AP Syllabus focus: ‘In ionic crystals, cations and anions form a systematic, repeating 3‑D array that maximizes attractions and minimizes repulsions; use this particulate model to explain properties of ionic solids.’
Ionic solids are best understood by linking microscopic structure to macroscopic observations. A repeating lattice of oppositely charged ions explains why these solids are crystalline, strong, brittle, and behave distinctively when heated or dissolved.
The ionic crystal lattice model
Ionic crystal lattice: a systematic, repeating 3‑D array of cations and anions arranged so that attractions are maximized and repulsions are minimized.
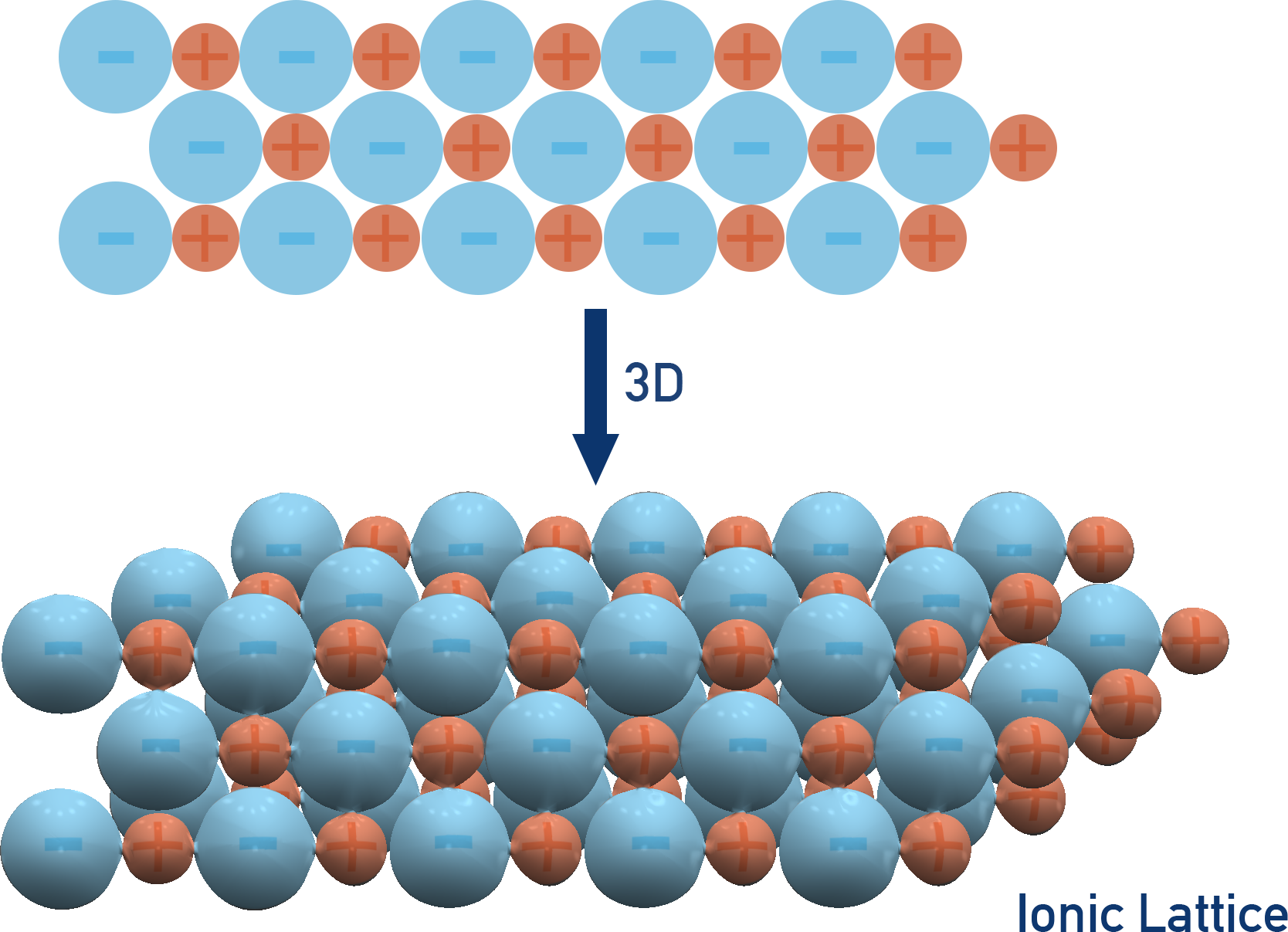
A particulate model of an ionic crystal lattice showing alternating cations and anions arranged in a repeating three-dimensional structure. The visual emphasizes that stability comes from placing opposite charges as nearest neighbors throughout the lattice while keeping like charges separated as much as possible. Source
This particulate model emphasises two competing electrostatic ideas:
Attractions between unlike charges (cation–anion) stabilise the solid.
