AP Syllabus focus: ‘Particles in matter move continuously and randomly; average kinetic energy relates to average speed by KE = ½mv², connecting microscopic motion to measurable properties.’
Molecular motion is the bridge between particle-level models and macroscopic measurements. In gases especially, particle speeds vary widely, but kinetic energy provides a quantitative way to describe and compare that motion.
Continuous, Random Particle Motion
Particles in all states of matter are in continuous motion, but in gases and liquids that motion is especially apparent because particles can translate (move from place to place). The motion is random, meaning there is no preferred direction in an unforced sample.
What “speed” means in particle models
Speed is the magnitude of a particle’s velocity (a scalar, not a direction). In a sample:
individual particles constantly change speed and direction due to collisions
macroscopic properties arise from averages over enormous numbers of particles
Kinetic Energy Links Microscopic Motion to Measurable Properties
The key quantitative descriptor of motion is kinetic energy, which depends on both particle mass and particle speed.
Kinetic energy (KE): energy associated with the motion of a particle; for translational motion it depends on mass and speed.
Kinetic energy is useful because it can be related to observable behaviour (for example, faster-moving particles collide more often and more forcefully with container walls).
The kinetic energy–speed relationship
For a single particle moving translationally, kinetic energy is related to speed by the classical expression below.
= translational kinetic energy of one particle (J)
= mass of one particle (kg)
= speed of the particle (m s)
Because the speed term is squared, changes in speed have a strong effect on kinetic energy (doubling increases by a factor of four).
Average Kinetic Energy and Average Speed
A real sample contains a huge number of particles with a range of speeds.
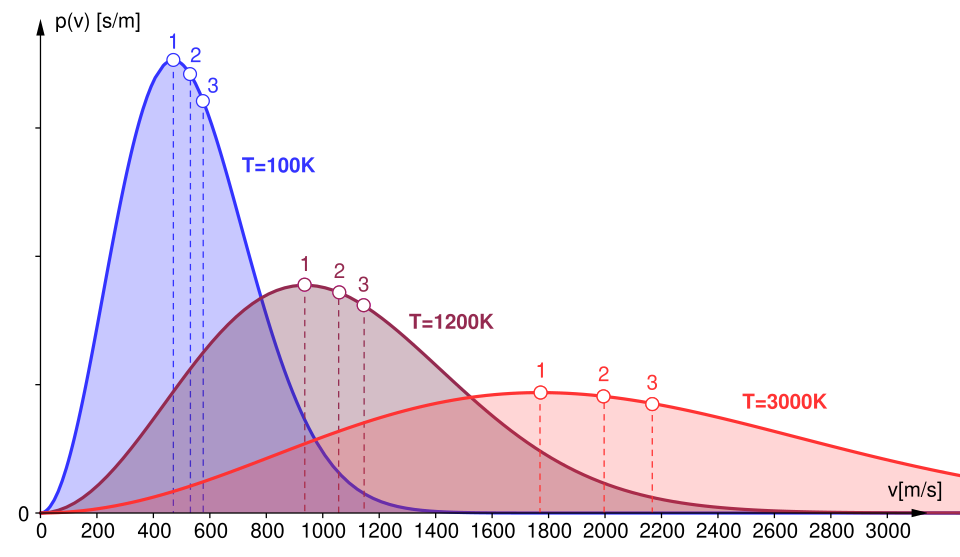
Maxwell–Boltzmann speed distributions for the same gas at different temperatures, showing how the peak shifts to higher speeds and the curve broadens as increases. The labeled markers emphasize that “typical speeds” (e.g., most probable and average-type speeds) are not the same value and move with temperature. This supports the idea that macroscopic averages come from an underlying spread of molecular speeds. Source
AP Chemistry commonly discusses average kinetic energy and how it connects to an average speed idea, even though no single particle permanently “has” the average value.
Why “average” matters
Collisions and intermolecular interactions constantly redistribute energy, so at any instant:
some particles are moving slowly
some are moving rapidly
most are in between
The average kinetic energy is a statistical measure that helps connect microscopic motion to properties you can measure in the lab.
