AP Syllabus focus: ‘Colligative properties and calculations of molality, percent by mass, and percent by volume are excluded from assessment; focus on particulate models and concentrations.’
These notes clarify what solution topics you are not responsible for on the AP Chemistry exam and what you should emphasise instead: particulate representations and straightforward concentration comparisons.
What the exam explicitly excludes
The AP Chemistry assessment for this subsubtopic does not test colligative properties or concentration calculations using molality, percent by mass, or percent by volume. You should be able to recognise these ideas, but you are not expected to perform exam calculations with them in this unit.
Colligative properties (not assessed here)
Colligative properties: Solution properties that depend on the number of dissolved solute particles (relative to solvent), not the solute’s chemical identity.
In many chemistry courses, colligative properties include freezing-point depression, boiling-point elevation, vapour-pressure lowering, and osmotic pressure. For AP Chemistry in this section, these are outside the required skill set, so avoid building explanations around them when answering questions tied to solution representations.
Alternative concentration units (not assessed here)
Molality (m): Moles of solute per kilogram of solvent.
Molality is commonly used when temperature changes matter (because it uses mass of solvent rather than solution volume), but molality calculations are excluded for this subsubtopic.
Percent by mass (% m/m): .
Percent by mass is frequently used for mixtures prepared by weighing, but it is not required for assessment in this section.
Percent by volume (% v/v): .
Percent by volume is often used for liquid–liquid solutions, but it is also excluded from assessment here.
What you should focus on instead
The emphasis is on particulate models and concentration in a qualitative or directly stated quantitative sense.
Your goal is to connect what a diagram shows (types of particles and their relative numbers) to what it implies about a solution.
Particulate representations as “concentration evidence”
