AP Syllabus focus: ‘Use Coulomb’s law to compare cation–anion attractions: larger ionic charges strengthen interactions, and smaller ionic radii (shorter distances) increase interaction strength.’
Ionic compounds are held together by electrostatic attractions between oppositely charged ions. Coulomb’s law provides a simple, powerful way to compare how strongly different cation–anion pairs attract based on charge and distance.
Core idea: electrostatic attraction controls interaction strength
Ionic interaction strength refers to how strongly a cation and anion attract each other due to opposite charges. In AP Chemistry, you mainly use Coulomb’s law qualitatively to compare relative strengths, not to compute exact forces.
Key quantities you compare
Ionic charge: e.g., , , ,
Interionic distance: approximated by the sum of ionic radii (smaller ions sit closer)
Larger charge magnitude and shorter distance both produce a stronger attraction.
Coulomb’s law applied to ions
In the point-charge model, ions are treated as charged particles whose attraction depends on charge and separation.
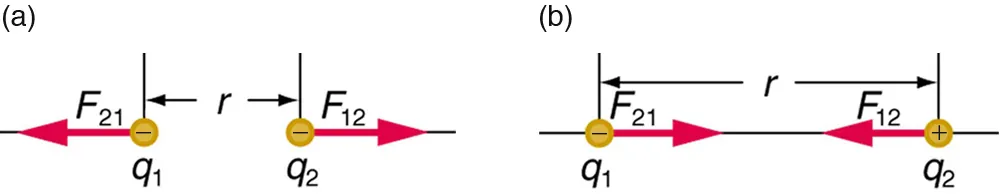
This diagram illustrates Coulombic interactions between two point charges separated by a distance , showing both repulsion for like charges and attraction for unlike charges. The equal-and-opposite force vectors emphasize that the interaction strength depends on charge and separation, while the direction depends on the signs of the charges. Source
Ionic radius: A measure of an ion’s effective size in a crystal; smaller ionic radius generally means a shorter cation–anion distance when paired with a given counterion.
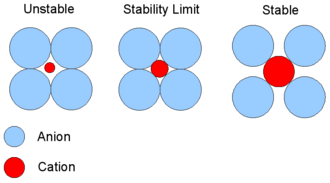
This radius-ratio diagram models ions as hard spheres and shows how the cation-to-anion size ratio controls whether a given coordination environment is geometrically stable. It helps you connect “smaller ionic radii” to a shorter center-to-center separation , which (via Coulomb’s law) increases the magnitude of electrostatic attraction. Source
Ionic radius is not a directly “visible” boundary; it is a model-based size that helps you reason about distances in ionic solids.
= magnitude of electrostatic force (N)
= charge on ion 1 (C)
= charge on ion 2 (C)
= distance between ion centres (m)
= Coulomb’s constant (N·m·C)
For AP comparisons, focus on proportionality: .
How charge affects attraction (qualitative comparisons)
Increasing the magnitude of ionic charge increases attraction strongly because the product increases.
If distance is similar, a / pair attracts more strongly than a / pair.
Comparing products:
So, with comparable distances, attraction strength tends to follow these charge products.
What to say in words (expected AP phrasing)
“Larger ionic charges strengthen interactions because the electrostatic attraction increases with .”
How ionic size (distance) affects attraction
Distance matters even more sharply because it appears as in the denominator.
Smaller ionic radii → smaller → much stronger attraction.
For ions with the same charges, the pair involving smaller ions typically has stronger attraction because the ions can approach more closely.
What counts as “smaller ions” in typical AP comparisons
Higher-charge cations (e.g., vs ) are often smaller due to stronger attraction to electrons.
Within a group, ions generally get larger down the group, increasing and weakening attractions (when charges are the same).
What to say in words (expected AP phrasing)
“Smaller ionic radii (shorter distances) increase interaction strength because increases as decreases.”
Using Coulomb’s law to rank interaction strength
When asked to compare two ionic interactions, use a consistent checklist:
Compare charge product first (bigger product → stronger).
If charge products are equal or similar, compare distance using ionic radii (smaller → stronger).
If one pair has larger charges but also much larger ions, explicitly state both factors and decide which dominates (charge changes often have a large effect, but very large distance can weaken attraction).
Limits of the model (what not to overclaim)
Coulomb’s law assumes point charges and ignores electron-cloud shape and short-range repulsions. It is best used for relative comparisons of ionic attraction, not exact numerical prediction in real crystals or solutions.
FAQ
The inverse-square form comes from how electric field strength spreads in three dimensions.
Small decreases in $r$ can cause large increases in attraction, so ionic radius differences can strongly affect interaction strength.
In materials, an effective constant $k_\text{eff} = k/\varepsilon_r$ is used.
Larger $\varepsilon_r$ reduces electrostatic attraction by screening charges, so attractions are weaker than in vacuum.
No. “Ionic radius” depends on coordination number and the measurement/model used.
Different crystal environments assign slightly different radii to the same ion because electron density is distributed, not sharply bounded.
At short range, electron-cloud overlap causes strong repulsion (Pauli/exchange effects).
So the net interaction is not purely Coulombic; there is an optimal separation where attraction and repulsion balance.
You approximate the interaction using an effective charge and an effective distance to where the negative charge is concentrated.
Because charge is delocalised, the “distance” is less precise, so conclusions should be stated as qualitative comparisons.
Practice Questions
(2 marks) Using Coulomb’s law, state which attraction is stronger and give one reason: or .
Identifies as stronger (1)
Correct reason: larger charge product / larger ionic charge increases attraction (1)
(5 marks) Rank the electrostatic attraction strength (strongest to weakest) for: , , . Justify using Coulomb’s law, referring to both charge and ionic size (distance).
States strongest (1)
Justifies with larger (e.g., 4 vs 1) (1)
Correctly compares vs (1)
States smaller than so smaller (1)
Links smaller to stronger attraction via (1)

