}2}}{{P{N2} \times P{O2}}} ). Substituting the given partial pressures, we have ( Kp = \frac{{4.02}}{{2.0 \times 1.0}} = \frac{16}{2} = 8.0 ). Therefore, the equilibrium constant ( Kp ) for the reaction is 8.0 atm. This calculation demonstrates a clear understanding of how to relate the partial pressures of reactants and products to the equilibrium constant in a gas-phase reaction, a key concept in chemical equilibrium.
Explain how the value of ( Kp ) would change if the temperature of the system described in Question 1 is increased, assuming the reaction is exothermic.
For an exothermic reaction, increasing the temperature shifts the equilibrium towards the reactants, according to Le Chatelier's principle. This is because the system absorbs the added heat by favouring the endothermic reverse reaction. Consequently, the partial pressures of the products, NO, will decrease, and those of the reactants, ( N2 ) and ( O2 ), will increase. This results in a decrease in the value of ( Kp ), as ( Kp ) is directly proportional to the partial pressures of the products and inversely proportional to those of the reactants. The student's response demonstrates an understanding of the dynamic nature of chemical equilibria and the effect of temperature changes on the position of equilibrium and the value of ( Kp ).

