Stability, isotopes and binding energy
Isotopes are nuclei with the same proton number but different neutron number .
A nucleus is stable when the strong nuclear force can overcome electrostatic repulsion between protons.
The strong nuclear force is short-range and attractive between nucleons.
Mass defect: the nucleus has less mass than its separated nucleons.
Nuclear binding energy = energy needed to separate a nucleus into free nucleons.
Use mass–energy equivalence: .
In calculations, binding energy comes from the mass defect.
Binding energy per nucleon is a measure of nuclear stability.
The binding energy per nucleon curve rises for small nuclei, reaches a maximum near iron, then is approximately constant above nucleon number 60 before slowly decreasing.
Higher binding energy per nucleon means a more stable nucleus.
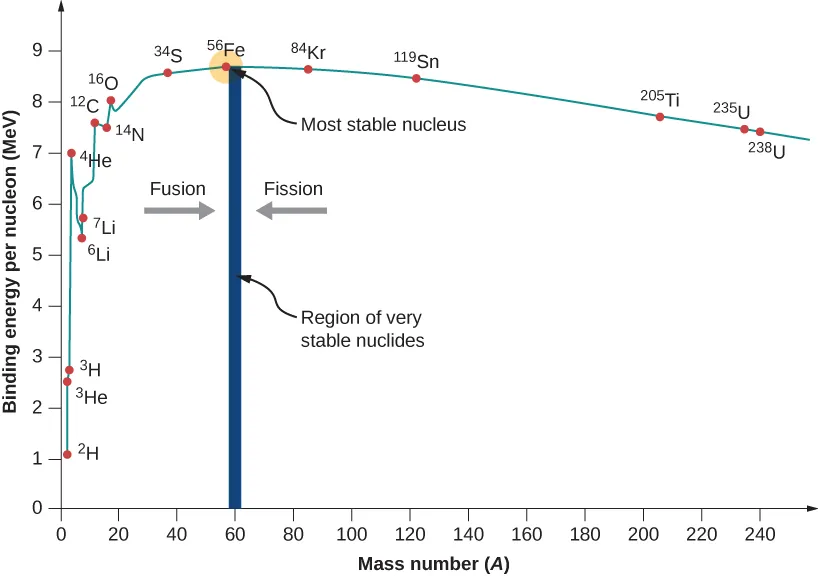
This graph shows how binding energy per nucleon varies with mass number and why nuclei near iron are especially stable. It is useful for linking stability to the shape of the curve and for explaining why changes in nuclear structure can release energy. Source
Why some nuclei are stable (HL only)
HL only: stability depends strongly on the neutron-to-proton ratio.
For small nuclei, stability is often close to .
For larger nuclei, stable nuclei need more neutrons than protons to reduce the effect of proton–proton repulsion.
HL only: nuclei above the line of stability tend toward decay; nuclei below it tend toward decay.
HL only: evidence for the strong nuclear force comes from the fact that nuclei exist at all, despite strong electrostatic repulsion between protons.
