AP Syllabus focus: ‘Lewis acid–base concepts are not assessed; the emphasis is on acid–base reactions in aqueous solution.’
Acid–base chemistry can be described using multiple models. For AP Chemistry, you are expected to use the model that best matches aqueous proton-transfer reactions, and to avoid relying on out-of-scope terminology.
What “scope” means on the AP Exam
The syllabus statement is an exam note that sets boundaries on what you are assessed on. In practice, it means:
You should explain acid–base reactions in water using proton transfer and species present in aqueous solution.
You should not need to use the Lewis framework to earn full credit, even if a prompt uses language that resembles it.
When in doubt, prioritise an explanation tied to transfer (often represented as formation in water) and the reactants/products in solution.
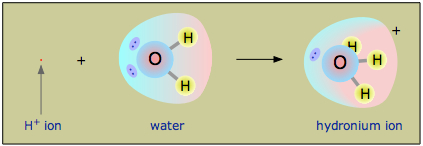
Diagram showing protonation of water to form hydronium, illustrating why is represented as in aqueous solution. This directly models acid–base reactions as proton transfer to a solvent molecule rather than as an abstract free proton. Source
The assessed focus: acid–base reactions in aqueous solution
On the AP Exam, acid–base reasoning is anchored in what happens in water:
Identify which species in solution can donate a proton and which can accept a proton.
Track how proton transfer changes formulas/charges (for example, gaining increases charge by ).
Use aqueous-species thinking (what is actually present as dissolved ions/molecules) rather than abstract “electron-pair” arguments.
Keep your explanations aligned to aqueous conditions stated or implied by the problem (e.g., “in water,” “aqueous,” beakers/solutions, titrations).
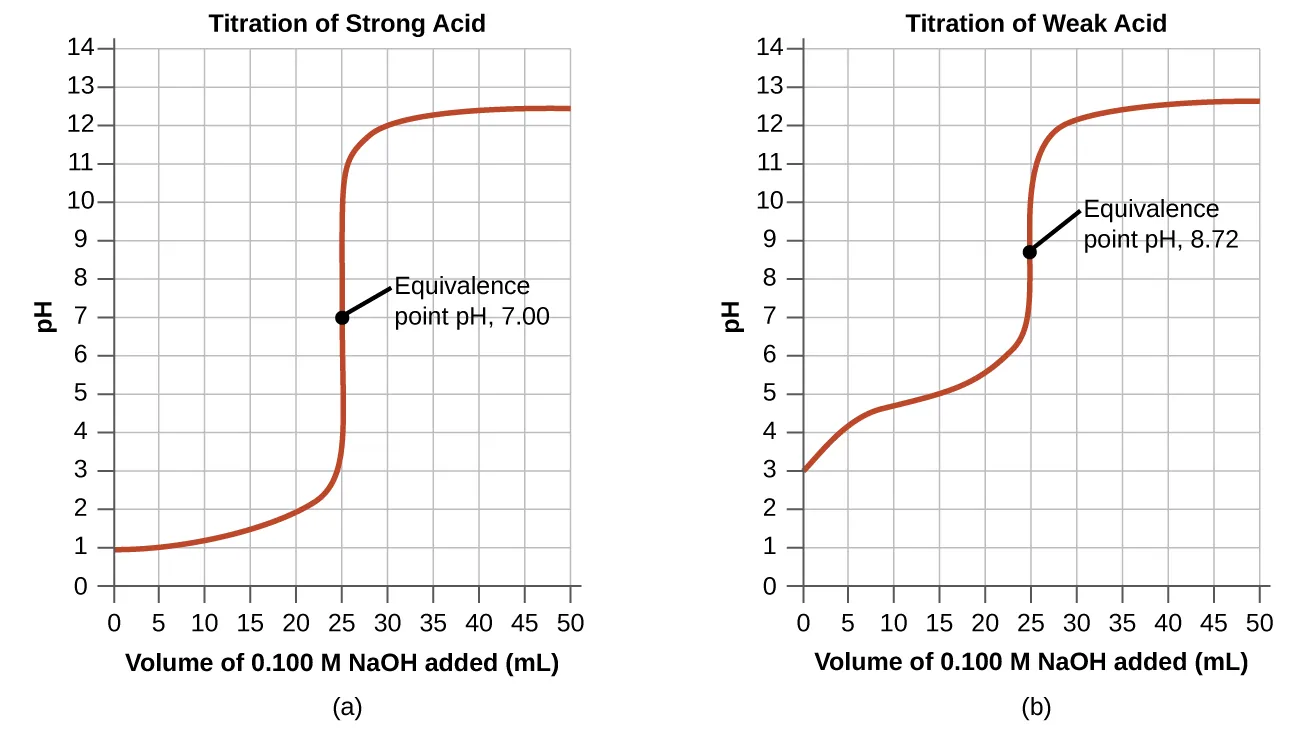
Titration curves comparing strong-acid/strong-base and weak-acid/strong-base titrations, with equivalence points clearly indicated. The graph shows how pH changes as base is added and why the equivalence-point pH depends on the acid/base strengths (i.e., the dominant aqueous species after neutralization). Source
What is explicitly not assessed: Lewis acid–base
Lewis ideas may appear in general chemistry, but the AP Chemistry exam does not require them here.
