AP Syllabus focus: ‘Maxwell–Boltzmann distributions graph the range of particle energies or speeds at a temperature, helping compare how distributions change as temperature changes.’
Maxwell–Boltzmann distributions are a central tool for connecting temperature to molecular motion. Interpreting their shapes and shifts lets you reason about how a sample’s particle speeds (or kinetic energies) are spread out, not uniform.
What a Maxwell–Boltzmann Distribution Shows
A Maxwell–Boltzmann distribution is a curve that displays how many particles in a gas have a particular speed (or kinetic energy) at a given temperature. The key idea is that particles occupy a range of speeds/energies due to constant, random collisions.
Axes and Meaning
Most AP Chemistry graphs use:
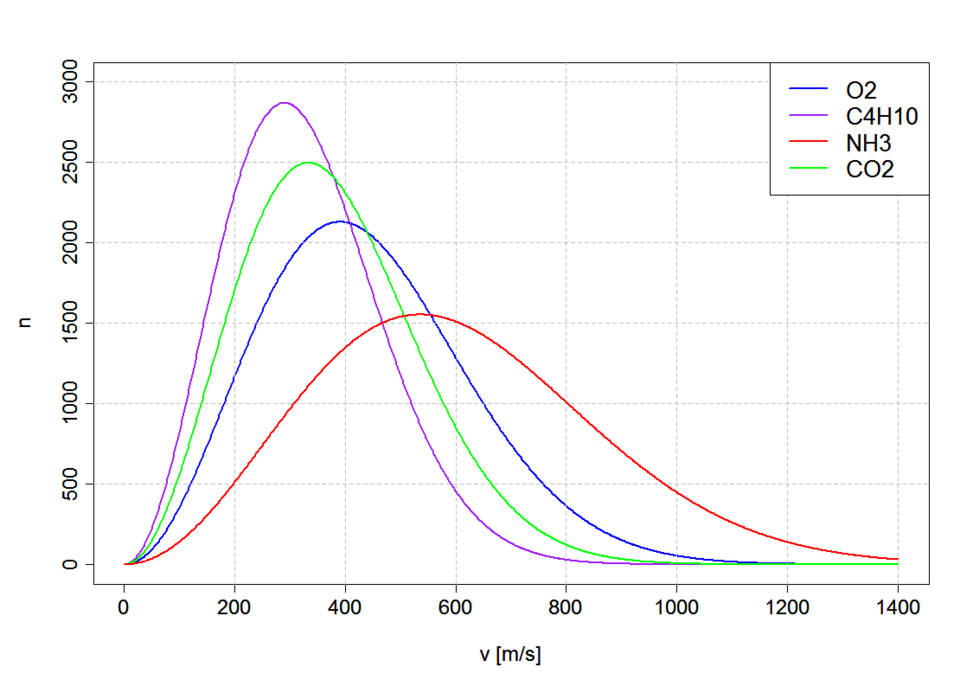
Maxwell–Boltzmann molecular speed distributions for several different gases at the same temperature (293.15 K), with speed on the x-axis and number of particles per million on the y-axis. The differing peak positions visually reinforce that heavier molecules tend to have lower most probable speeds, while lighter molecules peak at higher speeds. Source
x-axis: molecular speed (sometimes kinetic energy)
y-axis: number (or fraction) of molecules with that speed (or energy)
Interpret the y-axis as a relative count: higher y-values mean more particles fall in that small speed interval.
Area Under the Curve
The total area under the curve represents the total number of particles in the sample (or total fraction = 1, if normalised). When temperature changes but the number of particles is constant:
the area stays the same
the shape changes (peak height and width adjust)
Key Features to Read From the Curve
The curve is not symmetric: it rises quickly from near zero, reaches a peak, then trails off gradually at higher speeds.
Most Probable Speed
The peak occurs at the most probable speed, the single speed possessed by the greatest number of particles.
Most probable speed: The speed at which the Maxwell–Boltzmann distribution reaches its maximum; the largest fraction of particles have this speed.
A distribution also implies many particles have speeds near the peak, and fewer particles occur far to the left or right.
Spread and Tails
“Spread” refers to how wide the distribution is:
a narrower curve means particle speeds are more tightly clustered
a broader curve means a wider variety of speeds is present
The high-speed tail is particularly important: even at moderate temperatures, a small fraction of molecules have very high speeds compared with the average.
Comparing Distributions as Temperature Changes
This is the primary AP use of Maxwell–Boltzmann plots: comparing how the distribution changes as temperature increases or decreases.
