Combustion basics
Combustion = reaction of a fuel with oxygen releasing energy.
Fuels in this topic: reactive metals, non-metals, hydrocarbons, alcohols.
Most combustion reactions are exothermic because strong bonds form in products such as and .
You must be able to identify combustion as oxidation and write a balanced equation.
Typical examples:
Magnesium:
Carbon:
Methane:
Ethanol:
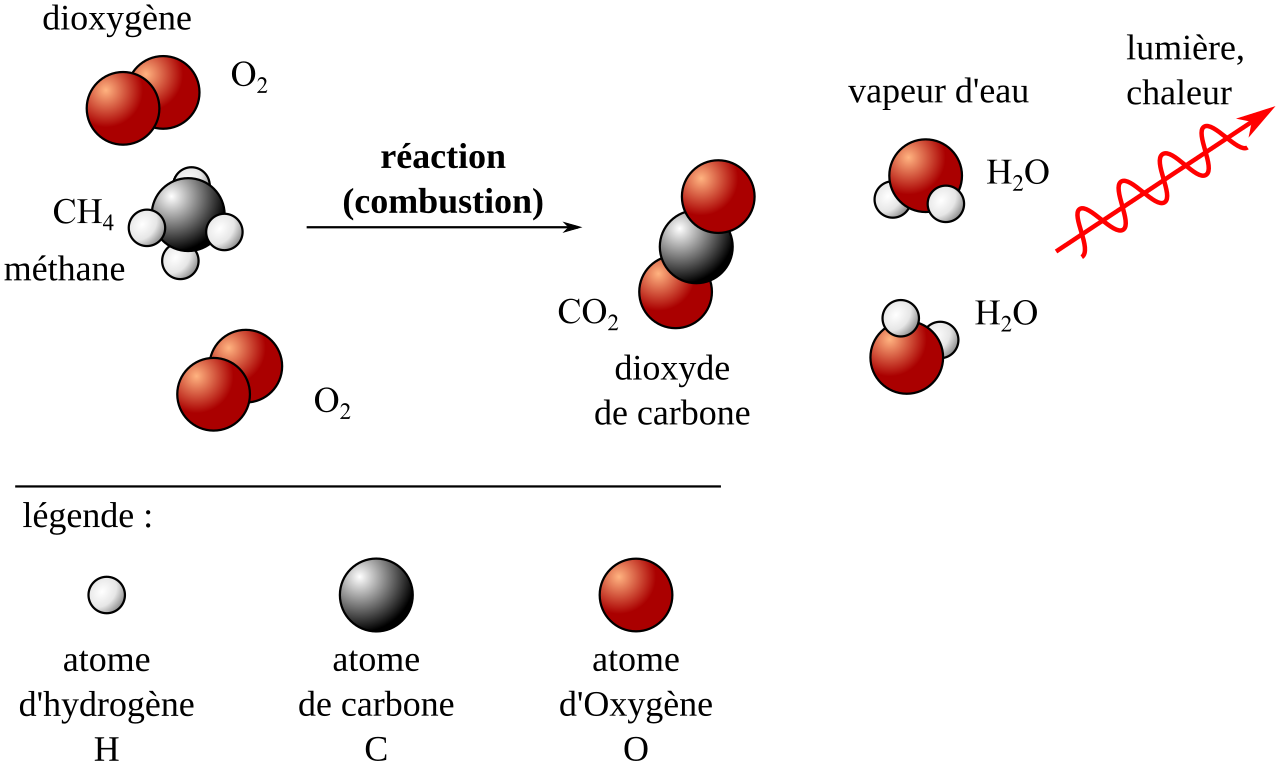
This diagram shows the balanced combustion of methane in oxygen, with atoms rearranging to form carbon dioxide and water. It is useful for checking atom conservation and product identification in complete combustion. Source
Writing combustion equations fast
For complete combustion of a hydrocarbon: products are always and .
General form:
For complete combustion of an alcohol, products are still and , but the fuel already contains oxygen, so less is needed than for a similar hydrocarbon.
Fast balancing strategy:
Balance C first.
Then balance H.
Balance O last.
Multiply through if you need whole-number coefficients.
Watch for exam traps:
Do not put or in complete combustion.
Include state symbols only if the question asks or data are given.
Incomplete combustion
Incomplete combustion happens when there is a limited supply of oxygen.
Organic fuels may produce:
Carbon monoxide,
Carbon (soot),
Water,
Examples:
Key consequences:
Less complete energy release
Formation of toxic carbon monoxide
Formation of particulates / soot
Carbon monoxide is dangerous because it is poisonous.
Larger hydrocarbons are generally more likely to burn incompletely because they are harder to mix fully with oxygen.