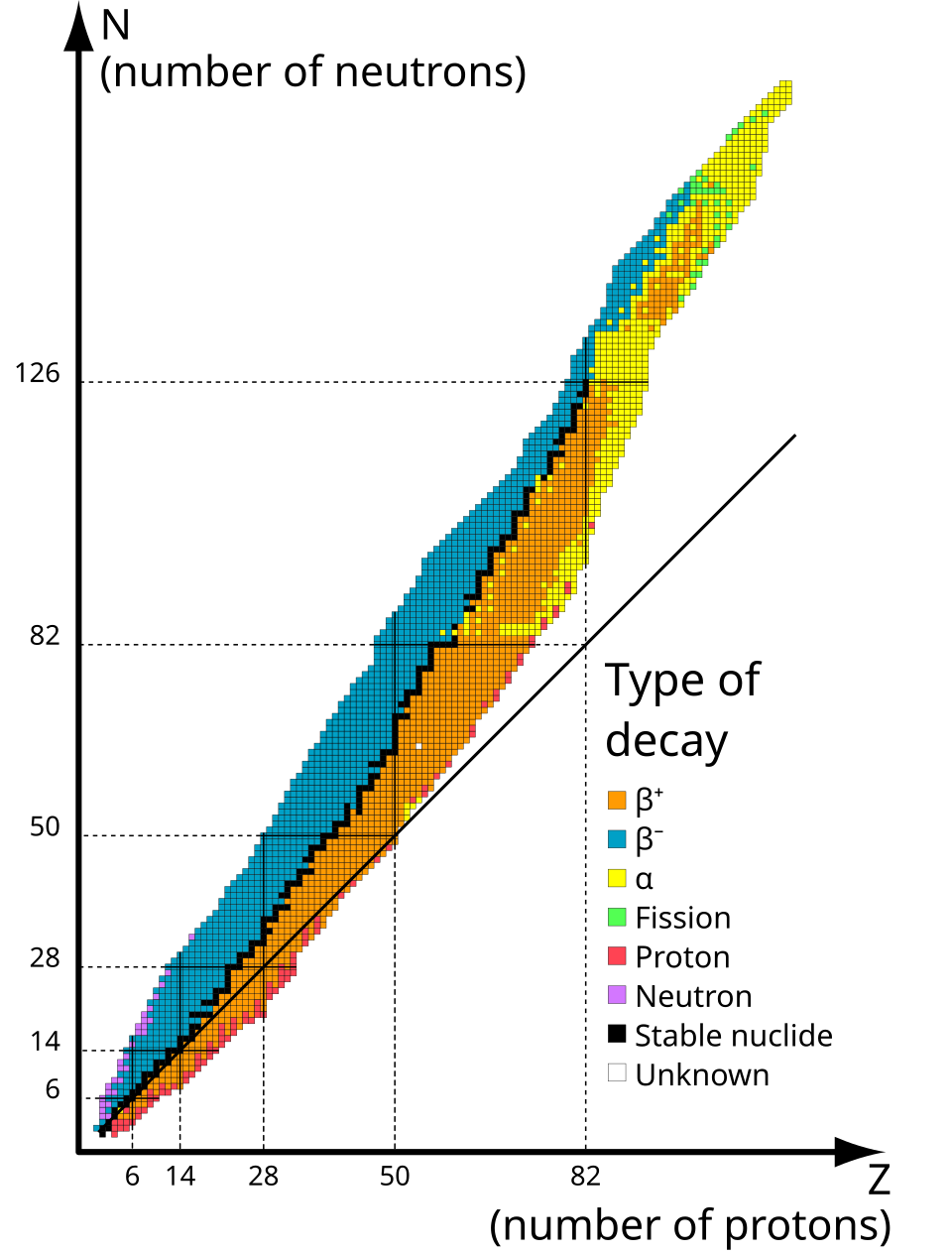
This chart shows the line of stability and how unstable nuclei on either side tend toward different decay modes. It is especially useful for understanding why heavier stable nuclei need more neutrons than protons. Source
Radioactive decay: core ideas
Radioactive decay is random and spontaneous.
You cannot predict when an individual nucleus will decay.
For a large sample, decay is predictable statistically.
In decay questions, conserve nucleon number and charge / proton number .
Parent nucleus decays into a daughter nucleus.
The nucleus may emit , , or radiation.
Neutrinos and antineutrinos are involved in beta decay.
Alpha, beta and gamma decay equations
Alpha decay: nucleus emits a helium nucleus .
In decay, decreases by 4 and decreases by 2.
General form: .
decay: a neutron changes into a proton, emitting an electron and an antineutrino.
In decay, stays the same and increases by 1.
General form: .
decay: a proton changes into a neutron, emitting a positron and a neutrino.
In decay, stays the same and decreases by 1.
General form: .
Gamma decay is emission of a high-energy photon from an excited nucleus.
In decay, and stay unchanged.
General form: ^A_ZX^ \rightarrow ^A_ZX + \gamma*.
