Reactivity 1.2 – Energy cycles in reactions
Core idea: Use the law of conservation of energy to calculate enthalpy changes that may be difficult or impossible to measure directly.
Big exam theme: Move confidently between bond enthalpies, Hess’s law cycles, standard enthalpy changes of formation/combustion, and Born–Haber cycles.
Sign convention: Negative = exothermic; positive = endothermic.
Bond breaking and bond forming
Bond-breaking absorbs energy because energy is needed to overcome electrostatic attractions in a bond.
Bond-forming releases energy because the new bonded arrangement is more stable than separate atoms.
A reaction is exothermic if more energy is released making bonds than is absorbed breaking bonds.
A reaction is endothermic if more energy is absorbed breaking bonds than is released making bonds.
Use the data-booklet equation: .
Average bond enthalpies are measured for gaseous molecules and are averages over many compounds, so calculated values are approximations.
Therefore, bond enthalpy calculations often differ from experimental enthalpy changes for a specific reaction.
Stronger bonds usually have higher bond enthalpy and require more energy to break.
How to calculate from bond enthalpies
Step 1: Write a fully balanced equation.
Step 2: Identify all bonds broken in the reactants.
Step 3: Identify all bonds formed in the products.
Step 4: Substitute bond enthalpy values from the data booklet.
Step 5: Apply .
Always check stoichiometric coefficients carefully; multiply bond enthalpies by the number of each bond involved.
Bond enthalpy values are usually in kJ mol.
Common exam mistake: counting only one bond when the equation shows multiple molecules.
Hess’s law
Hess’s law: The enthalpy change for a reaction is independent of the pathway taken between the same initial and final states.
This works because enthalpy is a state function.
You can add, subtract, or reverse equations, as long as you also correctly adjust the values.
If an equation is reversed, the sign of changes.
If an equation is multiplied by a factor, the value is multiplied by the same factor.
Hess cycles are especially useful when the target reaction cannot be measured directly.
In exams, first identify the target equation, then manipulate the given equations until they add up exactly.
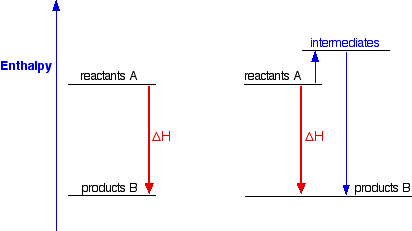
This diagram shows two different routes from the same reactants to the same products, illustrating that the overall enthalpy change is identical whichever path is taken. It is ideal for understanding why Hess’s law allows multi-step energy calculations. Source
How to use Hess’s law in calculations
Write the target equation clearly first.
Compare the target with the given equations/data.
Reverse equations if needed and change the sign of .
Multiply/divide equations if needed and scale accordingly.
Add the adjusted equations; cancel common species until the target equation remains.
Add the adjusted enthalpy values to obtain the final .
If using a cycle diagram, the total enthalpy change around the cycle must sum to zero.
Common exam mistake: adjusting the equation but forgetting to adjust the enthalpy value.
HL only – Standard enthalpy changes of formation and combustion
Standard enthalpy change of formation, = enthalpy change when 1 mol of a compound is formed from its elements in their standard states under standard conditions.
Standard enthalpy change of combustion, = enthalpy change when 1 mol of a substance undergoes complete combustion in oxygen under standard conditions.
Elements in their standard states have .
For formation data use: .
For combustion data use: .
These equations are applications of Hess’s law.
Watch the direction carefully: formation uses products minus reactants, while combustion uses reactants minus products.
Include correct state symbols because standard enthalpy data depend on the defined physical state.
HL only – Born–Haber cycles
A Born–Haber cycle is a Hess cycle used to analyse the formation of an ionic compound.
It connects enthalpy of formation with other measurable steps such as:
atomization
ionization energy/energies
electron affinity/affinities
lattice enthalpy
You may be asked to interpret a cycle or determine one missing value.
The syllabus requires compounds containing univalent and divalent ions.
Typical steps include converting elements to gaseous atoms, then to gaseous ions, then forming the ionic lattice.
Lattice enthalpy is linked to the strength of ionic attraction in the solid.
For ions with larger charges and smaller radii, lattice enthalpy is usually more exothermic in formation terms.
You are not required to construct a full Born–Haber cycle from scratch, but you must be able to read and use one.
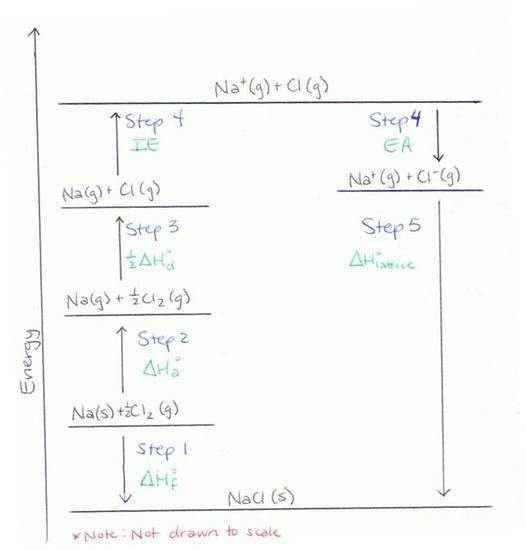
This diagram lays out the energy steps used to move from elements in standard states to gaseous atoms, then gaseous ions, and finally the ionic solid. It helps you identify where ionization energy, electron affinity, atomization, lattice enthalpy, and fit into one cycle. Source
HL only – Born–Haber cycle exam method
Start by identifying the unknown quantity the question wants.
Read the arrows carefully: each arrow represents a specific enthalpy change.
Follow one complete route between the same start and end points.
Apply Hess’s law: sum of enthalpy changes on one route = sum on the alternative route.
Keep a close eye on signs for electron affinity and lattice enthalpy.
If the question uses the formation of lattice from gaseous ions, this is usually exothermic.
If the question defines lattice dissociation enthalpy, the sign will be the opposite.
Common exam mistake: mixing up lattice formation enthalpy with lattice dissociation enthalpy.
High-yield exam traps
Bond enthalpy values are averages, so answers are approximate, not exact.
Do not confuse with .
Standard state matters: for example, the standard state of bromine is liquid, not gas.
Reversing an equation means reversing the sign of .
Multiplying an equation means multiplying by the same factor.
In Hess cycles, the chemistry must cancel to the exact target equation.
In Born–Haber cycles, distinguish lattice enthalpy of formation from lattice dissociation enthalpy.
Always check units and include kJ mol where appropriate.
Checklist: can you do this?
Calculate from average bond enthalpy data and explain why it is only an approximation.
Apply Hess’s law by reversing/scaling equations and adjusting correctly.
Use and data to determine the enthalpy change of a reaction.
Interpret a Born–Haber cycle and calculate one missing enthalpy term.
Spot sign errors and data-booklet formula errors in exam-style calculations.