AP Syllabus focus: ‘Kinetics describes how fast reactants are converted to products: the reaction rate is the change in amount (or concentration) per unit time.’
Chemical kinetics focuses on how quickly reactions occur. This page defines reaction rate, clarifies what “change per unit time” means in chemistry, and shows how rate is represented mathematically and graphically.
Core idea: what a reaction rate measures
Kinetics: The study of how fast reactants are converted to products (reaction speed) and how that speed is quantified.
A reaction proceeds as reactant particles are transformed into product particles, so measurable quantities (like amount in moles or concentration) change as time passes. The reaction rate turns that change into a single numerical description of “how fast.”
Reaction rate: The change in amount (or concentration) of a reactant or product per unit time.
Rates are always tied to a clearly stated chemical species and a chosen measurement basis:
Amount-based rate: uses moles, typically in a fixed-volume system or when tracking total moles directly.
Concentration-based rate: uses molarity (), most common for solutions because concentrations reflect particle density in the reacting mixture.
Mathematical description of rate
When a rate is reported over a time interval, it is an average rate. Average rates depend on the chosen interval because the speed of many reactions changes as concentrations change.
Average rate: The change in amount or concentration over a finite time interval, divided by the length of that interval.
A standard way to express an average rate for a single species is shown below. The sign convention matters: reactants are consumed (decrease), products are formed (increase).
= change in concentration of over the interval,
= elapsed time over the interval, (or another time unit)
= concentration of species ,
To keep “rate” as a positive quantity for reactants, chemists often report rate of disappearance as (because is negative when is consumed). For products, is typically positive.
Units and what they communicate
Common rate units depend on what is measured:
Concentration-based rate: (often written )
Amount-based rate:
Interpreting the unit is conceptual: tells you how many moles per litre are consumed or produced each second, on average over the interval used.
Rate at an instant (idea of instantaneous rate)
Many reactions slow down as reactants are depleted, so a single average rate may hide how the speed changes.
Instantaneous rate: The rate at a specific moment in time, corresponding to the slope of a concentration–time curve at that point.
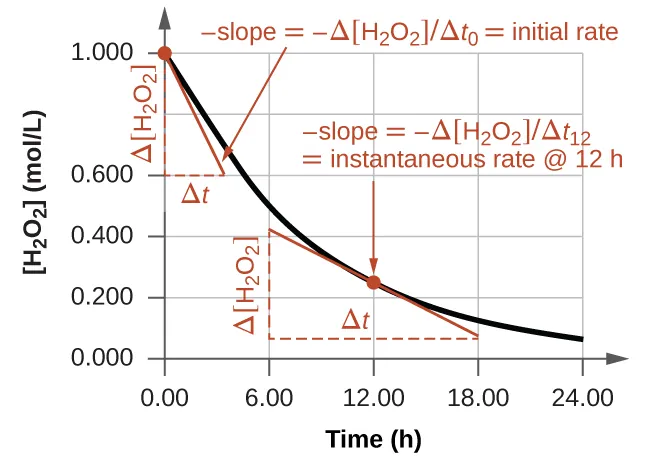
A concentration–time plot for the decomposition of with tangent lines drawn at two times. The instantaneous rate at each time is the (negative) slope of the tangent, illustrating why reactant rates are often reported as to keep rate positive. The right-triangle “rise/run” markers show how a tangent slope is estimated from the graph. Source
You can visualise this by plotting concentration (or amount) versus time:
For a reactant, the curve typically slopes downward; the instantaneous rate relates to the steepness at that time.
For a product, the curve typically slopes upward.
Instantaneous rate is a local concept: it is obtained by considering a very small time interval around the moment of interest (a tangent slope on the graph). In AP Chemistry, the key skill is interpreting what “steeper” or “less steep” means physically: steeper slope = faster change = larger magnitude of rate.
Choosing “amount” versus “concentration” carefully
The syllabus statement emphasises that rate can be defined using amount or concentration. The best choice depends on what stays controlled and what you can measure reliably:
Use concentration when solution volume is essentially constant and mixing is uniform.
Use amount when you track total moles directly (for example, from a mass change or a total-mole measurement).
In all cases, a reported rate should clearly indicate:
the species being monitored (reactant or product),
whether the rate is average (over an interval) or instantaneous (at a time),
the units and time scale used.
FAQ
Use an amount-based rate (e.g. $\mathrm{mol,s^{-1}}$) or correct concentrations using the measured volume at each time.
If only concentration data are available, note that apparent rate may reflect both reaction progress and dilution/contraction.
Yes. For an ideal gas at constant volume, pressure is proportional to concentration.
A rate can be written as $\Delta P/\Delta t$ with units like $\mathrm{kPa,s^{-1}}$, provided temperature is controlled.
As reactants are used up, fewer reactive collisions occur per unit time, so the concentration changes more slowly.
Graphically, this means the slope magnitude approaches zero.
Delay in starting the timer relative to mixing
Incomplete mixing (non-uniform concentration)
Sensor lag or sampling interval too large
These especially affect early-time (fast) behaviour.
Rate can be reported with a sign (negative for reactant consumption, positive for product formation).
More often, chemists report a positive magnitude and specify “rate of disappearance” or “rate of formation” to avoid ambiguity.
Practice Questions
Q1 (2 marks) Define reaction rate and state a suitable unit when rate is expressed using concentration.
Defines rate as change in concentration (or amount) per unit time (1)
Gives a correct unit such as / (1)
Q2 (5 marks) A student plots concentration of reactant against time for a reaction. (a) Explain what feature of the graph represents the instantaneous rate at time and how it is obtained. (3 marks) (b) For reactant , explain why the expression is often used instead of . (2 marks)
Instantaneous rate corresponds to the gradient (slope) of the curve at that time (1)
Obtained by drawing a tangent at time (or considering an extremely small time interval about ) (1)
Steeper gradient implies larger magnitude of rate (faster change) (1) (b)
decreases so is negative for consumption (1)
Negative sign makes the rate of disappearance a positive value by convention (1)

