IB Syllabus focus:
'- Understanding the relationship between pressure, volume, temperature, and amount of an ideal gas as shown in the ideal gas equation (PV = nRT) and the combined gas law (P1V1/T1 = P2V2/T2).
- Solving problems related to the ideal gas equation.
- Using SI units for volume and pressure.
- Noting the value of the gas constant (R), the ideal gas equation, and the combined gas law from the data booklet.
- Discussing how the ideal gas law can be used to calculate the molar mass of a gas from experimental data.'
The behaviour and properties of ideal gases are governed by various laws, which in turn dictate mathematical relationships between pressure, volume, temperature, and amount of gas. In this segment, we'll delve into the key equations and their practical applications.
The Ideal Gas Equation (PV = nRT)
At the heart of understanding gaseous substances lies the Ideal Gas Equation. Here's how each variable connects:
P: Pressure of the gas
V: Volume occupied by the gas
n: Amount of gas (in moles)
R: Universal gas constant
T: Absolute temperature of the gas (in Kelvin)
Key Points:
The equation encapsulates the behaviour of an 'ideal' gas.
It represents the combined effects of Boyle's, Charles', and Avogadro's laws.

Image courtesy of Ali
The Combined Gas Law (P1V1/T1 = P2V2/T2)
Combined Gas Law (P₁V₁/T₁ = P₂V₂/T₂)
P₁, P₂ = Initial and final pressures (Pa or atm)
V₁, V₂ = Initial and final volumes (m³ or L)
T₁, T₂ = Initial and final absolute temperatures (K)
When dealing with changing conditions, the Combined Gas Law offers a relationship:
P1 & P2: Initial and final pressures
V1 & V2: Initial and final volumes
T1 & T2: Initial and final temperatures
This equation proves useful when any two parameters (like pressure and volume) change while the quantity of gas remains constant.
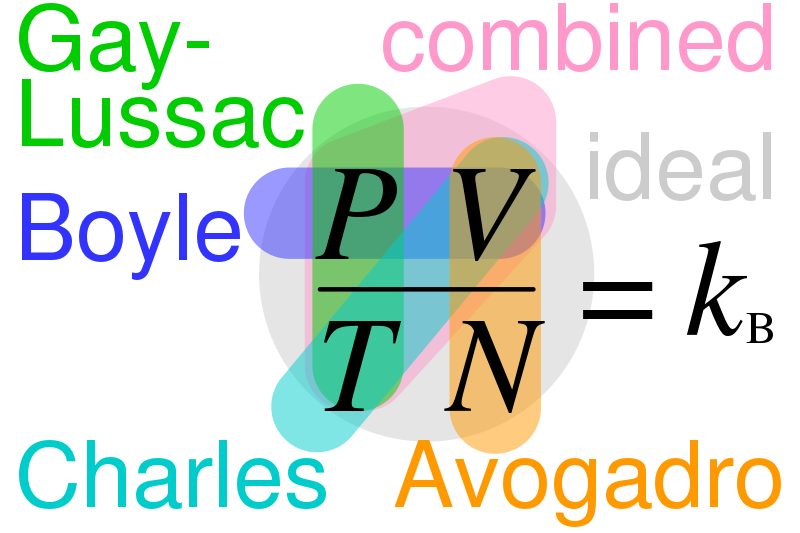
The basic formula for combined gas law showing a relation between Boyle's law, Charles's law, Gay-Lussac's law, Avogadro's law, combined and ideal gas laws. The constant k is a true constant, as long as the number of moles of a gas remains the same. If the amount of gas varies, then k changes.
Image courtesy of Cmglee
IB Chemistry Tutor Tip: Mastering gas laws equations enables predicting and explaining gas behaviour under various conditions, essential for practical experiments and understanding real-world applications in Chemistry.
Problem-Solving with the Ideal Gas Equation
Direct Proportions: When temperature increases, so does the volume (assuming pressure remains constant). Similarly, more moles of gas will lead to a higher volume if temperature and pressure stay unchanged.
Inverse Proportions: If the volume decreases, pressure increases, provided temperature and gas quantity are constant.
