AP Syllabus focus: ‘For covalent bonds, bond length depends on core size and bond order; higher bond order bonds are shorter and have greater bond energies than single bonds.’
Covalent bond lengths are measurable structural features that reflect how strongly two nuclei are held together by shared electrons. In AP Chemistry, focus on how atomic size and bond order systematically control bond length and strength.
Core ideas: what controls covalent bond length?
Bond length is the equilibrium distance between the nuclei of two covalently bonded atoms; it occurs at the distance where attractions and repulsions balance.
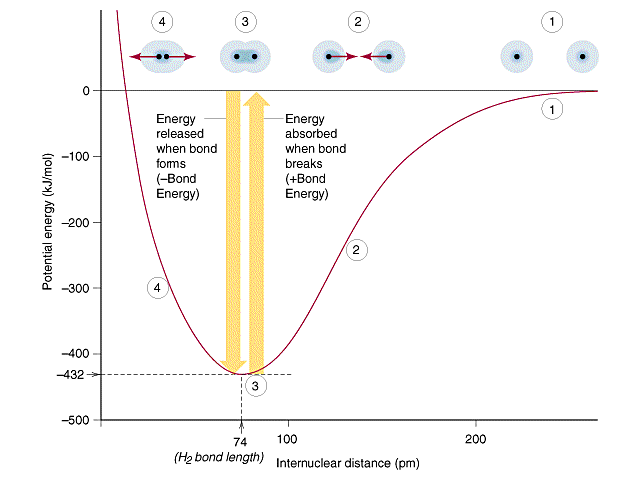
Potential energy is plotted versus internuclear distance for a covalent bond, producing a “potential well.” The minimum of the curve marks the equilibrium separation (the bond length), while the depth of the well corresponds to the bond energy required to separate the atoms. Source
Bond length: The average equilibrium internuclear distance between two bonded atoms in a molecule.
Two syllabus-aligned factors dominate comparisons of covalent bond lengths:
Core size (atomic radius): Larger atoms have valence electrons farther from the nucleus, so their covalent bonds tend to be longer.
Bond order: More shared electron density between the nuclei increases attraction and pulls nuclei closer, making bonds shorter and typically stronger.
Core size (atomic radius) trends
When comparing bonds with the same bond order, atomic size is a major driver.
Down a group, atoms have more electron shells, so atomic radius increases.
Larger valence orbitals generally overlap less effectively at short distances, so the equilibrium distance is larger.
As a result, bonds to heavier atoms (same type of bonding) are typically longer than bonds to lighter atoms.
Practical comparisons to keep consistent:
Compare single bonds to single bonds (or double to double) when isolating atomic size effects.
Compare similar bonding environments when possible, because surrounding atoms can slightly shift measured bond lengths.
Bond order and its effects on length and energy
Bond order is the number of bonding electron pairs shared between two atoms in a simple Lewis sense (single = 1, double = 2, triple = 3). Higher bond order means more electron density between the nuclei.
Bond order: A measure of the number of shared bonding electron pairs between two atoms; higher bond order indicates greater electron density between nuclei.
Higher bond order produces two key outcomes emphasized by the syllabus:
Shorter bonds: Increasing electron density between nuclei increases electrostatic attraction, pulling the nuclei closer.
Greater bond energies: More bonding interactions generally require more energy to break, so higher bond order bonds have greater bond energy than single bonds.
Relative comparisons you should be able to state
For the same pair of atoms (or very similar atoms), the typical ordering is:
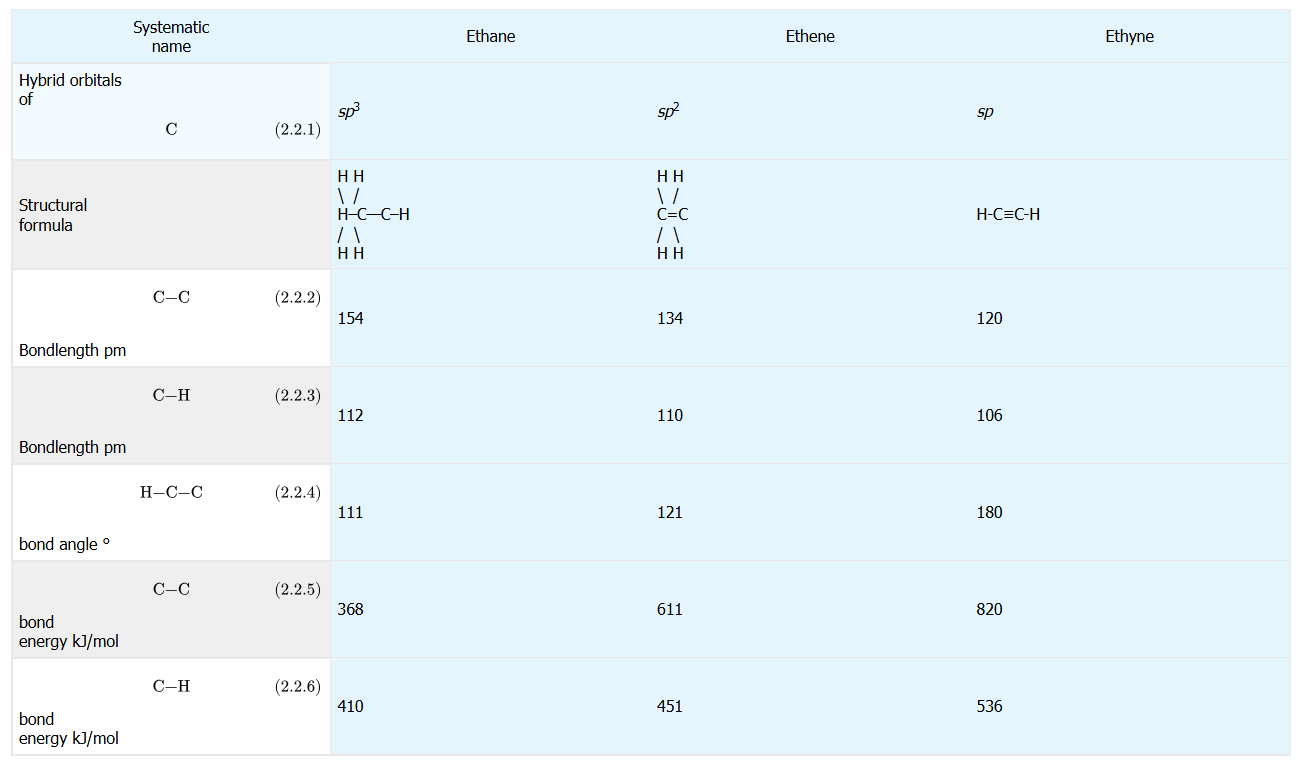
A comparison of ethane (C–C single), ethene (C=C double), and ethyne (C≡C triple) shows that higher bond order corresponds to shorter bond lengths and larger bond energies. The included structures and measured values make the “single > double > triple” bond-length trend and the opposite bond-energy trend explicit and easy to apply in reasoning problems. Source
