AP Syllabus focus: ‘Interstitial alloys form when much smaller atoms occupy the spaces between larger metal atoms (e.g., carbon in iron); relate this structure to changes in strength and hardness.’
Interstitial alloys are a major way engineers tune metallic properties. By inserting small atoms into the gaps of a metal lattice, the internal structure is disrupted in predictable ways that change hardness and strength.
What interstitial alloys are
Core idea: small atoms in the “gaps”
In a metallic solid, metal atoms pack into a regular lattice with small open regions between them.
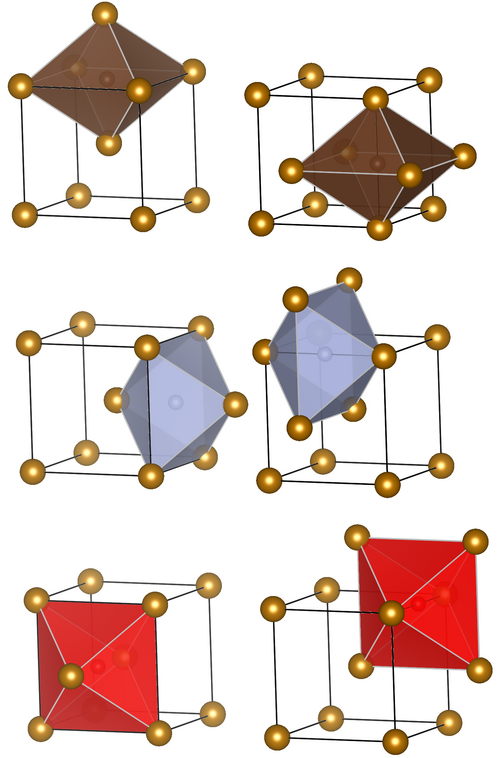
Representative octahedral interstitial sites in a body-centered cubic (BCC) metal lattice are highlighted, with an interstitial atom shown occupying the void. This visual makes the key geometric point of interstitial alloys: the solute sits between host metal atoms rather than replacing them, which sets up lattice strain and altered mechanical properties. Source
If a second element’s atoms are much smaller, they can fit into these spaces rather than replacing metal atoms.
Interstitial alloy: an alloy in which small atoms occupy spaces (interstices) between larger metal atoms in the crystal lattice.
These small atoms do not typically form a separate lattice of their own; instead, they become embedded throughout the metal structure, creating a modified metallic solid.
Interstitial sites and size mismatch
Interstitial atoms are constrained by the surrounding metal atoms. Because the “fit” is imperfect, the metal lattice becomes locally strained.
Interstitial site: an empty space in a crystal lattice (between the regular lattice positions) that can be occupied by a smaller atom or ion.
A key consequence is that the lattice is no longer an ideal repeating array; it is slightly distorted around each interstitial atom.
Structure–property links: why strength and hardness increase
Lattice distortion blocks deformation
Most metals are malleable because planes of atoms can slide past each other under stress. Interstitial atoms make this sliding harder because they:
Distort the lattice (introducing strain)
Create regions where metal atoms are pushed apart or pulled closer
Pin or hinder the movement of defects that enable plastic deformation
This makes the metal resist shape change more strongly, so both strength (resistance to permanent deformation) and hardness (resistance to indentation/scratching) increase.
Dislocations and “solid-solution strengthening” (conceptually)
While AP Chemistry does not require detailed metallurgy, it is useful to know that real metal crystals contain defects. Interstitial atoms interfere with the movement of these defects, so more force is required to deform the metal.
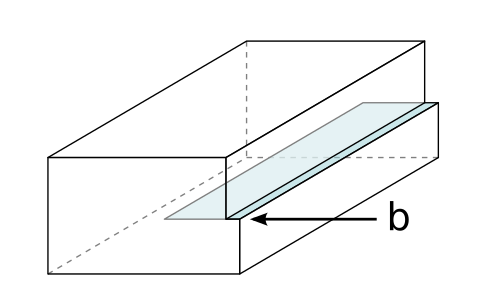
Schematic of an edge dislocation, a line defect that enables plastic deformation when it moves through a crystal under stress. Interstitial solute atoms create local strain fields that interact with dislocations, making dislocation motion more difficult and therefore increasing strength and hardness. Source
Example emphasised by the syllabus: carbon in iron
Carbon in iron (steel) is the classic interstitial alloy example:
Iron atoms form the primary lattice.
Much smaller carbon atoms occupy interstitial sites.
The resulting lattice strain makes it harder for iron layers to shift.
Observable property trends commonly linked to increasing interstitial content include:
Hardness increases
Tensile strength increases
Ductility decreases (less ability to draw into wires)
Malleability decreases (less ability to hammer into sheets)
Additional property changes you may notice (secondary effects)
Even though the syllabus highlights strength and hardness, interstitial alloying can also influence other macroscopic properties:
Electrical/thermal conductivity often decreases somewhat because lattice distortions scatter mobile electrons more.
Density may change slightly depending on the mass of the interstitial atom and how much is added.
Melting point can change, but the direction and magnitude depend on the specific metal–interstitial combination and how it alters bonding and lattice stability.
How to identify an interstitial alloy (AP-style cues)
Look for these clues in wording or diagrams:
A metal lattice with smaller atoms drawn in the spaces between metal atoms
A large atomic size difference between components
Property descriptions such as “harder/stronger but less malleable”
Explicit examples like C in Fe, or small atoms (e.g., B, C, N, H) in a transition metal matrix
FAQ
It must be sufficiently smaller than the host metal atom to fit into interstitial sites without replacing metal atoms.
A practical indicator is a large atomic radius mismatch; otherwise, substitution is more likely.
Carbon is very small and can occupy interstitial sites in iron at useful concentrations.
Its presence produces significant lattice strain, strongly impeding deformation.
Often they do, because lattice distortions increase electron scattering.
However, the extent depends on the concentration and identity of the interstitial element and the host metal.
Yes. Interstitial sites are limited, and excessive interstitial content can make the structure unstable.
Beyond a solubility limit, new phases/compounds may form instead of a single interstitial solid.
Even low concentrations can create widespread lattice strain because each interstitial atom disrupts many neighbouring metal atoms.
This efficiently blocks deformation pathways, so strength/hardness can rise sharply with small additions.
Practice Questions
(1–3 marks) Carbon is added to iron to make steel. Explain, in terms of structure, why steel is typically harder than pure iron.
States that carbon atoms are much smaller and fit into spaces between iron atoms / occupy interstitial sites. (1)
Explains that this causes lattice distortion/strain in the iron lattice. (1)
Links distortion to reduced ability of layers/planes of atoms to slide (so hardness increases). (1)
(4–6 marks) A student compares pure copper with an alloy made by adding a small amount of a much smaller element X to copper. The alloy is found to be stronger and less malleable than pure copper. (a) State the type of alloy formed and describe where X atoms are located in the structure. (b) Explain how the location of X atoms leads to increased strength and hardness.
Identifies the alloy as an interstitial alloy. (1)
Describes X atoms occupying spaces/interstices between copper atoms in the lattice. (1) (b)
Explains that X atoms distort/strain the metal lattice. (1)
Explains that distortion hinders movement of layers/planes (or dislocations) so deformation is more difficult. (1)
Links to increased force needed for deformation → higher strength and/or hardness. (1)
Links hindering movement to reduced malleability/ductility. (1)