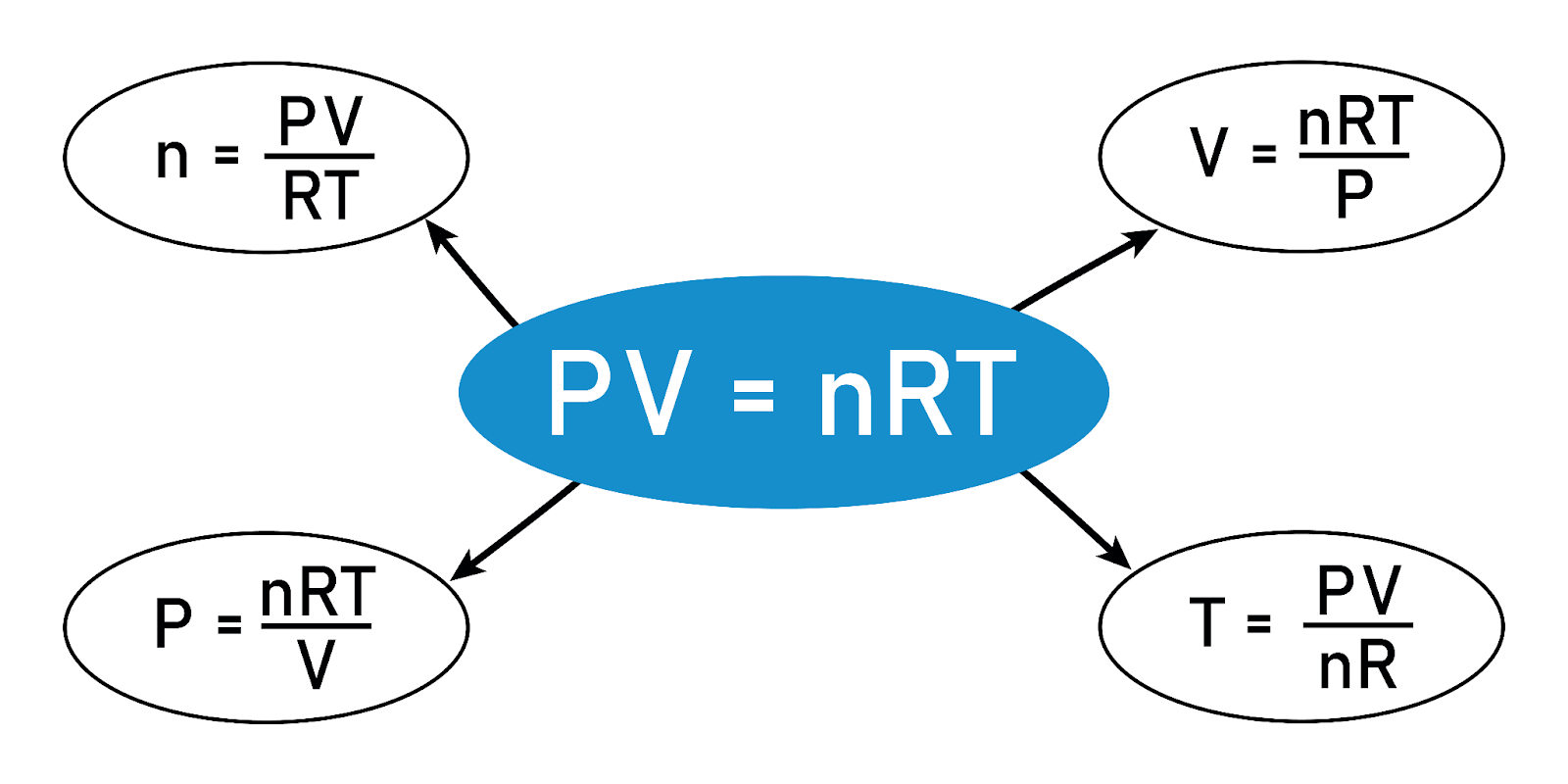
Image courtesy of SAMYA
SI Units and Conversions
It's imperative to use consistent units for accurate calculations.
Pressure: Pascal (Pa). However, many problems use atmospheres (atm) or torr. Conversions are crucial.
Volume: Cubic metres (m^3), though litres (L) are more common in chemistry.
Temperature: Kelvin (K). Always convert Celsius to Kelvin by adding 273.15.
