AP Syllabus focus: ‘Model metallic bonding as an array of positive metal ions surrounded by delocalized valence electrons; connect this structure to properties such as conductivity and malleability.’
Metallic solids behave differently from ionic and molecular solids because their valence electrons are mobile. The “sea of electrons” model links particulate-level structure to key observable properties tested in AP Chemistry.
Metallic bonding as a particulate model
In a metallic solid, metal atoms pack into a lattice and their valence electrons become shared by the entire solid rather than belonging to one atom. The result is an array of positive metal ions in a background of delocalized valence electrons.

Particulate model of a metallic solid showing a lattice of positive metal ion cores embedded in a mobile “sea” of delocalized electrons. This visualization supports the key AP idea that metallic bonding is an electrostatic attraction between the ion lattice and the electron sea, rather than discrete, directional bonds. Source
Metallic bonding: electrostatic attraction between a lattice of positive metal ions and a mobile “sea” of delocalized valence electrons that holds the solid together.
What “delocalized” implies
Delocalized electrons are not confined to a single bond or a single nucleus; they can move throughout the metal crystal.
The metal is best described as ions + electrons, not as discrete molecules.
Because electrons are mobile, metals can respond to electric fields and mechanical stress without the bonding “breaking” in a localized way.
What the metal ions represent
The “ions” in this model are metal nuclei plus core (non-valence) electrons. They are effectively fixed in place relative to the faster-moving valence electrons, although they vibrate more as temperature increases.
Connecting structure to metallic properties
The AP expectation is that you can use the ions-in-a-sea-of-electrons model to explain major properties, especially conductivity and malleability.
Electrical conductivity
Metals conduct electricity because they contain mobile charge carriers (the delocalized electrons).
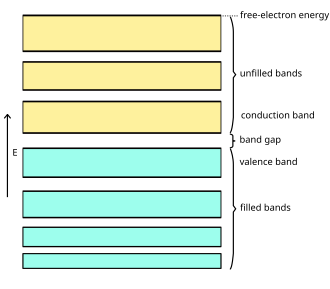
Electronic band diagram illustrating valence and conduction bands separated by an energy gap (typical of semiconductors/insulators). In metals, bands overlap or the gap is effectively absent, which aligns with the idea that electrons can move easily and act as mobile charge carriers during electrical conduction. Source
When a potential difference is applied, electrons drift through the lattice.
Since electrons are already present and mobile, there is no need to break bonds or move whole ions to carry charge.
Conductivity typically decreases as temperature rises because increased lattice vibrations scatter electrons more, reducing their mobility (conceptual linkage only).
Thermal conductivity
Metals are good thermal conductors because energy can be transferred efficiently by:
collisions among mobile electrons (rapid energy transport)
vibrations passed through the closely packed lattice
Malleability and ductility
Malleability (hammered into sheets) and ductility (drawn into wires) follow from the non-directional nature of metallic bonding.
In a metal, layers of ions can slide past one another under stress.
The sea of electrons continues to attract the ions after they shift position, so the solid deforms rather than shattering.
This contrasts with structures where shifting brings like charges next to each other and causes strong repulsion and fracture.
Luster (metallic shine)
Metallic luster can be described using the same electron mobility idea:
Delocalized electrons at the surface can absorb and re-emit a wide range of light frequencies, producing a shiny appearance.
Variations in strength and melting point (within the model)
Not all metals have the same melting point or hardness; the model explains trends qualitatively.
More delocalized valence electrons per atom and/or higher ionic charge can increase the attraction between ions and the electron sea.
Smaller ions (higher charge density) can also strengthen attractions because electrons are held more strongly near the ion lattice. These stronger attractions generally correlate with higher melting points and greater hardness (qualitative comparison, not calculation-based).
Using correct AP Chemistry language
When explaining metallic bonding, keep the cause-and-effect chain clear:
Structure: positive ions in a lattice + delocalized electrons
Mechanism: electrons move; bonding is non-directional and maintained during ion-layer shifts
Properties: conductivity, malleability/ductility, thermal conductivity, luster
FAQ
Softer metals typically have fewer delocalised electrons per atom and form lower-charge metal ions, reducing ion–electron attraction.
Larger atomic size can also lower charge density, weakening the lattice’s overall cohesion.
Band language reframes the same key point: electrons occupy closely spaced energy levels that extend through the solid.
A partially filled band (or small gap) allows electrons to move into nearby available states when an electric field is applied.
Heating increases ion vibration amplitude. The average ion–ion separation increases because vibrations sample more of the less-attractive region of the potential energy curve.
Stronger bonding reduces, but does not eliminate, thermal expansion.
Without an electric field, their motion is random overall.
With a potential difference applied, a small net drift develops opposite the electric field direction, superimposed on random motion.
The lattice ions remain in fixed positions (aside from vibration) because they are much more massive and are bound in the crystal lattice.
Charge transport occurs through electron drift, not by bulk ion migration through the solid.
Practice Questions
(2 marks) Using the “sea of electrons” model, explain why solid copper conducts electricity.
Mentions delocalised/mobile valence electrons in the metal lattice (1)
Explains that electrons move/drift to carry charge when a potential difference is applied (1)
(6 marks) Magnesium metal is harder and has a higher melting point than sodium metal. Use the metallic bonding model to explain these differences. Then explain why both metals are malleable.
Identifies metallic structure as positive metal ions in a lattice surrounded by delocalised electrons (1)
States Mg provides more delocalised electrons per atom than Na (e.g., 2 vs 1) (1)
Links more delocalised electrons to stronger electrostatic attraction/stronger metallic bonding (1)
States Mg forms higher-charge ions than Na (Mg vs Na) (1)
Links higher ionic charge (and/or stronger attraction) to higher melting point/hardness (1)
Explains malleability: layers of ions can slide while attraction to delocalised electrons remains (non-directional bonding) (1)