Repulsions between like charges (cation–cation, anion–anion) destabilise the solid and must be reduced by ion arrangement.
How the lattice “chooses” an arrangement
In a stable ionic solid, ions pack so that each ion is surrounded by as many oppositely charged neighbours as possible without forcing like charges too close.
The crystal must remain overall electrically neutral.
The lattice pattern repeats in all directions, producing a crystalline (ordered) solid rather than an amorphous one.
Formula unit: the lowest whole-number ratio of ions that represents an electrically neutral ionic compound.
The formula unit is reflected in the repeating lattice: the structure extends beyond any single “molecule,” so ionic compounds are not made of discrete molecular units in the solid state.
Explaining macroscopic properties using the lattice
High melting points and boiling points
Ionic solids typically have high melting points because:
Many strong cation–anion attractions must be overcome throughout the 3‑D lattice to allow ions to move past one another.
Melting is not breaking one bond; it is disrupting a large network of electrostatic interactions across the crystal.
This is why ionic solids are often solids at room temperature and require substantial heating to melt.
Brittleness (cleavage under stress)
Ionic solids are usually brittle rather than malleable. When an external force shifts layers in the lattice:
Like-charged ions can become adjacent (anion next to anion or cation next to cation).
The resulting strong electrostatic repulsions can cause the crystal to cleave along planes, producing fractures.
Thus, the same ordered structure that stabilises the solid also creates a mechanical weakness when the alignment is disturbed.
Electrical conductivity: solid vs molten vs aqueous
Electrical conductivity requires mobile charged particles.
Solid ionic crystals do not conduct: ions are locked into fixed positions within the lattice and cannot move to carry charge.
Molten ionic compounds conduct: heating disrupts the lattice enough for ions to move freely.

A three-panel laboratory demonstration of conductivity in sodium chloride: the bulb is off for solid NaCl, and on when the salt is molten. This illustrates that electrical conduction requires mobile ions, which are only available once the rigid lattice is disrupted by melting. Source
Aqueous ionic solutions conduct (when soluble): water separates and disperses ions, making them mobile charge carriers.
The key structural link is whether the lattice is intact (immobile ions) or broken apart (mobile ions).
Crystal shape and hardness
Because the lattice is repeating and ordered, ionic solids:
form crystalline shapes with flat faces and consistent angles,
are often hard due to the extensive 3‑D network of attractions resisting deformation.
Observed differences in hardness and melting point among ionic compounds can be interpreted by how strongly the lattice holds ions in place and how effectively it avoids like-charge repulsions.
FAQ
No. Different compounds adopt different repeating arrangements depending on ion sizes and charge balance, which changes neighbour counts and packing efficiency.
It is the number of nearest oppositely charged ions surrounding a given ion in the crystal. It varies with relative ion sizes.
Solubility depends on the competition between breaking the lattice and stabilising separated ions in water; stronger stabilisation of free ions favours dissolving.
Missing ions or misplaced ions can alter density and sometimes change conductivity by creating pathways or charge imbalances that increase ion mobility.
Cleavage occurs along planes where shifting aligns like charges, maximising repulsion; the crystal breaks cleanly along these repeating structural planes.
Practice Questions
State why an ionic solid does not conduct electricity, but the same substance conducts when molten.
Solid: ions are fixed in a lattice / not mobile (1)
Molten: ions are free to move and carry charge (1)
Using the ionic lattice model, explain (i) why ionic solids have high melting points and (ii) why they are brittle when struck.
Lattice is a 3-D array of cations and anions (1)
High melting point: must overcome many strong cation–anion attractions throughout the lattice (1)
Melting involves disrupting an extended network, not discrete molecules (1)
Brittleness: stress shifts ion layers so like charges become adjacent (1)
Repulsion between like-charged ions causes cleavage/fracture (1)