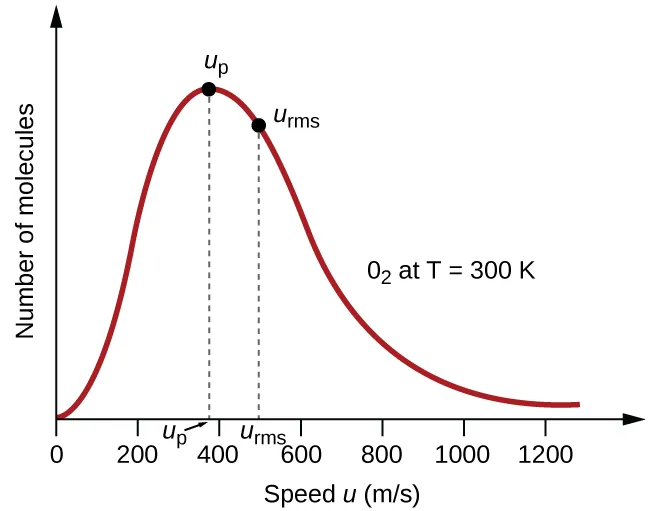
A Maxwell–Boltzmann molecular speed distribution for at 300 K, with the most probable speed and the root-mean-square speed marked on the curve. The diagram highlights that the distribution is skewed, so different “typical speed” definitions fall at different positions. This visual supports interpreting average-based macroscopic properties as arising from a distribution rather than a single representative particle speed. Source
Mass matters: same KE does not mean same speed
From , if two different particles have the same kinetic energy, the lighter particle must have the higher speed. Qualitatively:
at a fixed kinetic energy, increasing mass decreases speed
at a fixed mass, increasing speed increases kinetic energy sharply (because of )
This mass–speed connection is essential when comparing the motion of different gases or different particles within a mixture: differences in particle mass change typical speeds even when energy considerations are comparable.
Connecting Microscopic Motion to Measurable Behaviour
Molecular-level speed and kinetic energy are not measured particle-by-particle in typical introductory experiments, but they underpin macroscopic observations. When particles move faster on average, their collisions with surroundings change in ways that can be detected as changes in bulk properties.
Key particle-level links to observations (qualitative)
Faster particles transfer more energy and momentum during collisions, altering measured system behaviour.
Because particles are always moving and colliding, macroscopic properties represent collective outcomes, not the motion of any single “representative” particle.
Using and speed together provides a consistent language for explaining how microscopic motion scales up to the lab scale.
FAQ
No. “Average” is a mean over all particles, while “most probable” is the peak of the speed distribution.
They can be close, but they are not identical for real molecular speed distributions.
Frequent collisions continually exchange energy between particles.
Even without external changes, collisions randomise directions and redistribute speeds across the sample.
Not in this simple form. The expression is specifically for translational kinetic energy of a particle treated classically.
Rotational and vibrational energies are described with different models and quantisation ideas.
Because the equation describes the kinetic energy of an individual particle.
To use molar mass, you must first convert to mass per particle (divide by Avogadro’s constant), keeping units consistent.
At extremely high speeds approaching relativistic regimes, or when motion must be treated quantum mechanically, the classical relationship can break down.
For typical AP Chemistry conditions, it is an excellent approximation.
Practice Questions
(2 marks) State the equation that relates the kinetic energy of a particle to its mass and speed, and give the meaning of each symbol.
(1)
Correct identification of symbols (any two): kinetic energy, mass of one particle, speed (1)
(5 marks) Two gas particles, A and B, have the same kinetic energy. Particle A has twice the mass of particle B. Determine the ratio of their speeds and explain your reasoning using .
Uses and states KE is equal for both (1)
Sets (1)
Substitutes (1)
Deduces or equivalent (1)
Correct ratio (or ) with brief explanation that heavier particle must move more slowly for same KE (1)