Fossil fuels: strengths and weaknesses
Fossil fuels = coal, crude oil, natural gas.
They are non-renewable because they form over millions of years.
Advantages:
High energy output
Established infrastructure
Often cheap and easy to transport/store
Disadvantages:
Release , increasing the greenhouse effect
Can undergo incomplete combustion, producing and soot
Cause environmental, social, economic and ethical concerns
Relative ideas you should know:
Natural gas / methane generally gives less per unit energy than coal or larger hydrocarbons because it has a higher H:C ratio.
Fuels with more carbon per unit mass tend to add more to the atmosphere when burned.
Energy released per unit mass is an important comparison point when evaluating fuels.
Evaluating emissions from fuels
In exams, compare fuels by the amount of carbon dioxide formed when they burn.
Core idea: Every carbon atom in the fuel becomes one carbon atom in in complete combustion.
Therefore:
1 mol C in a fuel produces 1 mol
More moles of carbon burned more moles of
Useful exam method:
Write the balanced combustion equation.
Use the equation to find moles of .
Convert to mass of if needed.
For evaluation questions, compare released per unit mass or per unit energy.
Key judgement points:
A fuel may be energetically useful but still be environmentally worse if it releases more .
Methane is often favored over coal because it is cleaner-burning and produces less per unit energy.
Greenhouse effect link
is a greenhouse gas.
Higher atmospheric leads to an enhanced greenhouse effect.
This means more infrared radiation is retained by the atmosphere, increasing global temperatures.
IB focus: know the link, not just the definition:
Burning fossil fuels more
more stronger greenhouse effect
Do not confuse:
Greenhouse effect = heat trapping by gases
Ozone depletion = a different environmental issue
