AP Syllabus focus: ‘Estimate an element’s average atomic mass by finding a weighted average of isotopic masses using each isotope’s mass and its relative abundance.’
Average atomic mass connects the real isotopic mixture found in nature to a single number on the periodic table. AP Chemistry expects you to compute it using isotopic masses and their natural abundances.
What “Average Atomic Mass” Means
Elements typically exist as a mixture of isotopes (atoms with the same number of protons but different numbers of neutrons). Because isotopes have different masses, the “atomic mass” reported for an element is a weighted value.
Average atomic mass: The weighted average mass of an element’s naturally occurring isotopes, based on each isotope’s mass and its relative abundance.
A key idea is that more abundant isotopes contribute more strongly to the average than rare isotopes.
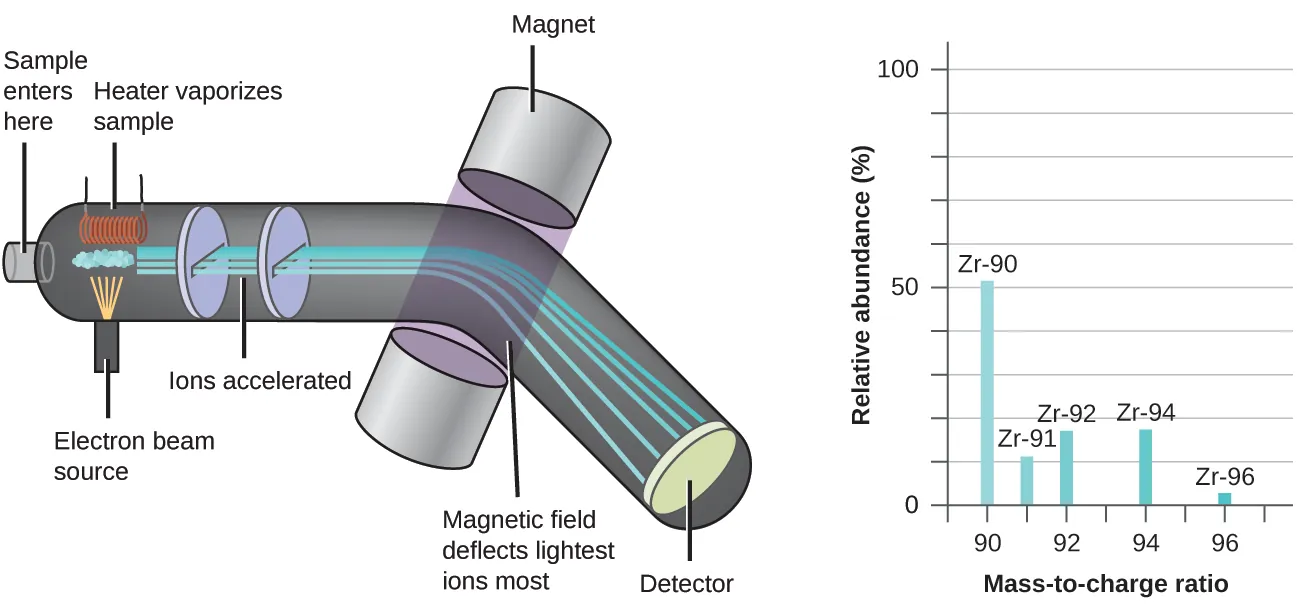
This diagram shows a mass spectrum, where each peak corresponds to ions with a particular mass-to-charge ratio () and the peak height represents relative abundance. In isotope problems, these peak heights (or the percentages derived from them) provide the fractional abundances used in the weighted-average calculation. Source
Isotopic Mass vs. Abundance
Isotopic mass: the mass of one atom of a specific isotope (commonly given in amu).
Relative (percent) abundance: how common that isotope is in nature.
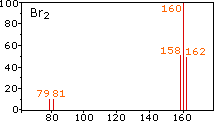
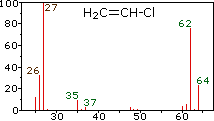
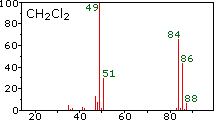
These example mass spectra illustrate how isotopes appear as distinct peaks, with peak heights proportional to their relative abundances. For chlorine-containing species, the characteristic two-peak pattern (separated by 2 amu) makes it easy to read off approximate isotope ratios that feed directly into the weighted-average atomic mass calculation. Source
Abundances should total 100% (or 1.00 if written as fractions), allowing for rounding.
Relative abundance: The fraction or percentage of atoms of an element that occur as a particular isotope in a natural sample.
Weighted Average Method
A “weighted average” multiplies each isotope’s mass by how common it is, then sums those contributions.
= average atomic mass of the element (amu)
= isotopic mass of isotope (amu)
= fractional abundance of isotope (unitless), where
This relationship is the computational heart of the topic: convert abundances into fractions, apply the sum, then interpret the result as the element’s average mass per atom.
Process (No Numbers)
Identify each isotope present and its isotopic mass.
Obtain each isotope’s relative abundance.
Convert percent abundance to fractional abundance by dividing by 100.
Multiply each isotopic mass by its fractional abundance.
Add the products to get the average atomic mass.
Units, Rounding, and Interpretation
Average atomic mass is commonly reported in amu (also called atomic mass units).
The value usually lies between the masses of the most common isotopes and is often closer to the most abundant isotope’s mass.
Use appropriate significant figures based on the given isotopic masses and abundances; rounding early can introduce avoidable error.
If abundances do not sum exactly to 100% due to rounding, treat the discrepancy as rounding error unless instructed otherwise.
Common Pitfalls to Avoid
Using percent abundance directly as a fraction (e.g., using 75 instead of 0.75).
Taking a simple average of isotopic masses (ignores abundance).
Forgetting that fractional abundances must sum to 1.00.
FAQ
They are used interchangeably in many textbooks and periodic tables.
Strictly, “atomic weight” can refer to a dimensionless relative quantity, but AP-level problems treat the listed periodic-table value as the average isotopic mass in amu for calculations.
No. Mass number is an integer count of protons + neutrons.
Isotopic mass (in amu) reflects measured atomic mass and includes nuclear binding effects, so it is typically not an integer.
First, check for rounding in the given data.
If the total is very close to 100%, proceed as given. If it is noticeably off, you may:
convert to fractions and normalise by dividing each by the total fraction (only if instructed or clearly necessary).
Yes, slightly. Natural isotopic abundances can vary by source and measurement method.
These variations are usually small; AP questions typically assume the provided abundances are exact for the problem.
Because it is a weighted average.
The isotope with the larger fractional abundance has more “weight” in the sum $\sum (m_i \times f_i)$, pulling the average closer to its isotopic mass.
Practice Questions
(1–3 marks) Chlorine exists naturally as two isotopes, and . Explain why the atomic mass of chlorine on the periodic table is not a whole number.
Atomic mass is an average of isotopic masses (1)
Isotopes have different masses and occur in different abundances, so the weighted average is not an integer (1)
(4–6 marks) An element has two naturally occurring isotopes with isotopic masses 9.012 amu and 10.013 amu. The relative abundance of the 10.013 amu isotope is 19.9%.
(a) State the fractional abundance of the 9.012 amu isotope.
(b) Calculate the average atomic mass of in amu.
(c) State, with a reason, which isotope contributes more to the average atomic mass.
(a)
(1)
(b)
Uses weighted mean: (2)
Correct numerical value with appropriate rounding (1)
(c)
Identifies 9.012 amu isotope (or the more abundant isotope) contributes more (1)
Reason: it has the larger fractional/percent abundance (1)

