HL only — Reactivity 1.4: Entropy and spontaneity
Entropy, = measure of the dispersal/distribution of matter and/or energy in a system.
The more ways energy can be distributed, the higher the entropy.
For the same substance under the same conditions: gas > liquid > solid in entropy.
Spontaneous means thermodynamically feasible under the stated conditions.
Spontaneous does not mean fast — rate and spontaneity are different ideas.
In IB questions, always separate:
sign of
sign of
temperature,
Entropy trends you must know
Entropy usually increases when:
a solid melts
a liquid vaporizes
a solid sublimes
the number of gas moles increases
substances mix or dissolve
Entropy usually decreases when:
a gas condenses
a liquid freezes
the number of gas moles decreases
a system becomes more ordered
Biggest entropy clue in reactions: check change in gaseous particles first.
Common exam shortcut:
more gas particles produced
fewer gas particles produced
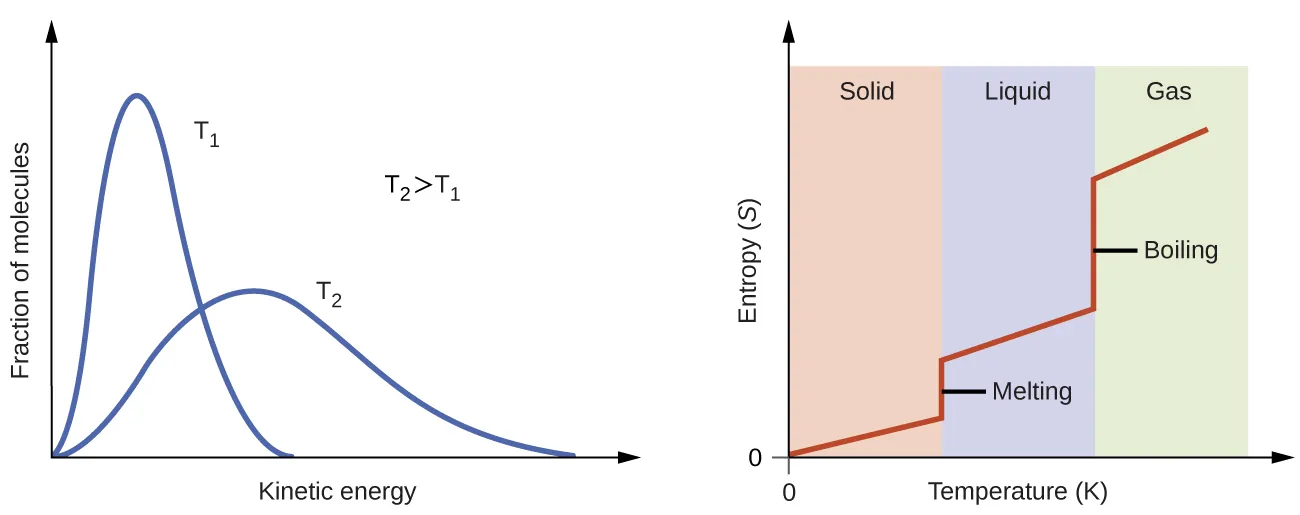
This figure shows that entropy rises as temperature increases and rises sharply during phase changes because particles gain more possible arrangements. It is excellent for linking state changes to increasing disorder/energy dispersal. Source
Predicting the sign of
Use:
values are given in the data booklet.
Include stoichiometric coefficients in the calculation.
Units of standard entropy, : J K mol.
Units of standard entropy change, : J K mol.
Exam habit: write the calculation clearly in two steps:
total products entropy
total reactants entropy
Then subtract: products − reactants.
Gibbs energy equation
Gibbs energy links enthalpy, entropy, and temperature:
Meanings:
= standard Gibbs energy change
= standard enthalpy change
= absolute temperature in kelvin
= standard entropy change
Units:
in kJ mol
usually in J K mol
in kJ mol
So before substituting, usually convert from J to kJ:
divide by 1000
Temperature must always be in K, never °C.
How to interpret
At constant pressure, a change is spontaneous if .
If , the change is non-spontaneous in the forward direction.
If , the system is at equilibrium.
IB wording to use:
negative = thermodynamically feasible / spontaneous
positive = not spontaneous under those conditions
A reaction can become more or less spontaneous as temperature changes because the term changes.
Temperature dependence of spontaneity
The sign combination of and tells you whether temperature matters.
Four essential cases:
, → always spontaneous
, → never spontaneous
, → spontaneous at low temperature
, → spontaneous at high temperature
Why?
negative helps make negative
positive makes more negative
Strong exam technique: do a sign analysis first before calculating.