Biofuels
Biofuels are fuels made from recently living material.
They are considered renewable because the carbon in them was fixed from the atmosphere over a short timescale.
This carbon is fixed by photosynthesis:
Why biofuels may reduce net atmospheric :
Plants remove first by photosynthesis.
Burning the biofuel later returns this recently fixed carbon.
Advantages:
Renewable source
Potentially lower net increase in atmospheric
Can reduce dependence on fossil fuels
Disadvantages:
Need land, water and energy to produce
May compete with food production
Still releases when burned
Overall sustainability depends on how the crop is grown and processed
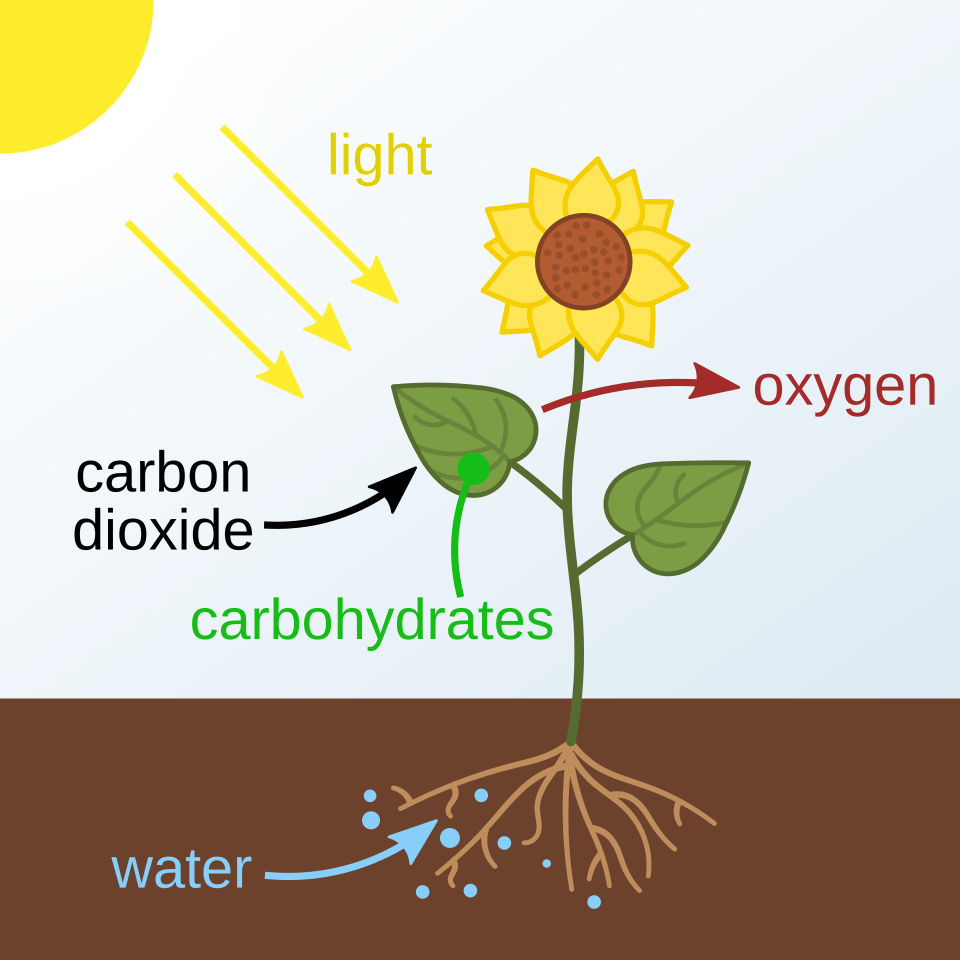
This diagram shows photosynthesis using sunlight, carbon dioxide and water to produce carbohydrates and oxygen. It is the key visual link for explaining why biofuels are described as using recently fixed carbon. Source
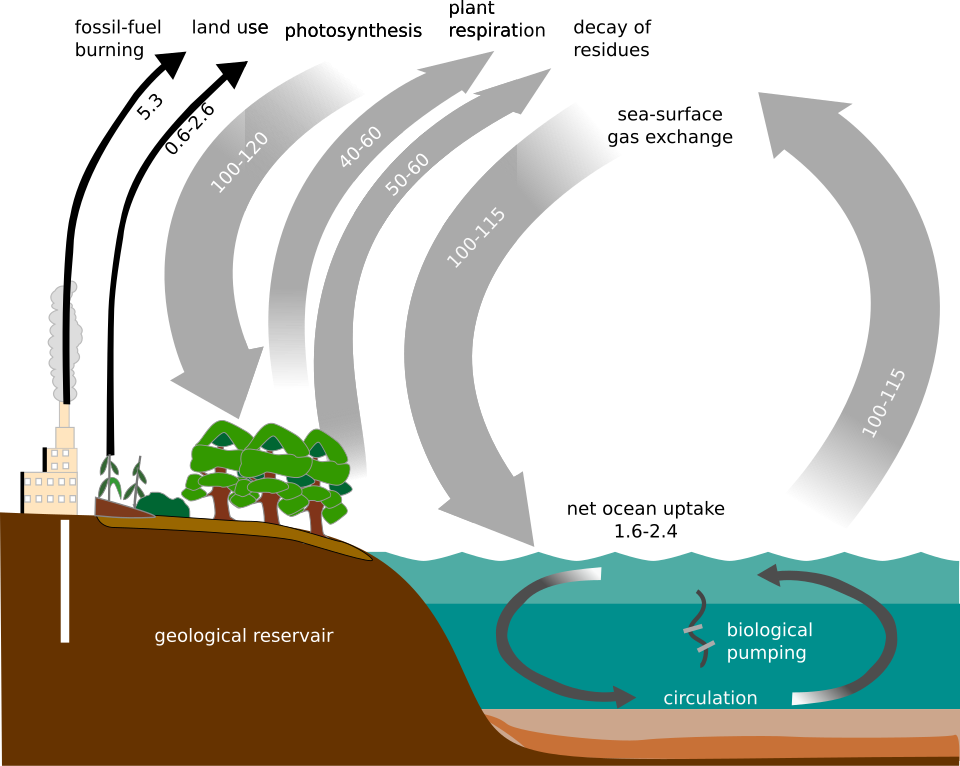
This diagram shows carbon moving between the atmosphere, organisms, and combustion processes. It helps compare fossil fuels with biofuels by showing whether carbon is being released from long-term stores or from a short-term cycle. Source
Renewable vs non-renewable
Renewable energy source = replaced on a human timescale.
Non-renewable energy source = not replaced quickly enough for continued use.
In this topic:
Biofuels = renewable
Coal, crude oil, natural gas = non-renewable
Evaluation questions often want a balanced judgement, not a one-sided answer.
Good exam style:
give one advantage
give one disadvantage
then make a reasoned conclusion
Fuel cells
A fuel cell converts chemical energy directly into electrical energy.
This is different from combustion in a power station, where chemical energy is first converted to heat.
IB examples: hydrogen fuel cells and methanol fuel cells.
