AP Syllabus focus: ‘Particulate representations communicate relative concentrations by showing appropriate ratios and counts of components in the same volume of solution.’
Particulate models make “invisible” composition visible by showing solute and solvent particles as counts in a fixed volume. Done correctly, these diagrams communicate relative concentration, dilution, and mixing without relying on numerical calculations.
Core idea: concentration as “particles per volume”
When you draw a particulate representation, you are encoding concentration with how many solute particles appear in the same-sized box (the same volume).
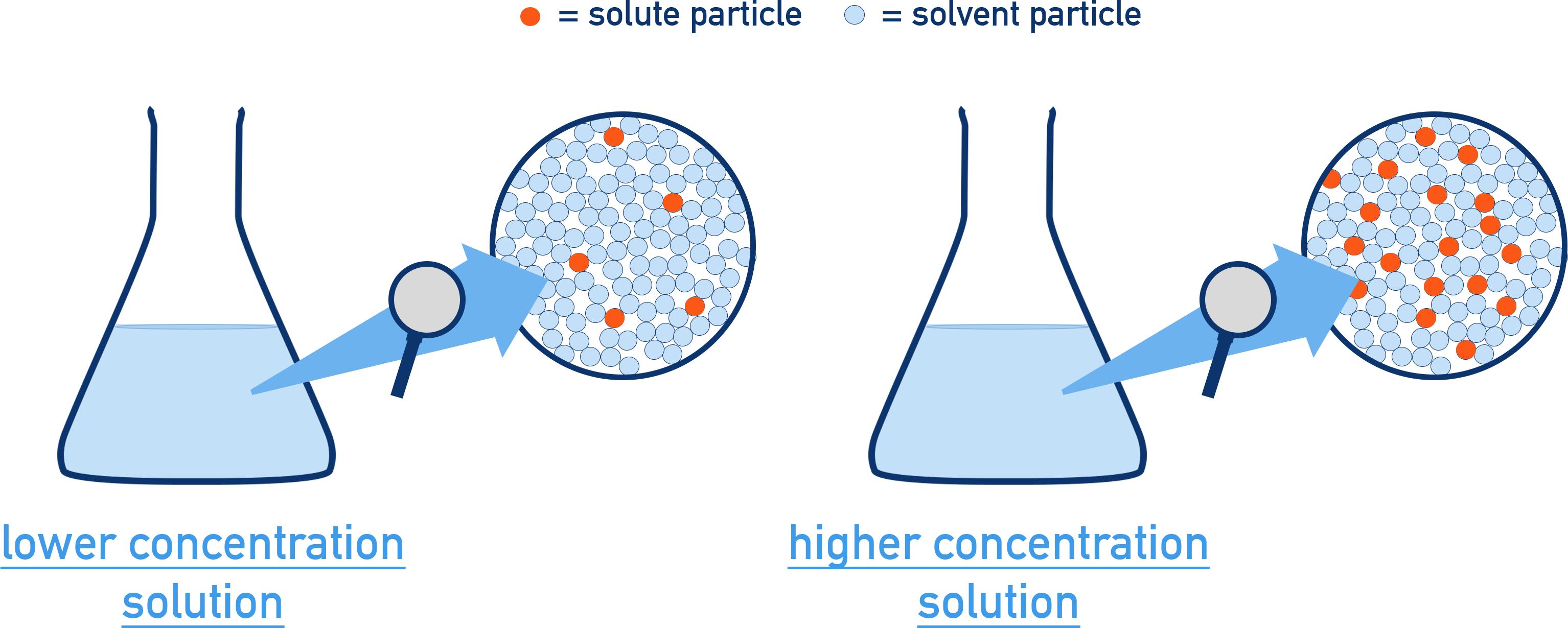
Side-by-side particulate “windows” depict a lower- vs. higher-concentration solution using the same pictured volume. The diagram makes concentration a number-density idea: more solute particles per fixed region (not bigger particles) corresponds to a higher concentration. Source
Concentration: The relative amount of solute present per a given volume of solution; in particulate models, it is represented by solute particle count in a fixed volume.
A key rule for AP-style diagrams is to keep the volume of the pictured region constant when comparing concentrations. If the pictured volume changes, the particle count alone is not meaningful.
How to show concentration in particulate diagrams
Use a constant “window” (same volume)
To compare two solutions’ concentrations:
Draw identical-sized containers/boxes (same volume).
Keep particle symbols the same size (don’t “shrink” particles to fit more).
Compare concentration using solute count per box.
Show concentration using particle counts and ratios
Concentration is communicated by:
Solute particle count: more solute particles in the same volume means higher concentration.
Solute-to-solvent ratio: a higher ratio indicates a more concentrated solution (even if the solvent count also changes).