Bond length: single > double > triple
Bond energy: single < double < triple
These are comparative trends rather than exact numerical rules; real values depend on the specific atoms involved and the molecular environment.
Non-integer and “average” bond orders (qualitative)
In some structures, the observed bond length can fall between typical single and double bond lengths because electron density is distributed in a way that makes multiple bonds equivalent. In such cases, an average bond order language can help you justify why a measured bond length is intermediate, without relying on exact calculations.
How to use these trends on AP-style reasoning tasks
When asked to compare bond lengths or bond energies, build a brief chain of reasoning:
Identify whether the comparison changes atomic size (core size) and/or bond order.
If bond order differs, that trend is usually decisive: higher bond order → shorter, stronger.
If bond order is the same, use atomic radius: larger atoms → longer bonds (and often weaker overlaps).
If both change, explicitly weigh both effects and state which one is expected to dominate based on the situation described.
FAQ
Bond lengths are obtained from structural techniques such as X-ray diffraction (solids) and microwave/rotational spectroscopy (gases). Reported values are typically averages and can depend slightly on temperature and phase.
The second bonding interaction is not identical to the first: electron–electron repulsions, orbital geometry, and diminishing returns in stabilisation mean added bonding increases energy less than proportionally.
Not reliably. If atoms differ, core size and orbital overlap change too, so bond order must be considered alongside atomic radius and how well the orbitals match in size and energy.
If electron density is shared across more than two atoms, individual bonds can become equivalent and intermediate in length. Chemists describe this using an average bond order to rationalise the measured geometry.
Yes. Packing forces in solids and molecular motion in gases can shift average distances slightly. The intrinsic trend with bond order and atomic size remains, but reported values may differ by method and context.
Practice Questions
State which bond is longer and justify: a C–C single bond or a C=C double bond.
Correct identification that the C–C single bond is longer (1)
Justification: higher bond order (double bond) has more electron density/stronger attraction pulling nuclei closer, so it is shorter (1)
A student compares the covalent bond lengths and bond energies for the following bonds: N–N, N=N, and N≡N.
(a) Rank the three bonds from longest to shortest. (2 marks)
(b) Rank the three bonds from lowest to highest bond energy. (2 marks)
(c) Explain, in terms of bond order and electron density between nuclei, why the rankings in (a) and (b) are related. (1 mark)
(a)
Correct order: N–N > N=N > N≡N (2) (allow 1 mark if two are correctly ordered)
(b)
Correct order: N–N < N=N < N≡N (2) (allow 1 mark if two are correctly ordered)
(c)
Explanation linking higher bond order to greater electron density between nuclei, giving shorter bond length and greater energy to break (1)

