AP Syllabus focus: ‘For AP Chemistry, you do not need to interpret spectra with multiple elements or peaks from species other than singly charged monatomic ions.’
Mass spectrometry is rich with experimental details, but AP Chemistry narrows what you must interpret. This page clarifies exactly which mass-spectrum features are out of scope and how to avoid overthinking exam items.
What is explicitly excluded (know this wording)
The AP expectation is restrictive: you are not responsible for interpreting either of the following in mass spectra:
Spectra containing multiple elements (i.e., overlapping isotope patterns from different elements)
Peaks from species other than singly charged monatomic ions (e.g., molecular ions, fragments, multiply charged ions)
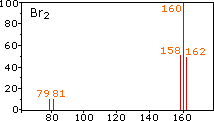
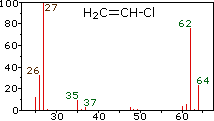
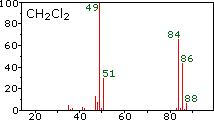
Example mass spectra illustrating isotope signatures for halogens, including chlorine’s two stable isotopes with peaks separated by 2 amu (consistent with and ). The page also shows how molecular/fragment ions create additional peaks, which is exactly the kind of complexity AP Chemistry tells you not to interpret. Source
This means AP questions will be written so that meaningful interpretation does not require sorting out complicated mixtures of elements or non-monatomic species.
The only kind of “species” you’re expected to read
AP problems will limit spectra to singly charged monatomic ions of a single element, so each major peak corresponds to an isotope of that element.
Monatomic ion: An ion made from a single atom that has gained or lost electrons (for example, ).
A “monatomic ion” constraint eliminates molecular composition issues (no bonds, no fragments), so the spectrum is essentially a direct window into isotopes.
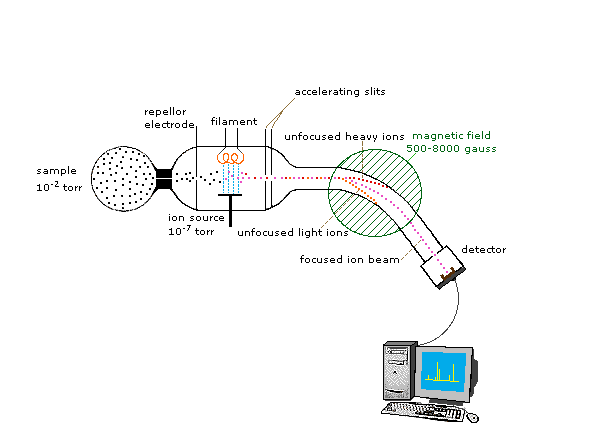
Schematic diagram of a mass spectrometer showing the functional blocks: ion source (creates ions), mass analyzer (separates ions by ), and detector (records ion counts to generate a spectrum). This reinforces why AP-style isotope questions can treat peaks as direct signals from isolated ions rather than needing a detailed model of the instrument. Source
Singly charged (1+) ion: An ion with a net charge of , meaning one electron has been removed; its measured equals its mass number (in amu) divided by 1.
You do not need to compute or interpret how charge states change the location of peaks; AP avoids that by sticking to ions.
What “multiple elements” would look like (and why AP avoids it)
Real spectra can involve more than one element (intentionally or via contamination), creating:
Peak sets that do not match a single isotope pattern
Overlapping peaks at similar masses from different elements
Ambiguity about which element “owns” a peak without extra context
On the AP Exam, if a spectrum is provided for an element, it will be designed so that you can treat it as one element’s isotopes only, without needing to deconvolute mixed signals.
Common real-world peaks you can ignore on AP
If a prompt mentions or shows features like these, AP will not require you to interpret them as part of answering:
Molecular ions (e.g., ) that reflect bonded species rather than atoms
Fragment ions (pieces formed when molecules break apart)
Multiply charged ions (e.g., , ), which shift peaks to lower
Adducts or clusters (species combined with something else during ionisation)
Peaks from instrument background or impurities, unless the problem explicitly reframes the task
The exam’s intent is to keep the reasoning focused on isotope identity and relative abundance for a single element, not on instrumentation artifacts or complex ion chemistry.
How to stay within scope when you see a spectrum
Use this quick “scope filter” before interpreting anything:
Check whether the context says the sample is a single element
Look for signals that would imply non-monatomic species (odd masses, unexpected extra peak families, charge labels)
If the task can be answered by considering only monatomic isotopes, do exactly that and ignore the rest
When in doubt, prioritize the simplest reading consistent with the AP constraint: one element, isotopes only, ions only.
FAQ
Some ionisation methods can remove more than one electron, especially from atoms with higher energies involved.
Multiply charged ions shift to lower $m/z$, complicating patterns—one reason AP restricts items to $1+$.
Mixed-element spectra arise from impurities, incomplete separation, or analysing compounds rather than pure elements.
You may see distinct isotope patterns that cannot belong to the same element.
Not typically. AP framing for elemental spectra assumes cations (often shown as $^+)$ and focuses on isotopes of one element.
Negative-ion modes add extra interpretation that is beyond the intended scope.
Not perfectly. Detector response, ionisation efficiency, and overlapping signals can distort intensity.
AP will avoid relying on subtle intensity distortions when asking you to interpret spectra.
Higher resolution separates nearly identical $m/z$ values into distinct peaks; low resolution can merge them.
AP questions are designed so peak identification does not depend on advanced resolution considerations.
Practice Questions
(1–3 marks)
A spectrum is described as having peaks for , , and . Is this spectrum within AP interpretation scope? Give one reason.
Yes, within scope (1)
It contains only a single element and singly charged monatomic ions (1)
(4–6 marks)
A mass spectrum for a sample contains peaks labelled and , plus smaller peaks labelled and .
(a) Identify which peaks you are expected to interpret for AP Chemistry.
(b) State why the other peaks are out of scope, using AP language.
(c) State one valid conclusion you can still make using only the in-scope peaks.
(a) and only (2; 1 each)
(b) is not monatomic (1) and is not singly charged (1)
(c) Any one: chlorine has (at least) two isotopes; or the sample shows isotopes of a single element; or relative abundances can be compared qualitatively from the two peaks (2; 1 for a valid conclusion, 1 for linking it to in-scope peaks)