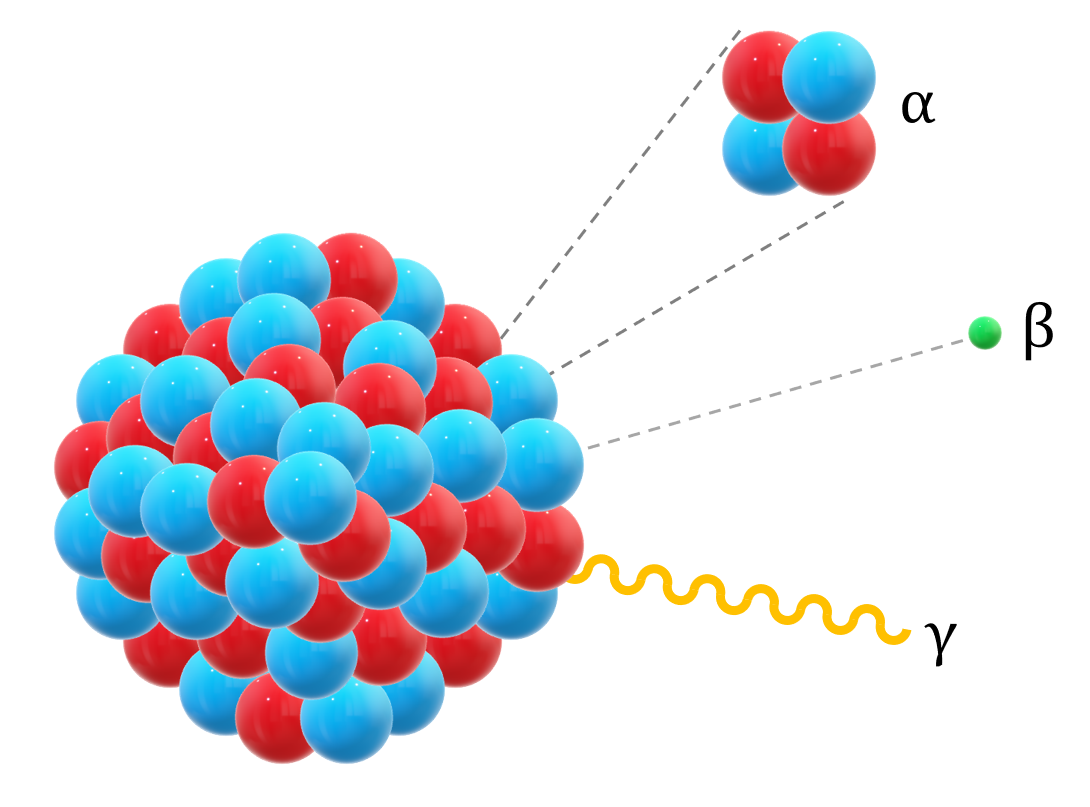
This diagram compares alpha, beta, and gamma emission directly from the nucleus. It is useful for visualizing which particles are emitted and how the nucleus changes in each process. Source
Penetrating and ionizing power
Alpha radiation is most strongly ionizing but least penetrating.
Beta radiation has moderate ionizing ability and moderate penetration.
Gamma radiation is least ionizing but most penetrating.
Typical shielding: alpha stopped by paper / skin, beta by thin aluminium, gamma needs thick lead or concrete.
Exam link: greater ionization generally means shorter range in matter.
Background counts must be considered when measuring low count rates.
Nuclear energy levels and spectra (HL only)
HL only: alpha and gamma spectra are discrete, showing discrete nuclear energy levels.
HL only: a nucleus can only emit certain allowed alpha energies or gamma photon energies.
HL only: the continuous beta spectrum shows that beta decay energy is shared.
HL only: this is evidence for the neutrino, which carries away part of the energy and momentum.
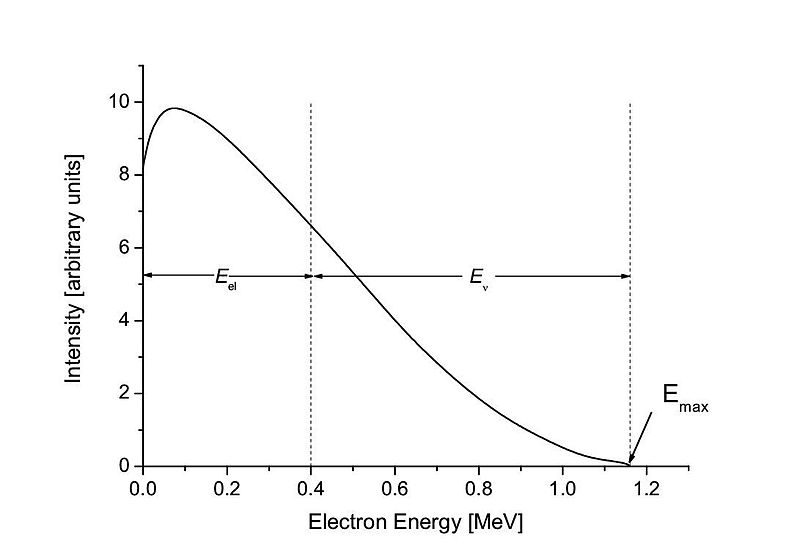
This graph shows the continuous energy spectrum of beta decay, unlike the discrete energies expected if only the electron were emitted. It supports the idea that a neutrino also carries away energy. Source
Activity, count rate and half-life
Activity = rate of decay of a radioactive sample.
