AP Syllabus focus: ‘Substitutional alloys form when atoms of similar size replace each other in the metal lattice (e.g., zinc substituting for copper in brass); explain how substitution affects properties.’
Substitutional alloys are a key materials application of metallic bonding. Understanding how one metal atom can replace another in a crystal lattice helps you predict changes in strength, hardness, conductivity, and overall performance.
What a substitutional alloy is
A substitutional alloy forms when atoms of a second element with similar atomic size replace (substitute for) host metal atoms at normal lattice positions while the overall metallic bonding framework remains.
Substitutional alloy: A metallic solid solution in which solute atoms occupy regular lattice sites of the solvent metal by replacing some of its atoms.
Substitution is most successful when the two atom types fit into the same repeating arrangement with minimal strain, so “similar size” is a practical structural requirement, not just a memorised phrase.
The metal lattice context
Metals consist of a regular, repeating array of atoms (often described as positive ion cores) immersed in delocalised valence electrons. Substitution changes the identity of some lattice sites but keeps the overall “sea of electrons” model intact.
Crystal lattice: A repeating three-dimensional arrangement of particles (here, metal atoms/ion cores) that defines the solid’s structure.
Because the bonding is collective and non-directional, metals can tolerate some substitution without losing the metallic character, but the lattice becomes less uniform.
How lattice substitution happens (particulate view)
In a pure metal, each lattice site is occupied by the same type of atom. In a substitutional alloy:
Solvent metal atoms form the main lattice framework.
Solute metal atoms of similar radius occupy some of the same lattice positions.
The delocalised electrons move throughout the entire solid, now interacting with a mixture of ion cores.
A classic example is brass, where Zn substitutes for Cu in the copper lattice. On a particulate diagram, you would show Zn atoms scattered among Cu atoms at normal lattice points (not in the holes between them).
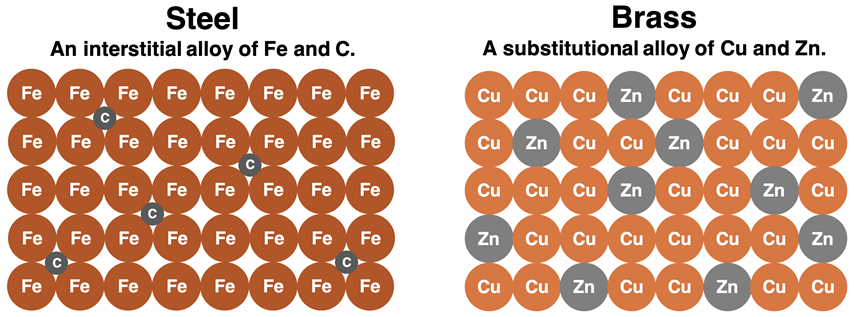
Particle diagram comparing an interstitial alloy (steel, Fe–C) with a substitutional alloy (brass, Cu–Zn). In the brass panel, Zn atoms replace some Cu atoms at regular lattice positions, reinforcing that substitutional alloys place the solute on lattice sites rather than in interstitial gaps. Source
Why “similar size” matters
If the substituting atom is much larger or smaller than the host atom:
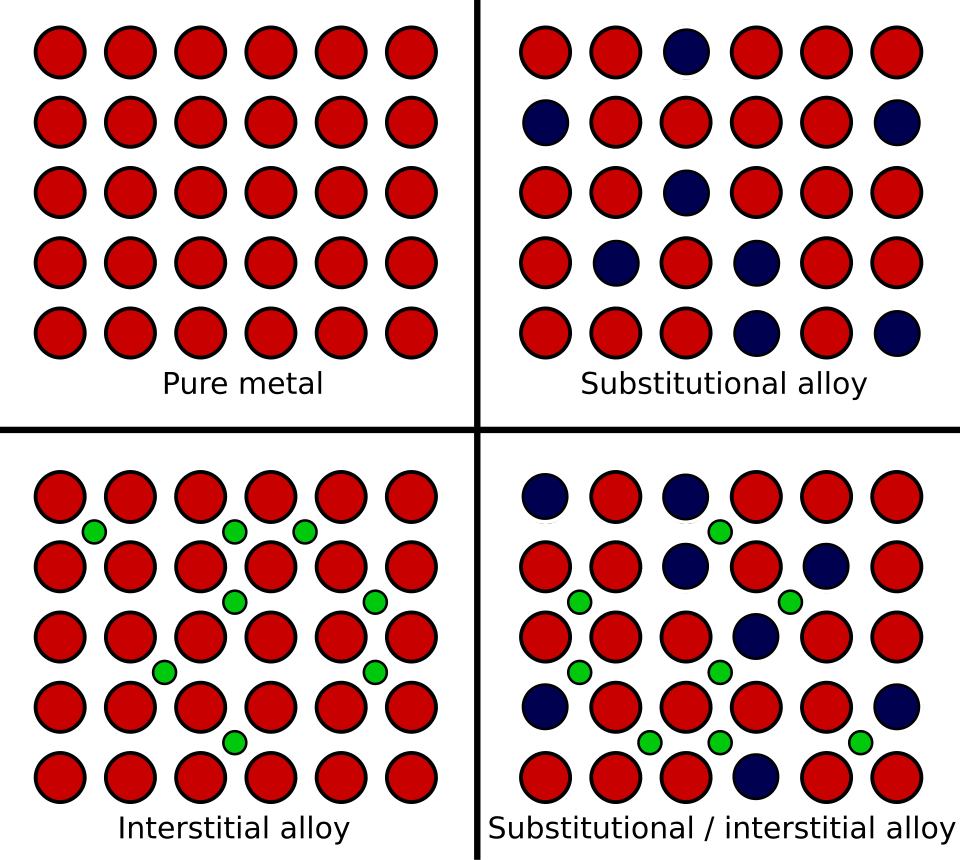
Schematic of atomic arrangements for a pure metal, a substitutional alloy, an interstitial alloy, and a mixed case. The substitutional panel shows similarly sized atoms occupying the same lattice sites, which connects directly to the “similar size” requirement and the idea that the overall lattice framework is retained (but becomes less uniform). Source
the lattice would distort too strongly,
packing becomes inefficient,
the solid may separate into multiple phases rather than forming a uniform substitutional alloy.
