Energy released in fission
Fission = splitting of a heavy, unstable nucleus into two smaller nuclei, releasing energy and usually neutrons.
Energy is released in both spontaneous fission and neutron-induced fission.
In exam questions, the energy released comes from a decrease in total mass of the system: mass defect.
Use to calculate the energy released.
In nuclear equations, conserve both nucleon number and proton number.
A typical fission process for uranium-235 is triggered when it absorbs a slow neutron, forms an unstable nucleus, then splits into fission fragments plus more neutrons.
The released energy appears mainly as kinetic energy of the fission fragments, with some carried by neutrons and emitted as gamma radiation.
If energy is needed in MeV, use .
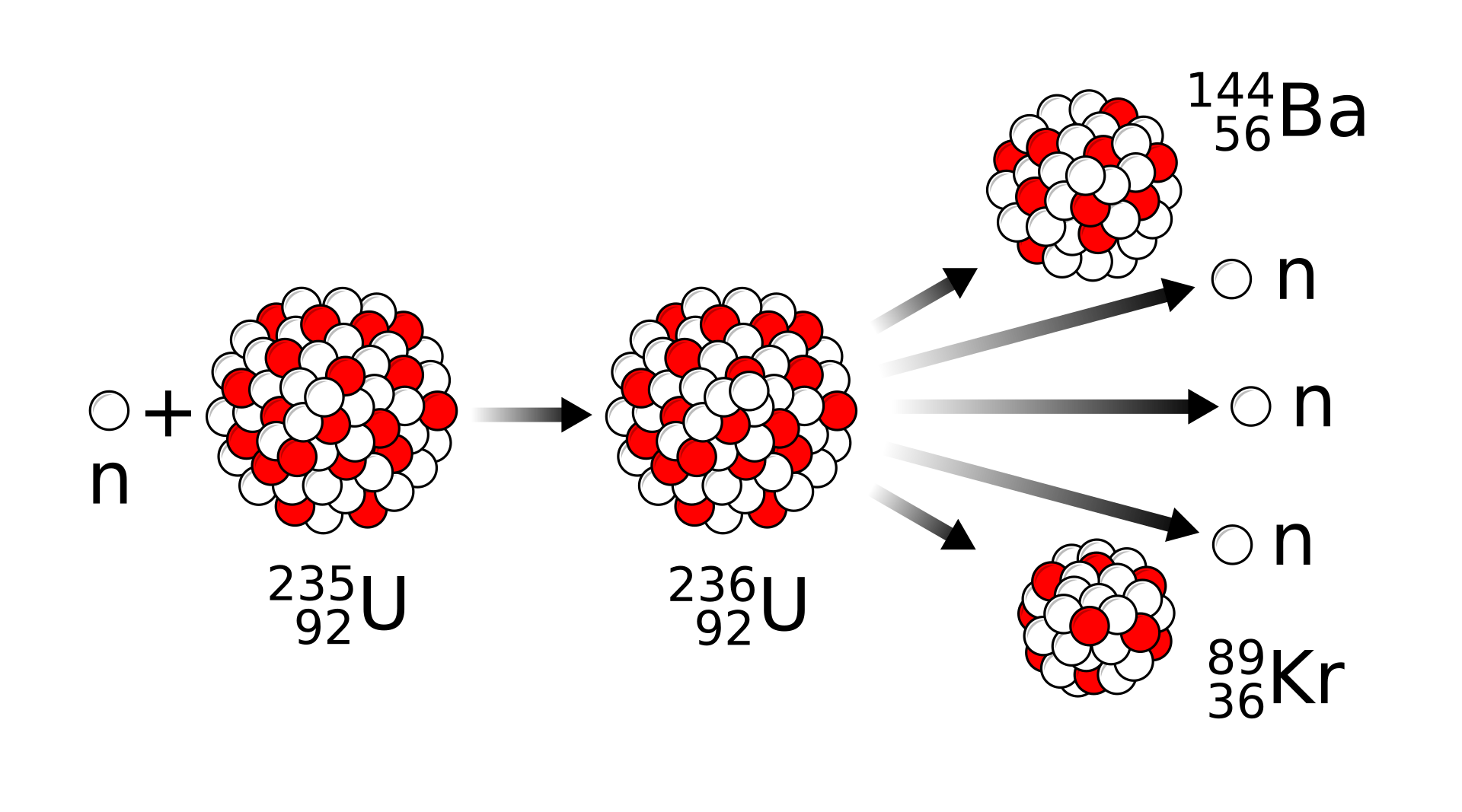
This diagram shows a typical uranium-235 fission reaction after neutron absorption. It is useful for seeing how one heavy nucleus splits into two lighter nuclei and releases additional neutrons. It directly supports balancing fission equations and identifying the source of released energy. Source
Chain reactions
A chain reaction occurs when neutrons produced by one fission event cause further fission reactions.