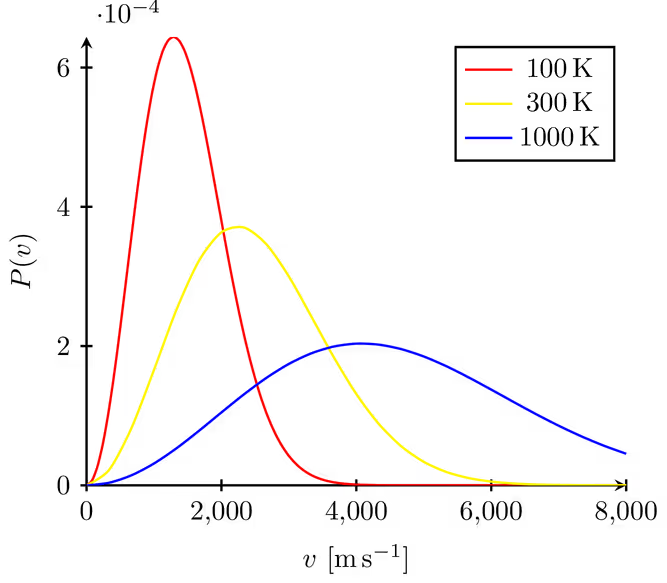
Maxwell–Boltzmann speed distributions for the same gas at three temperatures (100 K, 300 K, and 1000 K), plotted as probability density versus speed . As temperature increases, the peak shifts to higher speeds while the curve broadens and lowers, illustrating how higher increases the fraction of molecules in the high-speed tail. Source
When Temperature Increases
If a gas is heated (same substance, same amount):
the curve shifts to the right (typical speeds increase)
the peak becomes lower
the curve becomes broader (greater spread of speeds)
the high-speed tail lengthens (more particles reach very high speeds)
the area remains constant
Interpretation: higher temperature means the sample contains more particles with larger kinetic energies, but not all particles speed up equally; instead, the whole distribution redistributes.
When Temperature Decreases
If temperature is lowered:
the curve shifts left
the peak becomes higher
the curve becomes narrower
the high-speed tail shrinks
the area remains constant
Interpretation: more particles cluster near lower speeds, and relatively few particles have high speeds.
What “Shift Right” Actually Means
A rightward shift does not mean every molecule moves faster by the same amount. Instead:
collisions constantly reshuffle energy among molecules
at higher temperature, the probability of finding molecules at higher speeds increases
at any temperature, there are still some slow molecules and some fast molecules
Speed Distributions vs Energy Distributions
Maxwell–Boltzmann distributions may be shown as speed or kinetic energy distributions.
Similarities
In both cases, increasing temperature leads to:
a shift toward larger x-values
a lower, broader peak
a larger fraction of particles at high x-values
Differences in Graph Shape (Qualitative)
Because kinetic energy depends on speed squared, energy-distribution plots often appear “stretched” compared with speed plots. For AP interpretation, focus on the same comparative ideas:
position (left/right)
peak height
spread
tail behaviour
Common Interpretation Tasks (What to State Clearly)
When asked to interpret or compare two curves, explicitly address:
Which curve corresponds to the higher temperature (the one further right with a lower peak and broader spread)
How the fraction of fast particles changes (higher → greater fraction in the high-speed region)
Whether particle count changes (if the sample amount is unchanged, area under both curves is the same)
What remains true at both temperatures (a range of speeds exists; collisions maintain a distribution)
Use the graph’s language: “greater fraction beyond a chosen speed,” “peak shifts,” and “distribution broadens,” rather than vague statements like “molecules move faster.”
FAQ
Even though some molecules are slow, a speed of exactly zero is extremely unlikely in a moving gas.
The distribution reflects probability: continual collisions make it improbable for molecules to have precisely zero translational motion at any instant.
Not necessarily.
If the number of molecules is unchanged, a lower peak typically means the curve has broadened, so the same total area is spread across a wider range of speeds.
Energy is shared unevenly through collisions, so a small fraction of molecules can acquire much higher-than-typical speeds.
Because probabilities decrease gradually at high speed, the curve tapers off rather than ending abruptly.
The overall interpretation stays similar (higher temperature shifts the curve towards larger values and broadens it), but the shape appears more stretched.
This is because kinetic energy depends on $v^2$, so equal changes in speed correspond to larger changes in energy at higher speeds.
It assumes the same number of molecules is being represented and the graph is scaled consistently (often as a fraction or probability density).
If one sample has more moles, or the plot is not normalised, areas may differ even if the shapes are comparable.
Practice Questions
Question 1 (3 marks) A Maxwell–Boltzmann speed distribution for a gas at temperature is compared with the distribution for the same gas at a higher temperature . Describe three changes you would observe in the distribution when going from to .
Curve shifts to higher speeds (to the right). (1)
Peak becomes lower (maximum y-value decreases). (1)
Distribution becomes broader / high-speed tail extends / wider spread of speeds. (1)
Question 2 (6 marks) Two Maxwell–Boltzmann speed distributions, A and B, are shown for the same gas sample containing the same number of molecules. Curve A has a taller peak at a lower speed; curve B has a lower peak and extends further into the high-speed region. (a) Identify which curve corresponds to the higher temperature and justify using features of the curves. (3 marks) (b) A vertical line is drawn at speed in the high-speed tail region. Compare the fraction of molecules with speeds greater than for A and B, and explain your reasoning. (3 marks)
Curve B is the higher temperature. (1)
Justification: B is shifted to the right (higher most probable/typical speeds). (1)
Justification: B is broader with a lower peak (greater spread at higher ). (1) (b)
Fraction with is larger for curve B. (1)
Reason: area under the curve to the right of is greater for B (more molecules in high-speed tail). (1)
Clear comparative statement linking higher to increased population of high-speed molecules while total area remains the same. (1)