On the AP level, you should connect “similar size” to the ability to maintain a stable lattice with manageable distortions.
How substitution affects properties
Replacing some host atoms changes how easily metal layers slide and how freely electrons move. The key idea is that a less regular lattice creates obstacles to motion at the atomic scale.
Strength and hardness (typically increase)
In many substitutional alloys:
Strength increases because differently sized atoms create lattice strain.
This strain hinders the movement of atomic layers past each other, making the metal harder to deform.
As a result, the alloy is often harder and less malleable/ductile than the pure metal.
This is why alloying is a common strategy for improving mechanical performance compared with a pure metal.
Electrical and thermal conductivity (often decrease)
Pure metals conduct well because delocalised electrons move with relatively few disruptions. Substitution tends to:
increase electron scattering due to the less uniform arrangement of ion cores,
reduce the ease of electron flow,
therefore lower electrical conductivity (and often thermal conductivity, since electron motion contributes strongly to heat transfer in metals).
Other property shifts you may be asked to explain
Depending on the specific metals involved, substitution can also change:
melting point (often altered because the lattice is no longer perfectly uniform)
density (because different atoms have different masses while occupying similar volumes)
colour/appearance (notably in alloys like brass)
For AP responses, focus on clear cause-and-effect: substitution → lattice irregularity/strain → changed layer motion and electron flow → property changes.
How to communicate this on the AP Exam
When prompted to “explain how substitution affects properties,” use these linked statements:
Similar-sized atoms substitute into normal lattice positions.
The lattice becomes distorted/less regular.
Distortion makes slipping harder → increased strength/hardness.
Irregularity increases electron scattering → decreased conductivity.
Avoid describing solute atoms as occupying gaps; that language belongs to a different alloy type. Keep the explanation anchored to substitution at lattice sites and the resulting structural disruption.
FAQ
They use empirical “size and compatibility” ideas (often called Hume–Rothery-type rules). Beyond similar atomic radii, favourable mixing is more likely when the metals have similar crystal structures and comparable tendencies to attract electrons.
Yes. At certain compositions/temperatures, atoms can adopt partially ordered patterns (superlattice ordering). Ordering can increase hardness and change electrical resistance compared with a fully random distribution.
Substitution can alter surface reactivity and the stability of protective oxide layers. In some alloys, a minor component promotes formation of a thin, adherent oxide that slows further oxidation.
Heating can increase atomic diffusion, allowing more uniform mixing or enabling ordering; quenching can “freeze in” a high-temperature arrangement. These changes can shift hardness and conductivity without changing the overall alloy type.
Not always, but it is common. The outcome depends on how strongly the substituted atoms scatter electrons; small amounts may have modest effects, while higher substitution levels typically raise resistivity noticeably.
Practice Questions
(2 marks) Brass is an alloy of copper and zinc. State what is meant by a substitutional alloy and explain why zinc can form a substitutional alloy with copper.
Defines substitutional alloy: solute atoms replace host atoms at regular lattice sites in a metal lattice. (1)
Zinc can substitute because Zn and Cu atoms are of similar size/radius so they can occupy the same lattice positions without excessive distortion. (1)
(5 marks) A student compares pure copper with brass (Cu–Zn). Using a particulate-level explanation, describe how substituting zinc atoms into the copper lattice affects (i) strength/hardness and (ii) electrical conductivity.
Describes substitution: Zn atoms replace some Cu atoms at normal lattice points (not in interstitial spaces). (1)
States lattice becomes less regular/distorted due to size mismatch/strain. (1)
Links distortion to hindered layer/plane slipping (or dislocation motion) leading to increased strength/hardness. (2)
Links irregular lattice to increased electron scattering/reduced electron mobility leading to decreased electrical conductivity. (1)