Lewis acid–base (not assessed): A Lewis acid accepts an electron pair, and a Lewis base donates an electron pair, often described using lone pairs and bond formation.
Even though the Lewis model can sometimes describe the same overall chemical change, you should avoid using it as your primary justification on AP Chemistry acid–base questions, because:
It may shift your explanation away from the exam’s required emphasis on protons in water.
It can introduce claims about electron-pair donation/acceptance that are not necessary for scoring and may not match the expected rubric language.
Common “Lewis-looking” situations and how to stay in scope
Some scenarios can tempt Lewis reasoning; stay within the AP scope by reframing them:
If a species has a lone pair (e.g., ), focus on its role as a proton acceptor in water rather than “electron-pair donor.”
If a species seems “electron-deficient,” do not argue “electron-pair acceptor”; instead, look for the proton-transfer reaction it undergoes in aqueous solution (or note that it may be outside acid–base classification if no proton transfer is involved).
If coordinate (dative) bonding language appears, do not treat that as required acid–base reasoning; return to net proton transfer and the aqueous products formed.
How to write AP-scoring explanations (without Lewis)
Use the expected vocabulary
Use phrasing that directly signals the assessed model:
proton donor / proton acceptor
conjugate forms (conjugate acid/base language if needed by the prompt)
aqueous solution, hydronium, and water acting as acid or base (when relevant to the stated reaction)
Avoid common off-scope phrasing
These are typically unnecessary on AP acid–base scoring guidelines:
“electron-pair acceptor/donor” as the main justification
“forms a coordinate covalent bond” as the decisive reasoning step
“Lewis acidity/basicity” rankings as evidence
Practical exam strategy for “scope” prompts
When a question seems ambiguous, use these priorities:
First, ask: “Is there a proton transfer in water?” If yes, treat it as the acid–base basis of your explanation.
If a prompt mentions “Lewis,” you can acknowledge it briefly but still answer using aqueous proton transfer, because that is what is assessed.
If no proton transfer is possible from the given species under aqueous conditions, do not force an acid–base label; explain what the syllabus focuses on and stick to what can be justified in water.
FAQ
Yes, but keep it secondary.
Lead with proton-transfer in water so your answer matches the marking focus.
Restate the idea in aqueous proton-transfer terms.
Then answer using species present in solution (e.g., $H_3O^+$, conjugates).
Typically no for this scope statement.
If mentioned, translate it into what changed via $H^+$ transfer in water.
Yes.
It signals you should discuss protons, hydronium, and solution species rather than electron-pair frameworks.
Use a mental swap:
“electron pair” $\rightarrow$ proton acceptor/donor
“adduct formation” $\rightarrow$ conjugate formation in water
Practice Questions
(2 marks) A student explains an aqueous reaction by stating, “ is a base because it donates an electron pair to .” Using AP Chemistry scope guidance, state whether this reasoning is required and what wording should be emphasised instead.
1 mark: States Lewis electron-pair reasoning is not required/assessed for AP acid–base.
1 mark: Emphasises proton transfer in aqueous solution (e.g., as a proton acceptor; formation of in water).
(5 marks) A question asks about an acid–base reaction “in aqueous solution.” Two explanations are offered: A: “The base is the species that accepts in water, forming its conjugate acid.” B: “The base is the species that donates a lone pair to form a coordinate bond.” (a) Identify which explanation aligns with AP Chemistry assessment scope. (1) (b) Give two reasons the other explanation is not the expected basis for scoring. (2) (c) Write one sentence describing the exam’s stated emphasis for acid–base reactions. (2)
(a) 1 mark: Chooses A.
(b) 1 mark each (any two): Lewis concepts not assessed; focus is proton transfer; coordinate-bond/electron-pair language is unnecessary and may not match rubrics; aqueous context prioritises / transfer.
(c) 2 marks: Clear sentence stating Lewis acid–base is not assessed and emphasis is acid–base reactions in aqueous solution via proton transfer (both parts needed).