Gas Constant (R) and Its Values
The value of the gas constant, R, depends on the units employed:
If pressure is in Pa and volume in m3, then R = 8.31 J/(mol·K).
For pressure in atm and volume in L, R = 0.0821 L·atm/(mol·K).
Always refer to the data booklet for exact values to ensure precision in calculations.
Gas Constant (R): A physical constant that relates the energy scale to temperature and amount of substance. Common values include 8.31 J mol⁻¹ K⁻¹ when using SI units, and 0.0821 L atm mol⁻¹ K⁻¹ when using litres and atmospheres.
IB Tutor Advice: Always check your units before calculation; converting to SI units when necessary ensures accuracy in using the Ideal and Combined Gas Laws in exams.
Determining Molar Mass from Experimental Data
The ideal gas equation isn't just theoretical; it's a practical tool. Given the mass of a gas and its volume, pressure, and temperature, one can ascertain its molar mass.
Steps:
Utilise the ideal gas equation to determine the number of moles (n).
Calculate molar mass: Molar mass = given mass / n
Molar Mass (M) = m / n
M = Molar mass of the gas (g mol⁻¹)
m = Mass of the gas (g)
n = Amount of substance (mol)
Example: Suppose you have 10g of an unknown gas at 1 atm and 273 K, occupying 5.6 L. Using the Ideal Gas Equation, you can deduce the number of moles and consequently the gas's molar mass.
In conclusion, these laws and equations are foundational in gas chemistry, bridging theoretical concepts with tangible, measurable quantities. Through adept manipulation of these equations, one can gain profound insights into the nature and behaviour of gaseous substances.
FAQ
Employing SI units in gas calculations ensures uniformity and precision. The gas constant, R, has set values in various unit systems, so it's vital to use units that match the appropriate R value. Using m^3 for volume and Pa for pressure matches with the SI value of R, ensuring calculations are accurate and comparable across different contexts.
The combined gas law relates pressure, volume, and temperature of a gas undergoing a change in conditions, represented as P1V1/T1 = P2V2/T2. On the other hand, the ideal gas equation, PV = nRT, links pressure, volume, temperature, and the number of gas moles. While the ideal gas equation gives insights into a specific state of an ideal gas, the combined gas law is more comparative, detailing the relationship between two different gas states.
The ideal gas equation is based on certain assumptions – that gas particles have negligible volume and no forces act between them. In reality, gas particles do have volume, and they exert forces on one another, especially at high pressures or low temperatures. Such conditions make real gases diverge from the ideal gas equation's predictions. For instance, gases might liquify at low temperatures or not behave as predicted under high pressures. This highlights the importance of understanding the model's limitations.
Absolutely. For a gas with two distinct sets of conditions (initial and final), the ideal gas equation can be expressed for each state: P1V1 = nRT1 and P2V2 = nRT2. Given that both the amount of gas (n) and the gas constant (R) stay constant, dividing these two equations gives us P1V1/T1 = P2V2/T2. This is the combined gas law, effectively showcasing the relationship between two sets of conditions for a given gas.
The gas constant, R, is a pivotal constant in the ideal gas equation. It's a proportionality factor that bridges the relationship between the pressure, volume, and temperature of a gas to the number of moles of the gas. Its consistent value ensures the ideal gas equation's predictions are valid across a wide range of conditions for ideal gases. Essentially, it links the macroscopic properties of gases with the quantity of particles present, ensuring consistency in calculations.
Practice Questions
To solve this problem, we can utilise the Combined Gas Law: P1V1/T1=P2V2/T2. Substituting in the given values: (1.5 atm x 2.0 L) / 300 K = P2 x 1.5 L / 320 K. By simplifying, we get: P2 = (3.0 atm x 320 K) / (1.5 L x 300 K) = 2.133 atm. Thus, the new pressure of the helium gas after compression and heating will be approximately 2.133 atm.
At STP, 1 mole of an ideal gas occupies 22.4 L. Since the volume of our unknown gas is also 22.4 L at STP, it implies the gas is equivalent to 1 mole. To determine the molar mass, we simply take the given mass and divide by the number of moles. Therefore, Molar mass = 28 g/mol / 1 mol = 28 g/mol. Thus, the molar mass of the unknown gas is 28 g/mol.