Unit of activity: becquerel (Bq), where .
Count rate is what the detector records; it may be less than the true activity.
Background radiation adds extra counts, so use corrected count rate = measured count rate background count rate.
Half-life is the time for the number of undecayed nuclei, activity, or corrected count rate to fall to half its value.
For integer numbers of half-lives, repeatedly divide by 2.
Example pattern: .
Since activity is proportional to number of undecayed nuclei, and fall with the same half-life.
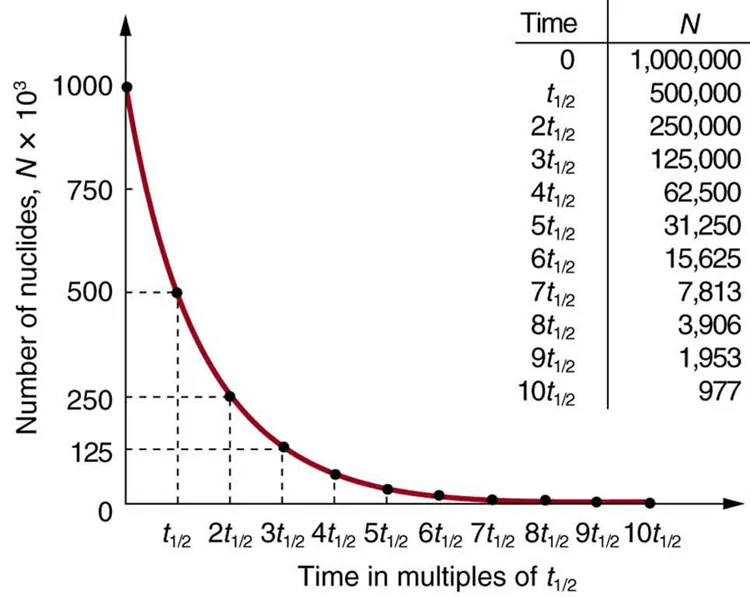
This page explains how activity and the number of undecayed nuclei both decrease with time. It is useful for connecting half-life calculations to the shape of the decay curve. Source
Decay law and decay constant (HL only)
HL only: radioactive decay follows .
HL only: is the decay constant.
HL only: activity can be written as .
HL only: half-life and decay constant are related by .
HL only: use the exponential law for arbitrary time intervals, not just integer multiples of the half-life.
HL only: approximates the probability of decay per unit time only when is very small.
Exam method and common traps
Always check whether the question wants activity, count rate, corrected count rate, remaining nuclei, or fraction remaining.
In decay equations, conserve total nucleon number and total charge.
Do not confuse count rate with activity.
Subtract background radiation before finding half-life from data.
On graphs, find half-life from the time for the corrected count rate or activity to halve.
If time is not an exact multiple of half-life, HL students should use the exponential equation.
A more stable nucleus usually has a higher binding energy per nucleon.
Applications and contexts
Medical tracers / treatment: choose isotopes with suitable half-life and penetration.
Leaks in underground pipes: use radioisotopes that are detectable outside the pipe.
Thickness control: use absorption of beta or gamma radiation through material.
Radioactive dating: choose isotopes with a half-life comparable to the age being measured.
Checklist: can you do this?
Write and balance nuclear equations for , , , and decay.
Calculate mass defect, binding energy, and binding energy per nucleon.
Interpret the binding energy per nucleon curve and explain relative nuclear stability.
Determine half-life from data or graphs, including using background-corrected count rate.
Use , , and for HL questions.