Discovery of the nucleus
Geiger–Marsden–Rutherford experiment: alpha particles were fired at thin gold foil.
Most particles passed straight through with little or no deflection.
A few were deflected through large angles.
A very small number bounced back.
Conclusion: the atom is mostly empty space, with positive charge and most mass concentrated in a tiny, dense nucleus.
Exam point: use the evidence to reject the plum pudding model and support the nuclear model.
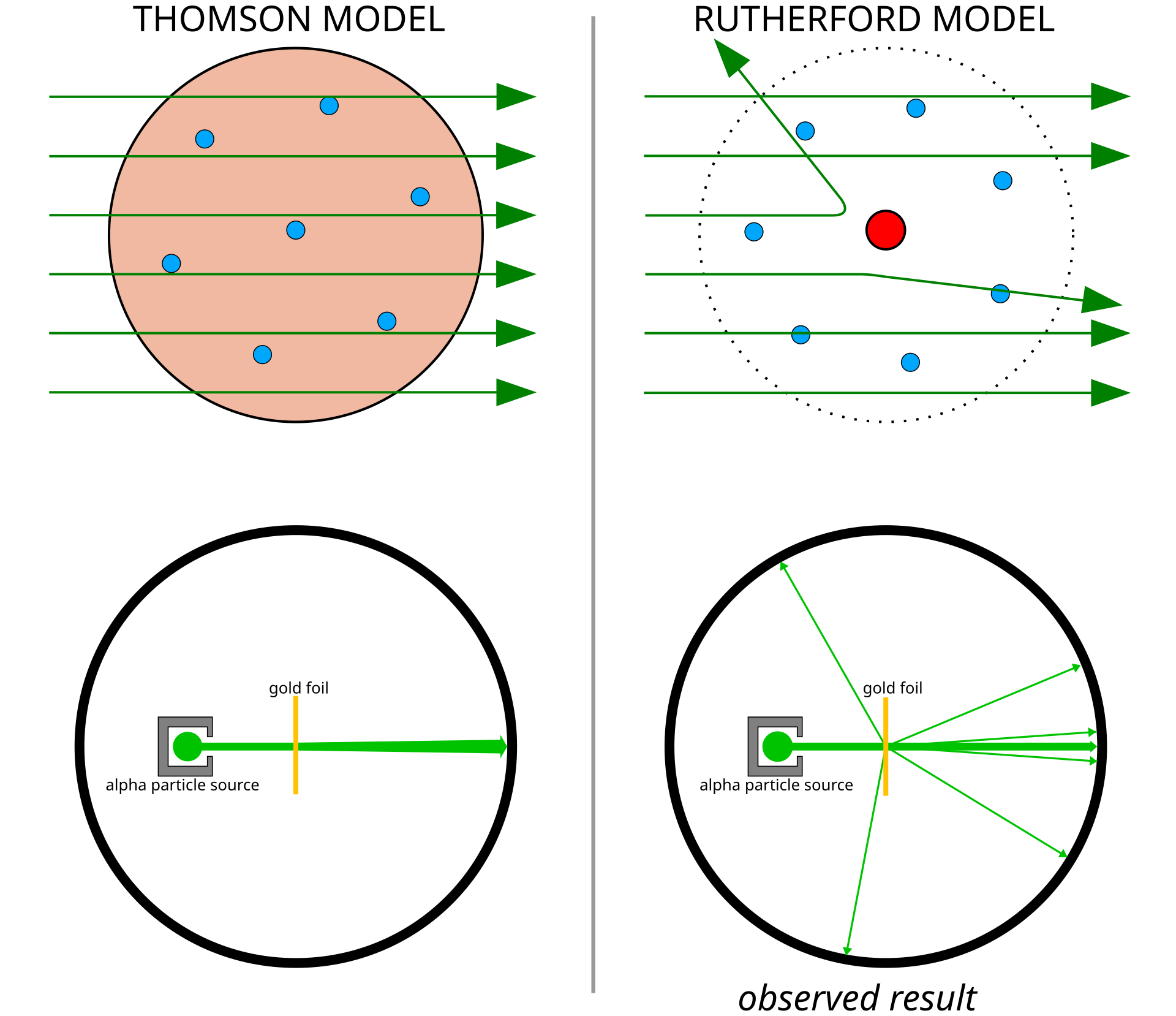
This diagram compares predicted and actual alpha-particle paths in the gold-foil experiment. It shows why only a small, concentrated nucleus can explain large-angle deflections and rare backscattering. Source
Nuclear notation and basic structure
Use nuclear notation: .
= nucleon number = protons + neutrons.
= proton number = number of protons.
= chemical symbol.
Number of neutrons = .
In a neutral atom, number of electrons = protons = .
