AP Syllabus focus: ‘A Brønsted–Lowry acid donates a proton and a Brønsted–Lowry base accepts a proton; identify conjugate acid–base pairs in proton-transfer reactions.’
Brønsted–Lowry theory explains acid–base reactions as proton-transfer processes. In AP Chemistry, you must define acids and bases by proton movement and correctly match each species to its conjugate partner.
Brønsted–Lowry acids and bases
A Brønsted–Lowry acid–base reaction always involves movement of an H⁺ (proton) from one species to another. The key skill is deciding which reactant loses H⁺ and which reactant gains H⁺.
Brønsted–Lowry acid: a species that donates a proton (H⁺) to another species.
The acid is the reactant whose formula can be viewed as decreasing by one H atom in the products.
Brønsted–Lowry base: a species that accepts a proton (H⁺) from another species.
The base is the reactant whose formula can be viewed as increasing by one H atom in the products.
Recognising proton transfer in equations
When comparing reactants to products, track where an H atom ends up:
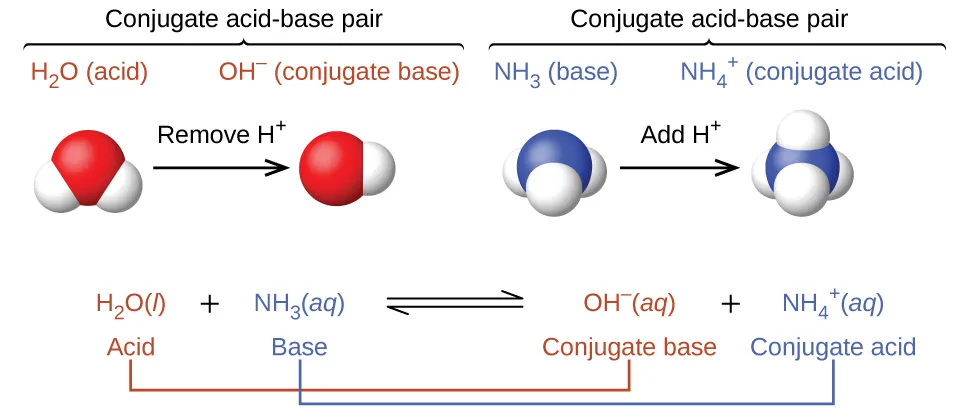
A Lewis-structure-style view of water autoionization: one donates (acting as an acid) while another accepts (acting as a base), producing and . The product charges provide a clear consistency check: the proton acceptor becomes more positive and the proton donor becomes more negative. Source
Identify a reactant that contains H and becomes a product with one fewer H (that reactant acted as the acid).
Identify a reactant that becomes a product with one more H (that reactant acted as the base).
Use charges as a consistency check: losing H⁺ makes a species more negative (or less positive); gaining H⁺ makes it more positive (or less negative).
Conjugate acids and conjugate bases
Each acid–base reaction produces products that are chemically related to the original reactants by exactly one proton.
Conjugate base: the species formed when an acid donates H⁺ (it has one fewer H than the acid).
A conjugate base is what remains after the proton leaves the acid; it can potentially accept H⁺ in the reverse direction.
Conjugate acid: the species formed when a base accepts H⁺ (it has one more H than the base).
A conjugate acid is the “protonated” form of the base; it can potentially donate H⁺ in the reverse direction.
Conjugate acid–base pair: two species that differ by exactly one proton (H⁺), interconverted by proton gain/loss.
General form of a Brønsted–Lowry reaction
This template helps you label roles and pairs in any proton-transfer equation.
= acid (proton donor)
= base (proton acceptor)
= conjugate base of
= conjugate acid of
In this pattern, HA/A⁻ is one conjugate pair, and B/HB⁺ is the other conjugate pair.
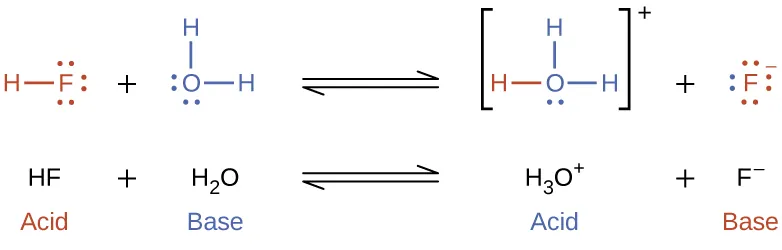
A labeled proton-transfer example showing how (acid) donates to (base) to form (conjugate base) and (conjugate acid). The brackets and color-coding make the “reactant → conjugate” pairing visually explicit: acid conjugate base, and base conjugate acid. Source
How to identify conjugate pairs (AP skill)
When given a proton-transfer reaction, identify pairs by matching “before” and “after” forms:
Find the species that lost H: reactant acid → product conjugate base.
Find the species that gained H: reactant base → product conjugate acid.
Pair each reactant with the product that differs by one H only (ignore coefficients; focus on formulas and charge).
Common pitfalls to avoid
Do not label acids/bases by the presence of OH alone; in Brønsted–Lowry, the deciding feature is H⁺ transfer.
Do not confuse a “conjugate pair” with any two species on opposite sides; they must differ by exactly one proton.
If multiple H-containing species appear, only one proton is transferred per elementary Brønsted–Lowry step as written; match the specific H change shown.
FAQ
A Brønsted–Lowry base can be neutral (e.g. $NH_3$) or negatively charged (e.g. $OH^-$).
What matters is whether it can accept $H^+$, typically using a lone pair.
Treat $H^+$ as the acid species.
The conjugate base of $H^+$ is “none” in a simple sense, but in real aqueous chemistry $H^+$ is associated with solvent; exam questions usually provide a fuller species if needed.
Polyprotic acids form a sequence of conjugate bases by losing one proton at a time, e.g. $H_3A \rightarrow H_2A^- \rightarrow HA^{2-} \rightarrow A^{3-}$.
Each adjacent pair differs by exactly one $H^+$.
Because $H^+$ carries a $+1$ charge.
Donating $H^+$ makes the donor’s charge decrease by 1 (more negative).
Accepting $H^+$ makes the acceptor’s charge increase by 1 (more positive).
They are not a conjugate acid–base pair in a single Brønsted–Lowry step.
Look for an intermediate form that differs by one $H^+$, or re-check the equation: AP questions typically present pairing that is one-proton apart.
Practice Questions
Given the reaction , identify: (i) the Brønsted–Lowry acid, (ii) the Brønsted–Lowry base, and (iii) one conjugate acid–base pair.
Acid identified as (1)
Base identified as (1)
One correct conjugate pair stated, e.g. or (1)
For the reaction :
(a) State which reactant is the Brønsted–Lowry acid and which is the Brønsted–Lowry base. (2 marks)
(b) Identify both conjugate acid–base pairs. (4 marks)
(a) is the acid (proton donor) (1)
(a) is the base (proton acceptor) (1)
(b) Pair: (2)
1 mark for correct matching, 1 mark for recognising they differ by one
(b) Pair: (2)
1 mark for correct matching, 1 mark for recognising they differ by one