For a sustained chain reaction, at least one neutron from each fission must go on to trigger another fission.
If too many neutrons escape or are absorbed without causing fission, the chain reaction dies out.
If too many neutrons trigger more fissions, the reaction rate rises rapidly and becomes uncontrolled.
In a nuclear reactor, the aim is a controlled chain reaction so that energy is released at a steady rate.
In a nuclear weapon, the chain reaction is uncontrolled and extremely rapid.
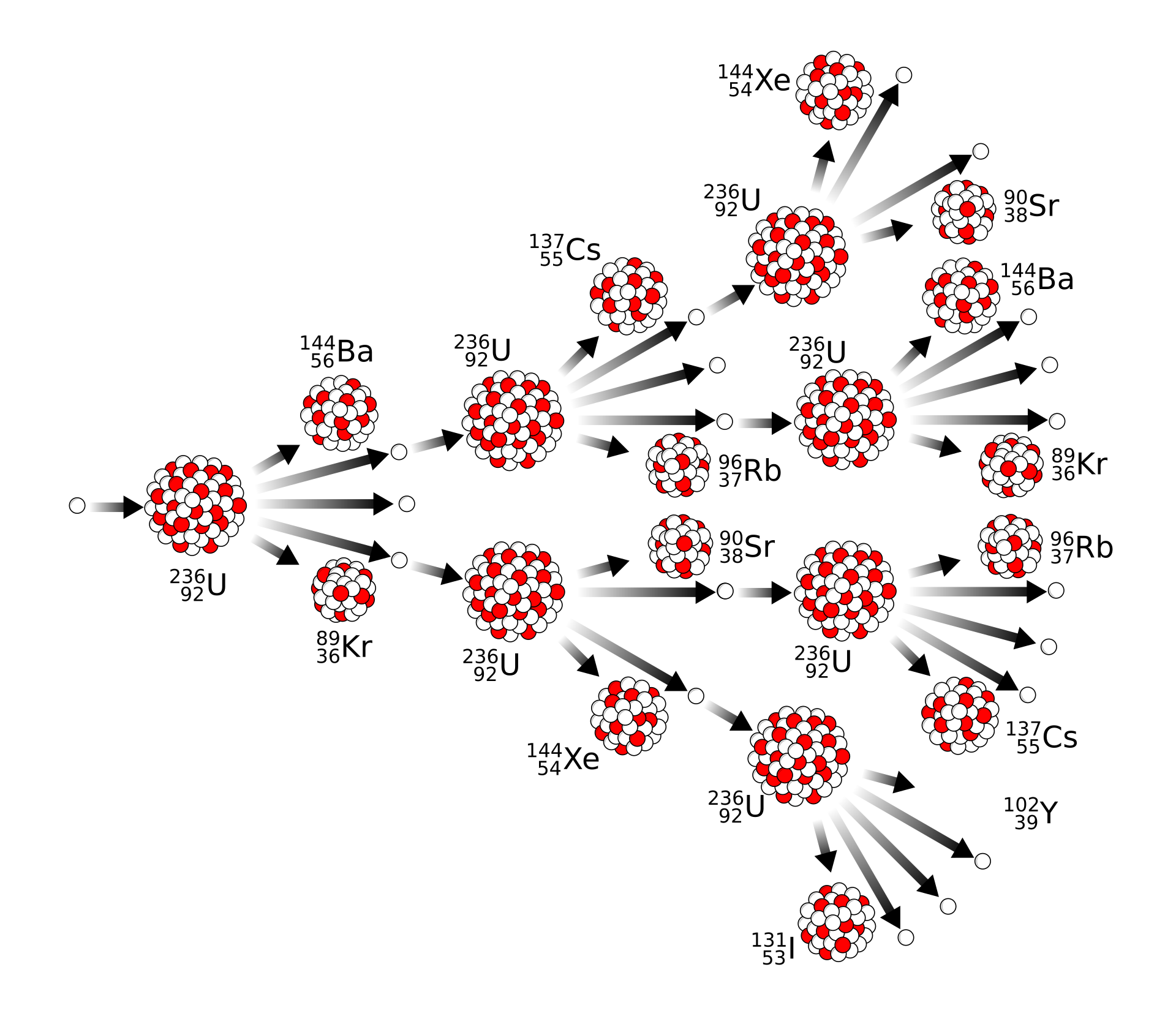
This illustration shows how neutrons from one fission can trigger further fissions, creating a chain reaction. It helps students visualize why reactor design must control the number of neutrons that continue the process. The repeated branching is the key idea behind sustained energy production. Source
Nuclear power plant components
Fuel rods contain the fissile material that undergoes fission.