Do not confuse nucleon number with mass number: in IB they refer to the same quantity here.
Atomic energy levels and photons
Electrons in atoms occupy discrete energy levels.
Electrons can move up an energy level by absorbing a photon.
Electrons move down an energy level by emitting a photon.
Photon energy is given by .
For a transition, photon energy = energy difference between levels: .
Larger energy gaps produce higher-frequency and shorter-wavelength photons.
Exam point: spectral lines are evidence that atomic energies are quantized, not continuous.
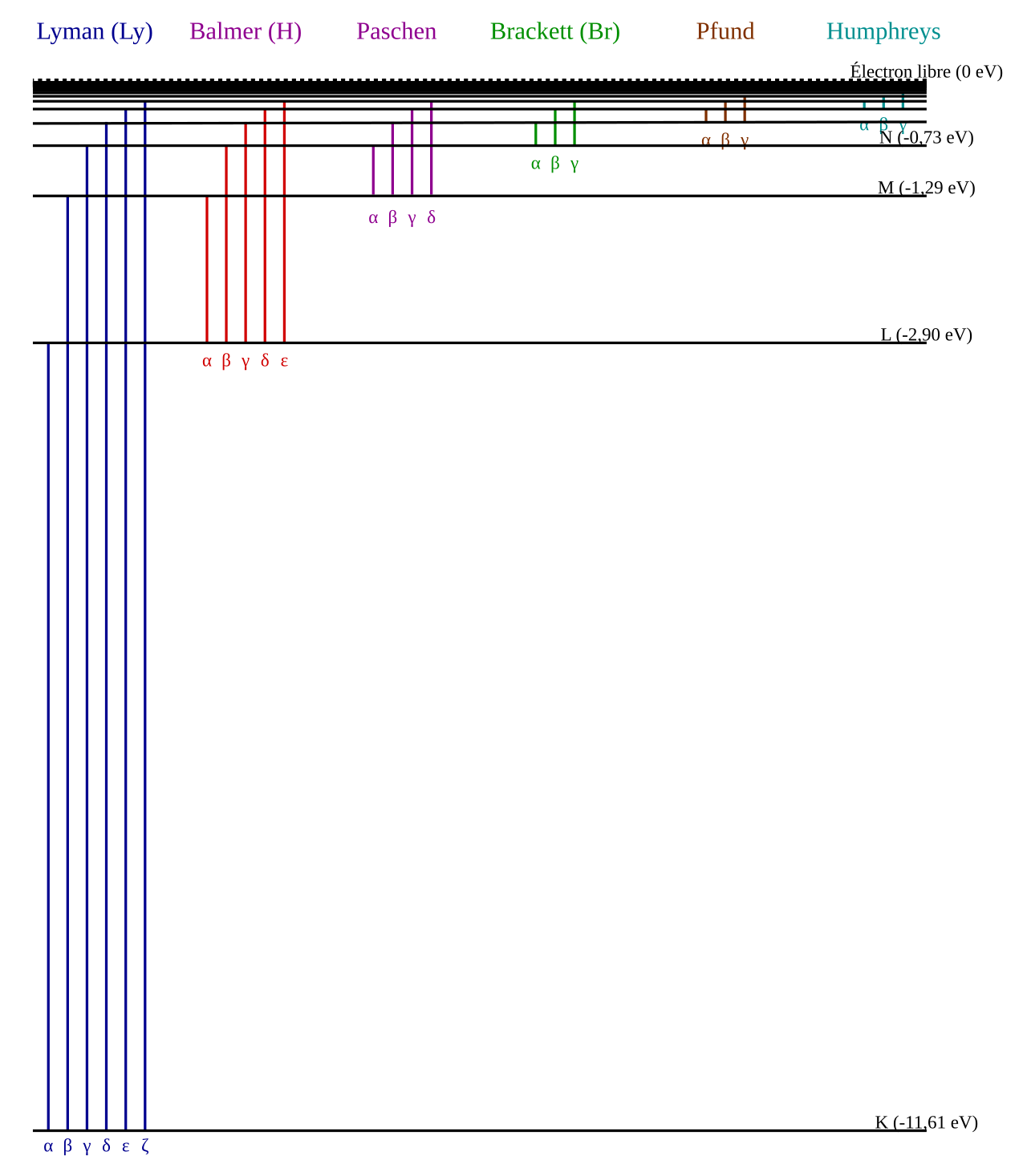
This spectrum shows that hydrogen emits light at specific wavelengths only, not a continuous spread. Each line corresponds to a particular electron transition between discrete energy levels. Source
Emission and absorption spectra
Emission spectrum: bright colored lines on a dark background.
Produced when excited atoms emit photons as electrons fall to lower levels.
Absorption spectrum: dark lines on a continuous background.