When shown a particulate diagram of a solution (or asked to draw one), prioritise these exam-relevant ideas:
Identity of particles present
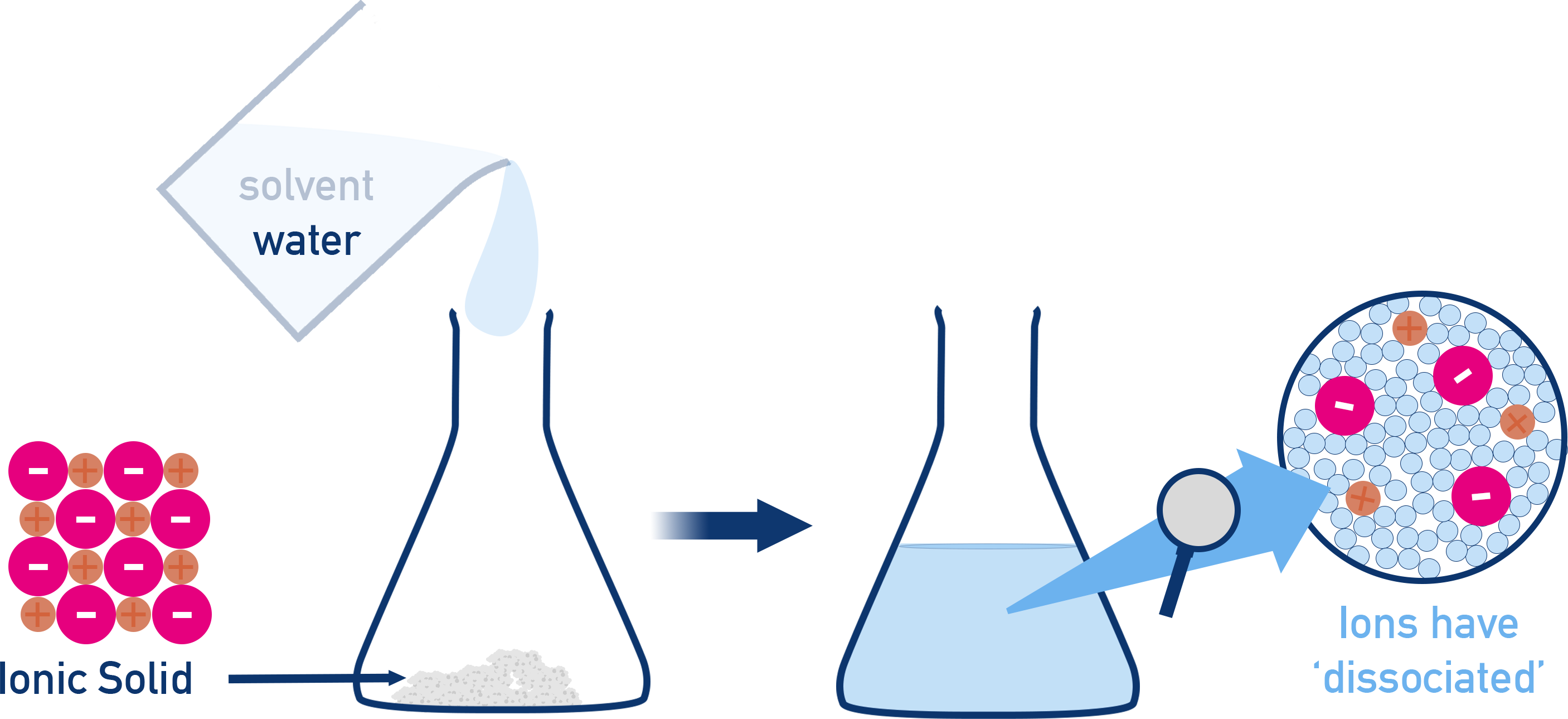
Schematic of an ionic solid dissolving to form an aqueous solution where the solute is represented as separate cations and anions. It reinforces that particle diagrams should show dissociation for soluble ionic compounds, so the ‘particles present’ are ions dispersed throughout the solvent. Source
Distinguish solute vs solvent particles as shown.
If multiple solutes are present, keep their particle types visually distinct.
Relative concentration
For the same depicted volume, more solute particles implies a more concentrated solution.
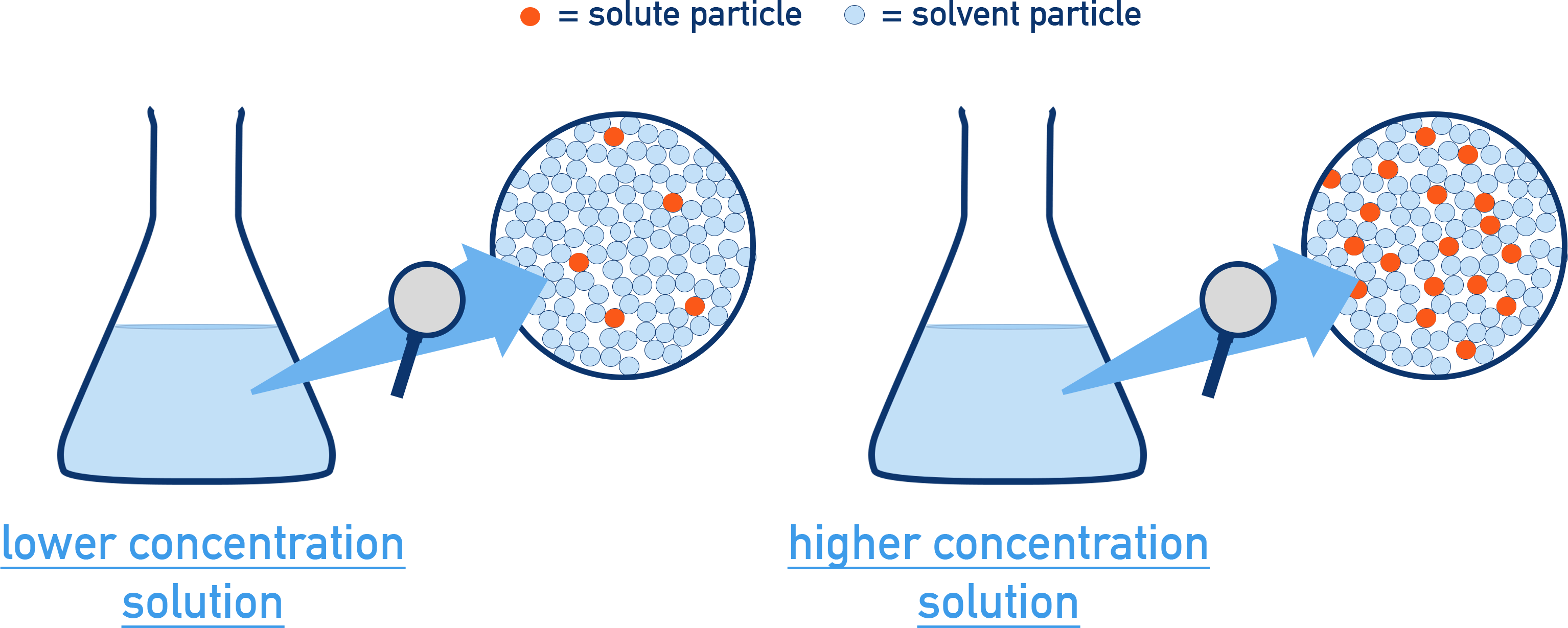
Particulate-level comparison of two solutions at the same depicted volume. The higher-concentration sample is represented by a greater number of solute particles dispersed among solvent particles, emphasizing concentration as “particles per volume” rather than any temperature-dependent unit. Source
For the same number of solute particles, a larger depicted volume implies a more dilute solution.
Consistency across diagrams
A “fair” comparison requires the same volume scale (or an explicitly stated equal-volume assumption).
Language to use in explanations
Because colligative-property reasoning is excluded, use diagram-based wording such as:
“Solution A has a higher concentration because it shows a greater number of solute particles per equal volume.”
“Both solutions have the same concentration because the solute-to-solution-volume ratio is the same in each representation.”
“The mixture is homogeneous because the particles are uniformly distributed throughout the sample shown.”
Common pitfalls to avoid (specific to this subsubtopic)
Do not justify a concentration claim by referencing boiling point, freezing point, or vapour pressure changes.
Do not switch to % by mass, % by volume, or molality to compare solutions in this unit.
Do not assume concentration from particle “clumping” unless the question explicitly frames an interaction; for this subsubtopic, the key is count-per-volume, not aggregation.
FAQ