Control rods absorb neutrons and are moved in or out to control the rate of fission.
More insertion of control rods means fewer neutrons available for further fission, so reactor power decreases.
A moderator slows down fast neutrons so they are more likely to cause further fission.
A heat exchanger transfers thermal energy from the reactor system to water that can produce steam.
The steam drives a turbine connected to a generator, producing electricity.
Shielding absorbs harmful radiation and protects workers and the surroundings.
The reactor must balance energy output, neutron economy, and radiation safety.
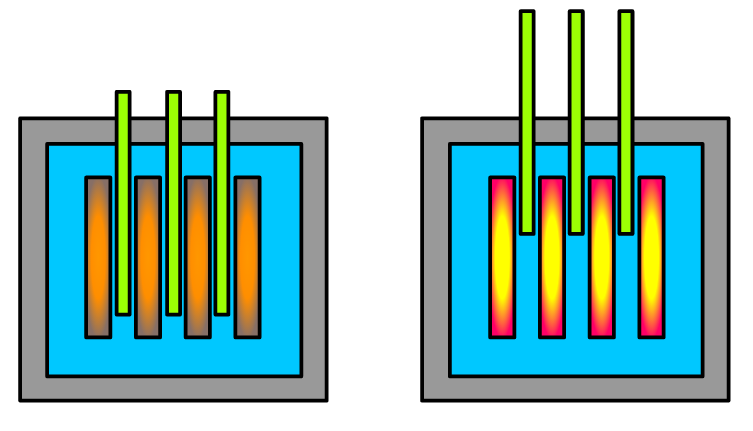
This schematic shows how control rods regulate reactor power by absorbing neutrons. It is especially useful for explaining why inserting rods further reduces the rate of the chain reaction. In exams, link control rods directly to reactor control and safety. Source
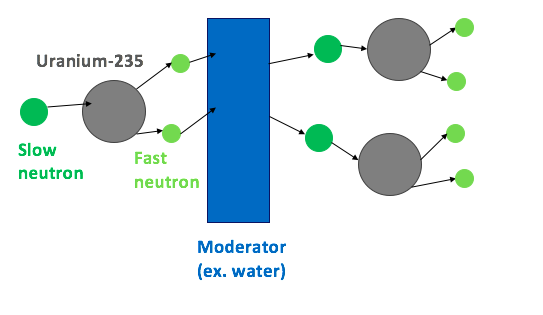
This figure shows the role of a moderator in slowing fast neutrons so they are more likely to cause further fission. It helps distinguish the job of the moderator from the job of control rods. Moderators make the chain reaction more efficient; control rods limit it. Source
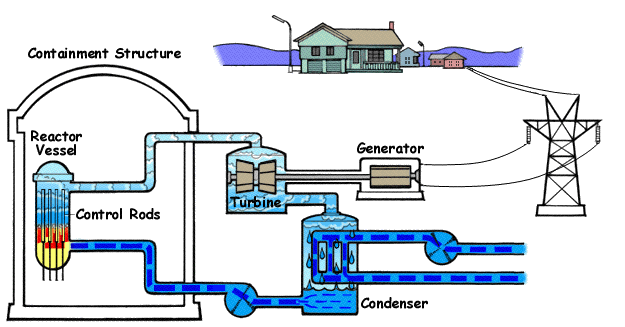
This diagram shows the major parts of a nuclear power plant, including the reactor, steam production, turbine and generator stages. It is ideal for linking fission in the core to electricity generation. Use it to explain the role of the heat exchanger and overall energy transfer pathway. Source
Fission products and waste management
Fission produces radioactive daughter nuclei known as fission products.