Produced when photons of specific energies are absorbed by atoms and electrons move to higher levels.
Each element has a unique set of spectral lines.
Therefore spectra can identify the chemical composition of a gas or star.
Exam point: same energy differences give matching emission and absorption line positions for the same element.
Calculations and exam skills
Use for photon energy.
Also use when wavelength is given.
Combine them when needed: .
In transition questions, set .
If energy is in eV, convert using .
A larger frequency means greater photon energy.
A shorter wavelength means greater photon energy.
HL only - nuclear size and density
Nuclear radius follows .
So radius increases slowly with nucleon number.
Since volume , nuclear volume is proportional to .
Therefore nuclear density is approximately constant for different nuclei.
Exam point: if increases by a factor of 8, radius increases by a factor of 2.
This supports the idea that nuclei have similar packing density.
HL only - Rutherford scattering at high energy
At low energies, alpha particles are treated as being repelled only by the electrostatic force.
At high energies, results can show deviations from Rutherford scattering.
Reason: alpha particles can get close enough for the strong nuclear force to become important.
This shows Rutherford’s pure electrostatic model is not complete at very small distances.
Exam point: mention deviation from expected scattering as evidence of additional nuclear interactions.
HL only - distance of closest approach
In a head-on collision, the alpha particle’s initial kinetic energy is converted into electric potential energy.
At the distance of closest approach, the alpha particle is momentarily at rest.
Use conservation of energy.
For an alpha particle and nucleus: .
Rearranged: .
IB guidance: use this only when the interaction is electrostatic repulsion.
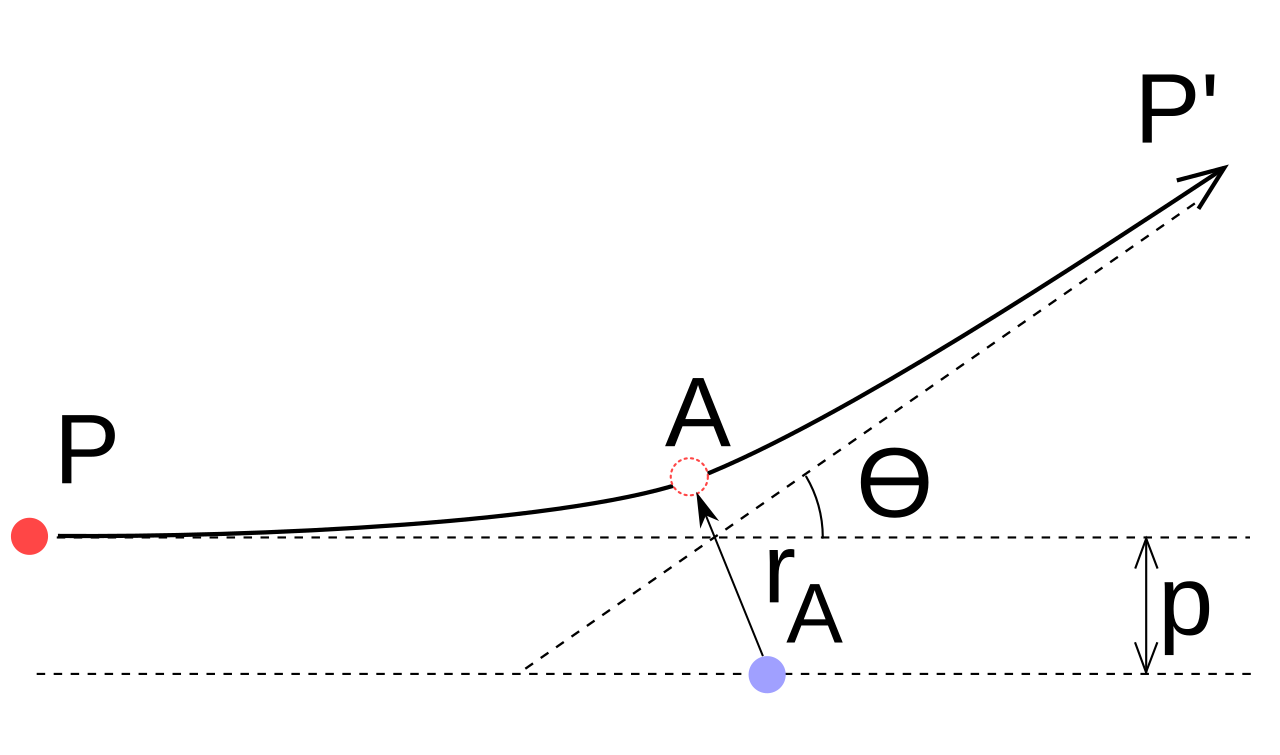
This geometry diagram helps visualize how an alpha particle is deflected by a positively charged nucleus. For a head-on approach, the turning point is the distance of closest approach, found using conservation of energy. Source
HL only - Bohr model for hydrogen
In the Bohr model, the electron in hydrogen occupies discrete circular orbits.
