AP Syllabus focus: ‘Kelvin temperature is proportional to the average kinetic energy of particles in a sample, so increasing temperature increases average molecular kinetic energy.’
Temperature is the bridge between what you can measure in the lab and what particles are doing microscopically. In AP Chemistry, using the Kelvin scale is essential for linking temperature directly to particle kinetic energy.
Kelvin Temperature as an Absolute Measure
Why Kelvin (not Celsius) matters
The Kelvin scale is anchored at absolute zero, the theoretical point where particle thermal motion is minimised.
Comparison diagram showing how Kelvin, Celsius, and Fahrenheit align on a single temperature axis, with Kelvin starting at 0 K (absolute zero). The figure makes the constant offset between Celsius and Kelvin explicit, which is why temperature ratios must be taken in Kelvin for energy-proportional relationships. Source
Because of this anchoring, Kelvin temperature increases in direct proportion to thermal energy measures used in molecular models.
Kelvin temperature (T): The absolute temperature scale with zero at absolute zero; it uses the same sized degree as Celsius, so .
A key AP-level idea is that proportional relationships in particle models use Kelvin because ratios of temperature must reflect ratios of energy. Celsius is offset by 273.15, so temperature ratios in °C do not correspond to energy ratios.
What “average kinetic energy” means
When discussing gases at the particulate level, kinetic energy refers to energy of motion of particles. “Average” is necessary because particles in any real sample have a range of speeds at the same temperature.
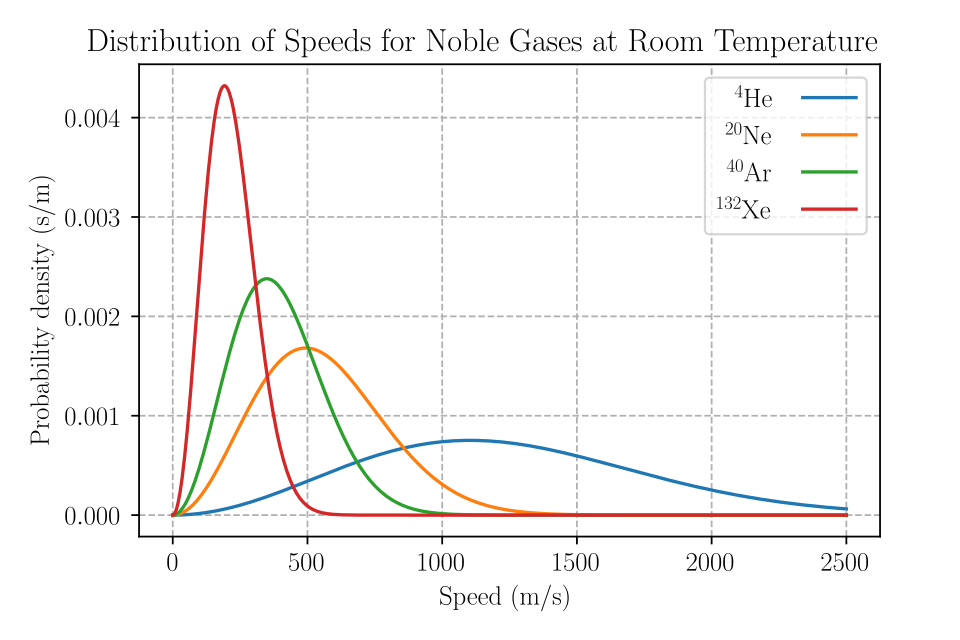
Maxwell–Boltzmann speed distribution curves for several noble gases at the same temperature, illustrating that particle speeds are spread over a range rather than being single-valued. The differing curve positions reflect how particle mass affects typical speeds at a given , which underlies the idea that temperature sets an average kinetic energy while individual particles vary. Source
Average kinetic energy: The mean translational kinetic energy of particles in a sample, reflecting the overall level of particle motion at a given temperature.
In this subsubtopic, the focus is the direct link: higher Kelvin temperature → higher average particle kinetic energy.
The Core Proportionality: and Average Kinetic Energy
The proportional relationship used in AP Chemistry
For a sample at thermal equilibrium, Kelvin temperature is proportional to the average kinetic energy of the particles. This captures the syllabus statement directly: increasing temperature increases average molecular kinetic energy.
One consequence is conceptual and testable: if temperature (in K) increases by a factor, average kinetic energy increases by the same factor.
= Boltzmann constant, J·K
= temperature in K
= gas constant, J·mol·K
= average kinetic energy per mole of particles, J·mol
This equation expresses the proportionality precisely; AP questions often emphasise the relationship even when constants are not required.
What changes (and what does not) when changes
When temperature increases:
Average kinetic energy increases (directly proportional to in K).
Particle speeds increase on average (since kinetic energy depends on speed).
Collisions become more energetic because particles are moving faster.
When temperature is constant:
The average kinetic energy stays the same, even though individual particles may speed up or slow down during collisions.
A faster particle colliding with a slower one can transfer kinetic energy, but the average remains fixed at that temperature.
Connecting Kinetic Energy to Molecular Speed (Qualitatively)
Relating kinetic energy to speed without heavy maths
Because kinetic energy depends on speed, raising Kelvin temperature increases the typical particle speed. However, the relationship between speed and temperature is not linear: kinetic energy scales with the square of speed, so speed increases more slowly than temperature.
At the AP level, you should be able to state:
Higher (K) means greater average kinetic energy, so particles move faster on average.
Heavier particles move more slowly than lighter particles at the same temperature, because reaching the same kinetic energy requires different speeds.
Common AP reasoning moves
To justify statements using the syllabus idea:
Use Kelvin temperature explicitly when discussing proportional changes.
Tie macroscopic change (heating) to microscopic change (increased average kinetic energy).
Distinguish individual particle motion (varies) from the average kinetic energy (set by ).
FAQ
Because $0\ \text{K}$ is defined at the limit of minimal thermal motion.
This makes energy–temperature proportionality meaningful: doubling $T$ in K corresponds to doubling average kinetic energy.
No. Average kinetic energy is per particle (or per mole).
Total kinetic energy also depends on how many particles are present.
No. Collisions constantly redistribute energy.
Temperature fixes the mean value, not individual particle energies.
In gases, translational motion is the component most directly connected to pressure and particle impacts.
The basic AP proportionality uses translational average kinetic energy as the reference.
At the same $T$, they share the same average kinetic energy.
Since $KE = \tfrac{1}{2}mv^2$, a larger $m$ implies a smaller typical $v$ for the same average $KE$.
Practice Questions
(2 marks) Explain why Kelvin temperature, rather than Celsius temperature, is used when stating that temperature is proportional to average kinetic energy.
Kelvin is an absolute scale with zero at absolute zero / no negative temperatures in typical conditions (1)
Proportionality and temperature ratios only work with an absolute scale; Celsius has an offset (1)
(5 marks) A sample of gas is heated from to at constant amount of gas.
(a) State how the average kinetic energy changes (as a factor).
(b) Describe what happens to average molecular speed.
(c) Explain, in terms of particles, why the average kinetic energy is the same for all gases at the same temperature.
(a) Average kinetic energy increases by a factor of (1)
(b) Average molecular speed increases (1)
Speed does not increase by a factor of 1.5; it increases less because (1)
(c) Temperature sets average translational kinetic energy (1)
At the same temperature, particles (regardless of gas identity) have the same average kinetic energy; lighter gases therefore move faster than heavier ones at that (1)

