AP Syllabus focus: ‘Use particulate models of mixtures to represent interactions between components, illustrating how particles attract or repel within a solution.’
Particulate models are visual tools that connect what cannot be seen (particles and forces) to what is observed (mixing, separation, viscosity, and conductivity). In AP Chemistry, these diagrams must clearly show interactions, not just identities.
What a particulate model must communicate
A high-quality particulate representation of a mixture or solution should make the cause of mixing behaviour visible: attractive and repulsive interactions among particles.
Particulate model: A particle-level drawing that represents the types, relative positions, and orientations of species in a substance or mixture to communicate interactions and resulting behaviour.
In this subtopic, the emphasis is not on exact geometry or counting particles precisely, but on whether the diagram correctly encodes how and why particles interact.
Showing attractions vs repulsions
Visual cues for attraction
Use features that explicitly imply particles are stabilised near one another.
Clustering or close approach of unlike particles when solute–solvent attraction is strong
Consistent orientation of polar molecules around charged or polar species (an organised “shell”)
Dashed lines (or similar) to indicate specific strong attractions when appropriate (e.g., hydrogen bonding), as long as the meaning is clear and used consistently
Interspersed distribution of solute throughout solvent (not segregated) to suggest favourable attractions that maintain mixing
Visual cues for repulsion or weak interaction
Repulsion is often shown indirectly, by a lack of stable contact or by separation patterns.
Segregation into regions (clumps of solute, or layers of two liquids) to suggest weak solute–solvent attraction
Random orientations with no persistent alignment near a solute when interactions are minimal
Greater average spacing between species that do not interact favourably (while still respecting that liquids remain close-packed overall)
Representing specific interaction types in mixtures
Polar molecular solute in a polar solvent
To show dipole-based attractions, include partial charges and orient molecules accordingly.
Label ends as and (or use clear shading) on polar molecules
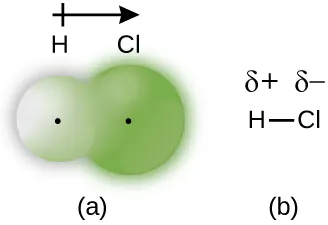
Electron density is unevenly distributed in a polar covalent bond, producing partial charges labeled and . This labeling convention is a standard way to encode dipole direction in particulate models so that electrostatic attractions are visually explicit. Source
Draw solvent molecules oriented so opposite partial charges face each other (attraction)
Indicate that these attractions occur throughout the sample, not just at one location
Ions in a polar solvent (hydration/solvation sketches)
When modelling ionic species in a polar solvent, the key idea is charge–dipole attraction.
Ions in water are stabilized by charge–dipole attractions, producing a hydration (solvation) shell of many surrounding water molecules. The diagram emphasizes orientation: the oxygen end of water points toward cations, while the hydrogen end points toward anions, illustrating why ions can remain dispersed in solution. Source
Place cations near the negative end of solvent dipoles
Place anions near the positive end of solvent dipoles
Show several solvent molecules surrounding each ion to imply stabilisation by many attractions, not a single “bond”
Avoid drawing ions as covalently attached to solvent; the interaction is noncovalent and dynamic
Nonpolar solute with a polar solvent
To illustrate weak attraction between unlike interaction types:
Show nonpolar particles with no partial charges
Keep polar solvent molecules primarily interacting with each other (no organised solvent orientation around the nonpolar solute beyond incidental contact)
If depicting poor mixing, show aggregation of nonpolar solute particles rather than uniform dispersion
Common diagram expectations and pitfalls
What graders look for
Correct identification of species (solute vs solvent) and their particle nature (ions vs molecules)
Correct relative arrangement/orientation that matches attraction/repulsion claims
A depiction that supports the stated macroscopic claim (e.g., “mixes well” corresponds to dispersed solute with clear attractions)
Frequent mistakes to avoid
Treating intermolecular attractions as rigid bonds (especially around ions)
Showing a “solution” with solute particles all at the bottom (unless modelling undissolved excess)
Omitting polarity cues (no labels or orientation) while claiming dipole-driven interactions
Drawing particles extremely far apart in a liquid (liquids are close-packed even when interactions are weak)
FAQ
Use consistent visual conventions: closer average spacing, more frequent unlike–unlike contacts, and clearer orientation patterns indicate stronger attractions.
You can also vary the number of solvent molecules drawn interacting with a solute to suggest more stabilising contacts.
Use $\delta+$ and $\delta-$ labels at molecular ends, or a simple shading convention (dark = negative end, light = positive end).
Be consistent across all species in the same diagram.
Only if they add clarity to the interaction claim you are making.
If you include them, use dashed lines and ensure they connect appropriate atoms (H on one molecule to an electronegative atom on another), rather than random links.
Indicate randomness plus local structure: draw a representative “snapshot” with correct orientation near key particles, while keeping the rest irregular.
Avoid perfectly repeating patterns, which can incorrectly suggest a crystal lattice.
They compress a 3D, moving system into a 2D, fixed picture, so they can exaggerate order or hide time-dependent rearrangements.
They also cannot directly show energy changes; they only imply them through interaction patterns.
Practice Questions
A student draws a particulate model of NaCl dissolved in water. Describe two features the model should include to show particle attractions in the solution.
Water molecules oriented with the oxygen (negative end) towards and/or hydrogens (positive end) towards (1)
Ions separated and surrounded by multiple water molecules (hydration shells), indicating attraction throughout the mixture (1)
Two liquids, X and Y, are mixed. X is polar; Y is non-polar. The mixture forms two layers after shaking.
(a) Describe what a correct particulate model would show immediately after shaking. (2 marks)
(b) Describe what the model would show after the layers reform, focusing on interactions. (2 marks)
(c) State one diagram feature that would be incorrect if used to justify the layering. (1 mark)
(a)
Temporary dispersion of Y particles among X particles, but without consistent dipole alignment between X and Y (1)
X molecules still shown close together with orientations reflecting X–X attractions more than X–Y (1)
(b)
Separation into two regions with mostly X in one layer and mostly Y in the other (1)
Minimal organised attraction between X and Y at the interface; stronger like–like interactions within each layer (1)
(c)
Any incorrect feature, e.g. showing strong dashed “bonds” between X and Y throughout the sample, implying strong attraction/miscibility (1)