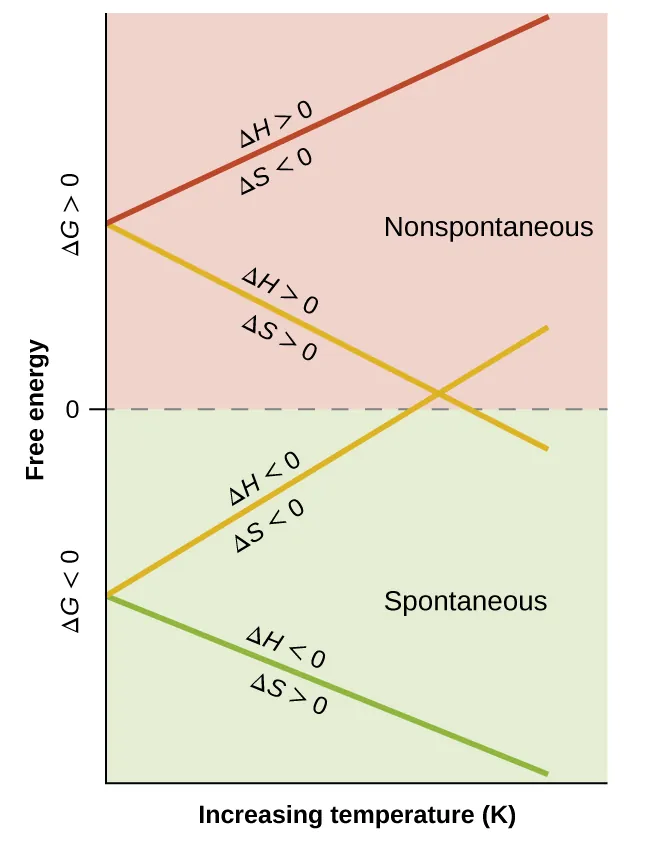
This page is useful for visualising why some reactions are spontaneous only at high or low temperature. It directly reinforces IB-style sign analysis for questions. Source
Finding the temperature where a reaction becomes spontaneous
At the boundary between spontaneous and non-spontaneous:
Therefore:
Use this only when asked for the temperature at which spontaneity changes.
Important conditions:
units must match before dividing
temperature answer must be in K
Interpretation:
if the question asks above or below this temperature, use the signs of and to decide.
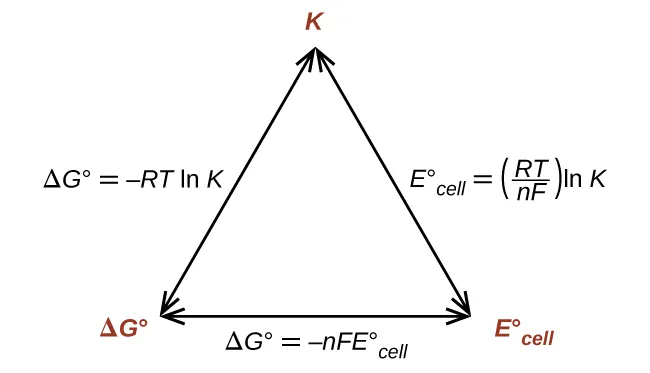
Equilibrium and Gibbs energy
As a reaction moves toward equilibrium, becomes less negative.
At equilibrium: .
Relationship with reaction quotient and equilibrium constant:
at equilibrium, , so
Meaning of :
→ , products favoured
→ , reactants favoured
→
This links thermodynamics to equilibrium position.
Exam calculation traps
Do not mix J and kJ.
Do not use °C in the Gibbs equation.
Do not forget stoichiometric coefficients in calculations.
A negative enthalpy alone does not guarantee spontaneity.
A reaction may be spontaneous but very slow.
refers to standard conditions; can change with actual conditions via .
Fast exam strategy
Step 1: identify what is being asked: sign, missing value, threshold temperature, or equilibrium link.
Step 2: check units.
Step 3: do a sign analysis before using numbers.
Step 4: use the correct equation:
Step 5: interpret the result in words, not just symbols.
This page is ideal for correcting a common IB error: confusing spontaneous with fast. It supports the exam language needed when explaining why feasibility and rate are not the same idea. Source
Checklist: can you do this?
Predict whether a change gives or from state change, mixing, dissolving, and change in gas moles.
Calculate and correctly with full unit conversions.
Interpret whether a process is spontaneous, non-spontaneous, or at equilibrium from the sign of .
Determine the temperature at which a reaction becomes spontaneous using .
Relate , , and to predict reaction direction and equilibrium position.
Memory box: absolute essentials
Entropy: measure of dispersal of matter/energy.
State order: solid < liquid < gas.
Spontaneous at constant pressure: .
Equilibrium: .
Core equation: .
High matters most when is significant.
Products favoured: , so .
This figure is helpful for the last part of the topic: connecting Gibbs energy to equilibrium position. It is especially useful for revision because it compresses several linked ideas into one diagram/table. Source