Relative comparisons: “A is more concentrated than B” should be obvious from the diagram without labels.
Distinguish different species clearly
If multiple species are present, concentration information depends on correct identification:
Use different colours/shapes for different solutes (or ions vs molecules).
Use a consistent legend (implicit or explicit) so that “counts” are unambiguous.
For ionic solutes, depict separate ions as the counted particles (each ion is a particle in the model).
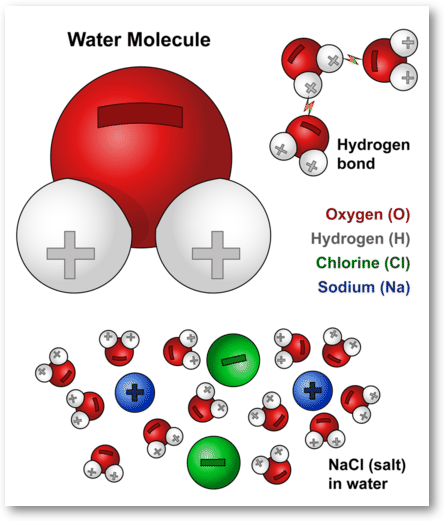
This molecular-level diagram shows how water’s partial charges interact with ions during dissolution: oxygen (δ−) is oriented toward cations (e.g., Na) and hydrogens (δ+) toward anions (e.g., Cl). It provides a clear visual basis for drawing hydrated, separated ions rather than intact formula units in particulate representations. Source
Common comparison tasks (conceptual, not computational)
Concentrated vs dilute
A dilute particulate diagram shows:
Few solute particles scattered among many solvent particles.
A concentrated particulate diagram shows:
Many solute particles in the same volume, still dispersed if dissolved.
Dilution as a visual change
To show dilution while staying particulate-level:
Keep solute particle count the same for a fixed amount of solute, but represent a larger volume with more solvent; or, for comparisons that must keep volume fixed, show that dilution corresponds to fewer solute particles per fixed box.
Avoid implying dilution by changing particle size or fading colour; dilution is a count/ratio idea.
Comparing mixtures with more than one solute
When two solutes are present, you may need to communicate:
Total solute concentration (count all solute particles).
Individual solute concentrations (count each solute type separately).
Relative proportions of solutes (compare solute A count to solute B count within the same volume).
Quality checks (what makes a diagram “AP-usable”)
A strong particulate representation of concentration should:
Use appropriate ratios and counts (not random spacing that obscures counting).
Keep the same volume of solution when the goal is concentration comparison.
Show solute particles evenly distributed to indicate a homogeneous solution (no clumps unless the intent is to show undissolved solute, which would change what “concentration” means).
FAQ
Use distinct symbols for each solute and count only the target solute within the same fixed volume. Keep the other solute’s symbols present so proportions remain visible.
Keep the viewing box the same size each time, then systematically reduce solute count per box while keeping particle symbols unchanged. Label each step consistently.
Use a larger representative sample window (still consistent across comparisons) or show fewer solute particles with clear spacing, but avoid implying “none” unless truly zero.
Particle size is not concentration; changing it introduces a second variable that can mislead the reader into perceiving “more matter” without an actual increase in particle count.
Use a repeated pattern (e.g., a cluster of 2 solute : 10 solvent repeated) and state that the pattern repeats uniformly, so the intended ratio is readable without overcrowding.
Practice Questions
Two particulate diagrams show solutions in equal-sized boxes. Diagram A contains 4 solute particles; Diagram B contains 8 solute particles. State which is more concentrated and justify using the particulate information.
Identifies Diagram B as more concentrated. (1)
Justifies: same volume but greater solute particle count (or higher solute-to-solvent ratio if mentioned). (1)
A student draws particulate models for two salt solutions in equal volumes. In Model 1, there are 6 cations and 6 anions. In Model 2, there are 6 “salt pairs” drawn as touching cation–anion units (still dispersed).
(a) Explain which model more clearly communicates concentration and why. (2)
(b) Describe two specific diagram rules that ensure concentration comparisons are valid between two particulate models. (2)
(c) State one common diagramming mistake that could falsely suggest a higher concentration. (1)
(a)
Model 1 is clearer because it shows the correct counted particles (separate ions) in solution. (1)
Concentration is communicated by counting particles per fixed volume; touching “pairs” can obscure counting/particle identity. (1)
(b) Any two:
Use equal-sized boxes to represent equal volumes. (1)
Keep particle sizes consistent. (1)
Use distinct symbols/colours for different species and count consistently. (1)
Keep distribution homogeneous so counting isn’t biased by clustering. (1)
(b) Any one:
Changing the pictured volume/box size without stating it. (1)
Shrinking particles to fit more in the same box. (1)
Using inconsistent symbols so solute counts are ambiguous. (1)

