AP Syllabus focus: ‘In aqueous solution, water can accept protons from or donate protons to dissolved species because of its molecular structure.’
Water is not a passive background in aqueous acid–base chemistry. Its polarity and lone pairs let it participate directly in proton transfer, control which species actually exist in solution, and shape how reactions are represented.
Water as a Brønsted–Lowry participant
In water-based acid–base reactions, water molecules frequently act as the proton-transfer partner, even when they are not written explicitly. This is possible because the oxygen atom has two lone pairs (proton-accepting sites) and the O–H bonds contain polar, partially positive H atoms (proton-donating sites).
Amphiprotic (amphoteric): able to donate a proton (act as a Brønsted–Lowry acid) or accept a proton (act as a Brønsted–Lowry base), depending on the reaction partner.
Because water is amphiprotic, the direction of proton transfer depends on the dissolved species:
With an acid, water commonly acts as the base (accepts ).
With a base, water can act as the acid (donates ).
Why “” becomes hydronium in water
A “free” proton is not stable in aqueous solution; it is immediately associated with water. For AP Chemistry purposes, aqueous acid reactions are typically described as producing hydronium rather than isolated .
Hydronium, : the protonated form of water produced when accepts a proton; it is the primary representation of “aqueous ”.
This matters for both particle-level understanding and for writing aqueous reactions: acids increase , and bases decrease it (often by producing that neutralizes ).
Water’s self-ionization sets the baseline for acidity
Even pure water contains small amounts of ions due to autoionization (self-ionization).
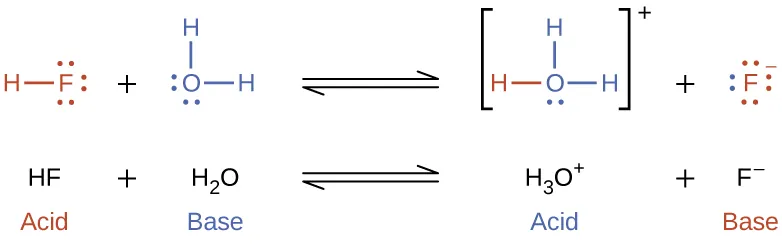
Autoionization of water shown with Lewis structures: one donates a proton (acid) while another accepts it (base), forming and . The color-coding highlights conjugate acid–base relationships and makes it clear why water is amphiprotic in aqueous acid–base chemistry. Source
This equilibrium establishes that both and are always present in aqueous solution, even before any solute is added.
= hydronium ion concentration in
= hydroxide ion concentration in
= ion-product constant of water (temperature-dependent)
= equilibrium concentration in
Water’s autoionization is the chemical reason aqueous acid–base chemistry can be tracked through and : changing one necessarily affects the other through .
Water as the solvent that stabilizes ions
Water’s polarity and ability to form hydrogen bonds stabilize ions in solution via strong ion–dipole attractions.
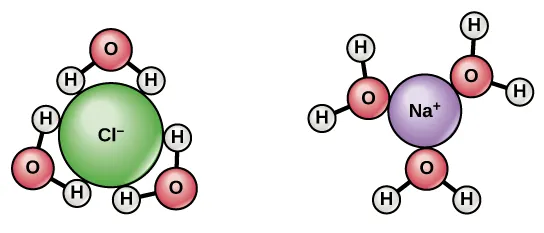
Hydration shells around ions in water: oxygen ends of water molecules point toward , while hydrogen ends point toward . This orientation illustrates ion–dipole attractions, a key reason ions are stabilized in aqueous solution and many acid–base processes proceed readily in water. Source
This stabilization:
Makes it energetically feasible for many acids/bases to form ions in water.
Influences which species predominate (molecules vs ions), because solvated ions can be substantially lower in energy than unsolvated ones.
Helps explain why aqueous acid–base reactions often proceed readily: the products (, , and other ions) are strongly solvated.
Writing aqueous acid–base reactions with water included
Because water is often a reactant or product, it may appear explicitly in balanced equations, particularly when showing proton transfer:
Acid in water: the acid donates a proton to water, producing and the acid’s conjugate base.
Base in water: the base accepts a proton from water, producing and the base’s conjugate acid.
When representing aqueous systems, remember that water can be both:
The proton acceptor that converts “acid strength” into formation of .
The proton donor that converts “base strength” into formation of .
FAQ
$H^+$ does not exist as an isolated ion in water; it is strongly associated with surrounding water molecules.
$H_3O^+$ is a convenient, AP-appropriate representation, but real proton solvation can involve larger clusters (e.g., $H_5O_2^+$).
$K_w$ increases as temperature rises, so $[H_3O^+]$ and $[OH^-]$ in pure water both increase.
“Neutral” still means $[H_3O^+] = [OH^-]$, but the numerical value is temperature-dependent.
Yes. Water strongly stabilises ions and supports rapid proton transfer through hydrogen-bond pathways.
In less polar or non-hydrogen-bonding solvents, acids/bases may ionise far less and different species can dominate.
Water’s bent shape and polar O–H bonds concentrate electron density on oxygen (lone pairs) and create a strong dipole.
This enhances attraction to $H^+$ and stabilises the resulting $H_3O^+$ via further hydrogen bonding.
Often not exclusively. At high acid concentrations, water becomes limited and protonated species can shift (e.g., more extensively protonated water clusters).
This is one reason very concentrated acids deviate from idealised dilute aqueous behaviour.
Practice Questions
(2 marks) Explain, using Brønsted–Lowry ideas, why water can react with both acids and bases in aqueous solution.
Water can accept from an acid (acts as a Brønsted–Lowry base). (1)
Water can donate to a base (acts as a Brønsted–Lowry acid). (1)
(5 marks) For aqueous ammonia, , write an equation showing water’s role in the acid–base process. Then state the two conjugate acid–base pairs present and identify which species is acting as the Brønsted–Lowry acid in this reaction.
Correct equation: . (2)
Conjugate pair: / . (1)
Conjugate pair: / . (1)
is the Brønsted–Lowry acid (proton donor) in this reaction. (1)