Because % by mass uses masses while molarity uses solution volume. Converting typically needs the solution’s density (to relate volume to mass) and a clear definition of “solution” vs “solvent”.
Molality is based on solvent mass, so it is less sensitive to temperature-driven volume changes. It is often preferred for work involving heating/cooling and for certain thermodynamic measurements.
Yes. It is usually treated as a very dilute mass fraction. Common approximations include:
aqueous solutions: $1\ \text{ppm} \approx 1\ \text{mg/L}$ (only under specific density assumptions)
Follow the prompt’s conventions. If ions are shown separately, label cations and anions; if intact formula units are shown, label them as the solute particles depicted, without adding unstated dissociation.
Volumes are not always additive on mixing (contraction/expansion can occur). So % v/v can depend on how “volume of solute” is defined and measured, especially for non-ideal liquid mixtures.
Practice Questions
(2 marks) State two solution topics or calculation types that are excluded from assessment for this subsubtopic, and state what students should focus on instead.
1 mark: Identifies any two excluded items (e.g. colligative properties; molality; percent by mass; percent by volume).
1 mark: States focus is on particulate models and concentration (e.g. relative number of particles per volume).
(5 marks) Two particulate diagrams show equal volumes of solution. Diagram A contains 6 solute particles dispersed among 24 solvent particles. Diagram B contains 12 solute particles dispersed among 24 solvent particles.
(a) Identify which diagram represents the more concentrated solution and justify using particulate reasoning. (3 marks)
(b) Give one statement of reasoning that would be inappropriate for this subsubtopic and explain why. (2 marks)
1 mark: Correct choice (Diagram B).
1 mark: Uses equal volume comparison.
1 mark: Justifies via higher solute particle count (higher solute-per-volume or higher solute-to-solvent ratio). (b)
1 mark: Provides an inappropriate statement (e.g. “B has a higher boiling point/freezing point change” or uses molality/%).
1 mark: Explains it is excluded from assessment here (colligative properties/alternative concentration units not tested in this subsubtopic).