Allowed energies are .
is the principal quantum number.
Higher means higher energy and levels that are closer together.
Ionization corresponds to eV.
Transitions between levels explain the hydrogen line spectrum.
Exam point: the model works well for hydrogen, but fails fully for more complex atoms.
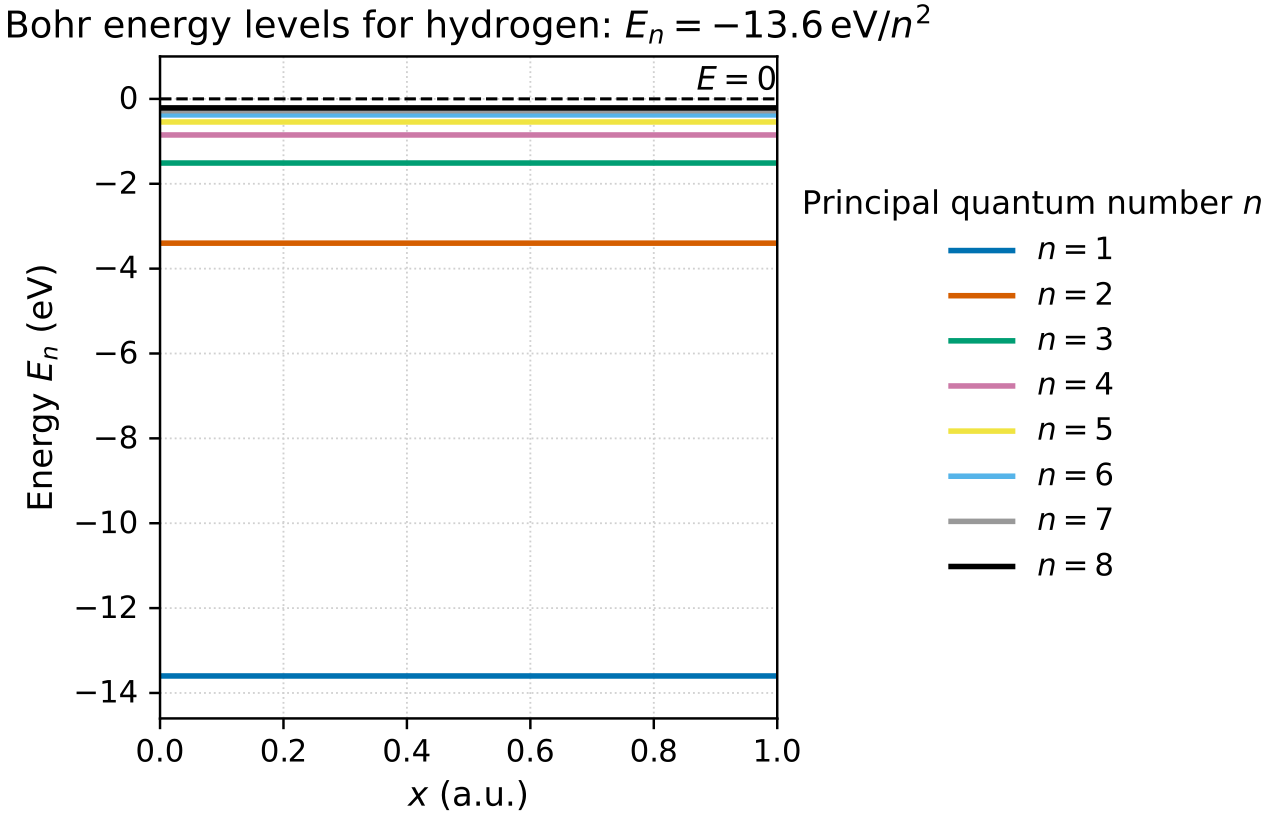
This energy-level diagram shows that hydrogen has quantized allowed energies. The levels get closer together as increases, approaching the ionization limit at eV. Source
HL only - quantization of angular momentum
In the Bohr model, allowed orbits arise because angular momentum is quantized.
Use .
Only orbits satisfying this condition are allowed.
Therefore the electron cannot have any radius or any energy.
This leads to discrete orbits and hence discrete energy levels.
Exam point: link quantized angular momentum to quantized energies.
Checklist: can you do this?
Interpret Rutherford scattering evidence to justify the existence of a small, dense nucleus.
Use notation to determine protons, neutrons, and electrons.
Calculate photon energy, frequency, or wavelength using and .
Explain how emission and absorption spectra show discrete energy levels and identify chemical composition.
Apply HL equations for , distance of closest approach, and Bohr energy levels where required.