These products are often unstable and may emit ionizing radiation, so they require careful handling.
Waste management is necessary because some products remain radioactive for long periods.
Key concerns are storage, shielding, cooling, containment, and long-term environmental safety.
Spent fuel is typically stored first to allow radioactivity and thermal output to decrease.
Long-term storage of nuclear waste is an important disadvantage and should be considered in evaluation questions.
Exam answers should connect waste management to the properties of the products: radioactive, energetic, and potentially long-lived.
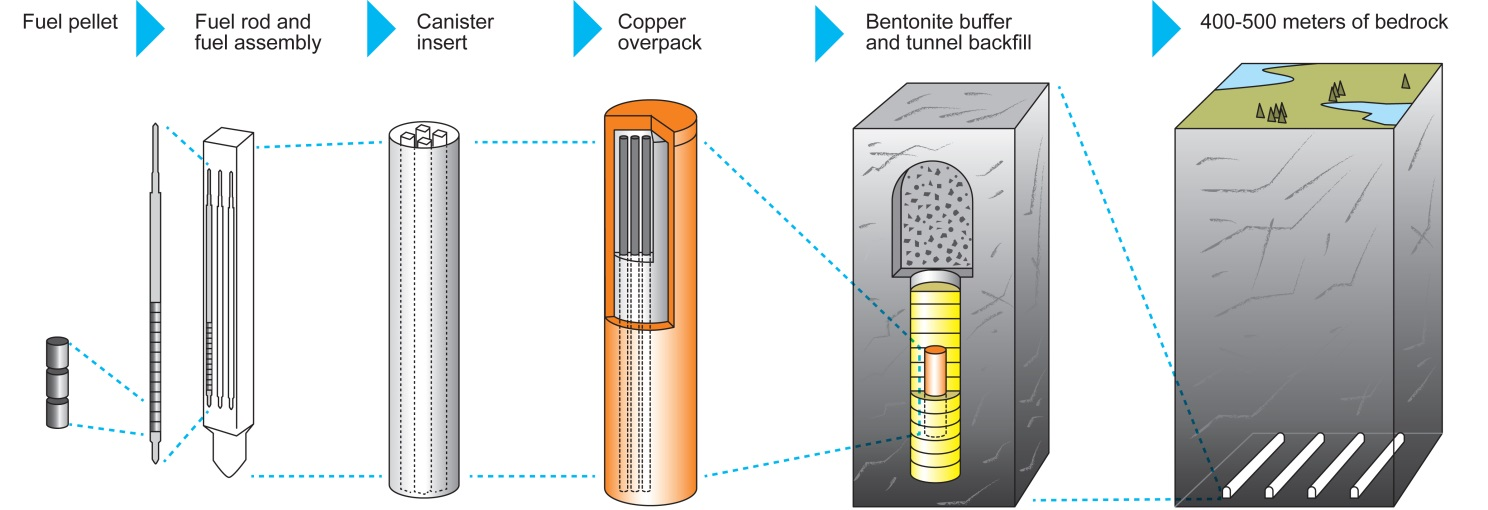
This resource explains how spent fuel is first stored, then managed for long-term disposal. It supports exam discussion of why fission products need shielding, cooling and secure containment. It is most useful for evaluation points about the impact of long-term nuclear waste storage. Source
Energy calculations in fission
Find the total mass of the reactants and the total mass of the products.
Calculate the mass defect: .
Then calculate the released energy using .
If masses are given in u, convert using .
To find energy in joules from MeV, use .
Always check that the nuclear equation is balanced first before calculating masses.
Be careful to include all emitted neutrons in the products.
In extended questions, relate large total energy output to the very large number of nuclei undergoing fission.
Checklist: can you do this?
Explain why energy is released in fission using mass defect and .
Balance a fission equation, including emitted neutrons.
Describe how a chain reaction becomes sustained, controlled, or uncontrolled.
State the roles of control rods, moderators, heat exchangers, and shielding in a nuclear power plant.
Discuss why fission products require careful long-term waste management.