Main advantage of fuel cells:
More direct energy conversion
Less reliance on combustion
Main limitations:
Fuel production and storage can be difficult
Some fuels are still obtained using fossil energy
Cost and infrastructure can limit use
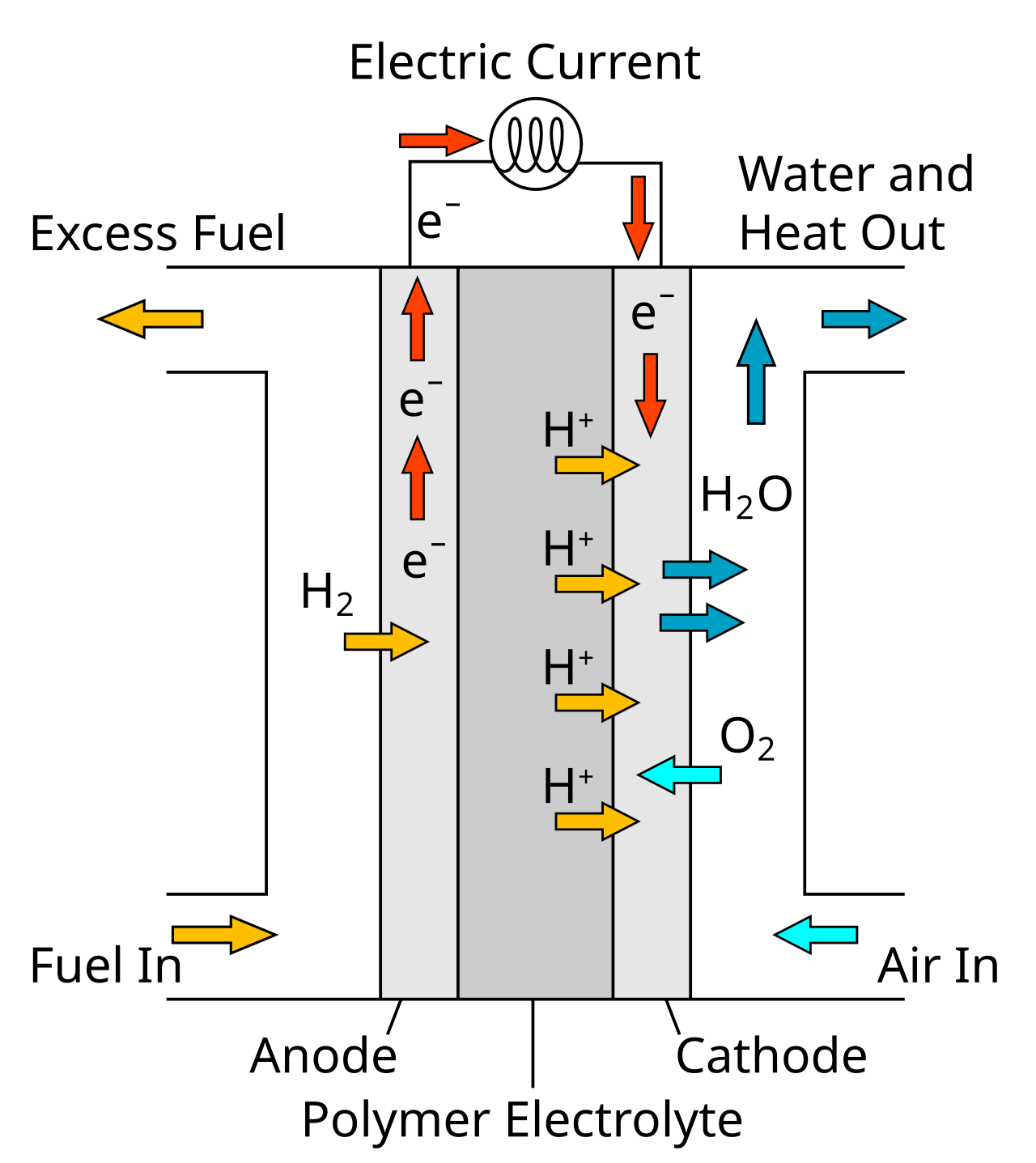
This schematic shows a hydrogen fuel cell with anode, cathode, electron flow through the external circuit, and ion movement through the membrane. It is ideal for learning how fuel cells produce electrical energy directly from a redox process. Source
Hydrogen fuel cell equations
You must be able to write half-equations and the overall equation.
Anode (oxidation):
Cathode (reduction):
Overall:
Key point: the only chemical product is water.
Important evaluation point:
The fuel cell itself does not emit when using hydrogen.
But the overall environmental benefit depends on how the hydrogen is produced.
Methanol fuel cell equations
You may also be asked for methanol as the fuel.
Anode (oxidation):
Cathode (reduction):
Overall:
Compared with hydrogen fuel cells:
Methanol is easier to store as a liquid.
It still produces .
Checklist: can you do this?
Write and balance equations for complete and incomplete combustion of hydrocarbons and alcohols.
Explain why incomplete combustion produces and , and why is dangerous.
Compare fossil fuels and biofuels using renewability, emissions, and energy released per unit mass.
Link rising levels to the greenhouse effect.
Deduce half-equations and the overall reaction for hydrogen and methanol fuel cells.