AP Syllabus focus: ‘Rates of change for reactants and products are linked by the stoichiometric coefficients in the balanced chemical equation.’
Reaction rates for different species in the same reaction are not independent measurements. Stoichiometry forces their time-dependent concentration changes to occur in fixed ratios set by the balanced chemical equation.
Core idea: one reaction, many linked rates
For a single chemical reaction, you can monitor the rate using any reactant being consumed or any product being formed. Because particles are consumed/produced in stoichiometric proportions, the measured rates differ in magnitude but correspond to the same underlying reaction progress.
Rate of disappearance vs. rate of appearance
Rate of disappearance/appearance: The rate at which a species’ concentration changes with time; negative for reactants (disappearance) and positive for products (appearance).
A concentration that is decreasing has a negative slope on a concentration–time graph, so reactant rates are often written with a leading negative sign to report a positive reaction rate.
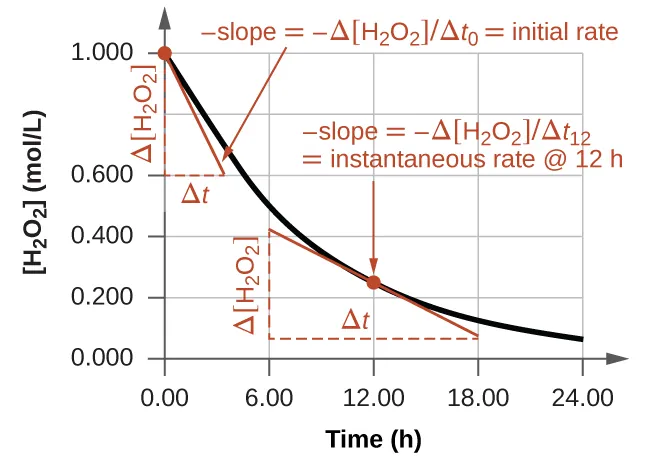
Concentration–time plot for a reactant with tangent lines drawn to show how the instantaneous rate is obtained from the slope at a specific time. Because the reactant concentration decreases, the slope is negative, and the rate is commonly reported as to give a positive value. Source
Using stoichiometric coefficients to relate rates
Consider the general balanced reaction:
The coefficients determine how much of each species is consumed or formed per “reaction event.”
Therefore, the time rates of change must scale inversely with these coefficients.
= single consistent rate for the overall reaction, typically in
= concentration of species in
= time in
This relationship means any one species can be used to compute the same reaction rate, as long as you divide by its stoichiometric coefficient and use the correct sign.
Interpreting the signs (what the calculus means)
For reactants, , so is positive.
For products, , so is positive.
The reaction rate is defined to be positive for the forward reaction when written as above.
Practical consequences for graphs and data
Comparing slopes on concentration–time plots
If you plot concentration vs. time for multiple species in the same run:
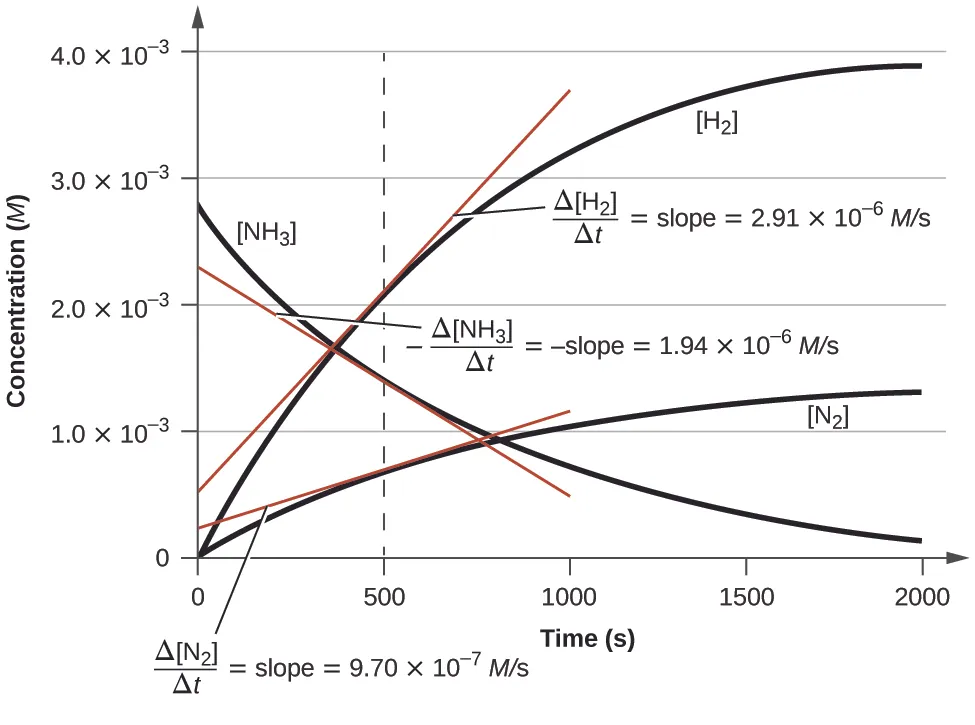
Concentration–time curves for reactant and products in a single reaction, with tangents drawn at the same time point to compare instantaneous slopes. The different slope magnitudes reflect the stoichiometric factors: species produced/consumed in larger coefficients change faster in concentration per unit time, even though they correspond to the same underlying reaction progress. Source
Slopes are steeper (larger magnitude) for species with larger stoichiometric coefficients, because their concentrations change faster per unit time.
The ratio of instantaneous slopes matches the coefficient ratio (with sign):
for (holding the same reaction direction).
Converting a measured rate for one species into another
You often measure one conveniently tracked species (via colorimetry, gas pressure, titration, etc.) and then convert:
From a reactant disappearance rate to a product appearance rate (or vice versa)
From one reactant’s rate to another reactant’s rate
The conversion is purely stoichiometric:
Multiply by the appropriate coefficient ratio
Keep track of sign conventions (reactants negative, products positive) unless reporting the single positive reaction rate.
Common AP Chemistry pitfalls to avoid
Using stoichiometric coefficients from an unbalanced equation (rates must be linked by the balanced equation).
Forgetting the coefficient scaling (e.g., assuming even when coefficients differ).
Mixing “rate of change of a species” with the “reaction rate” without dividing by the coefficient.
Dropping the negative sign for reactants when the question expects a positive overall reaction rate.
FAQ
Because the raw concentration change depends on how many moles of that species participate per reaction event. Dividing normalises different species to a single common progress rate.
Yes (for that balanced equation direction): $ \text{rate} = \frac{d[\text{product}]}{dt} $ when the product coefficient is 1.
The coefficients stay the same but reactants/products swap roles, so the sign pattern changes. The defined “forward” reaction rate corresponds to the written direction.
Yes, after converting the measured change to a proportional amount (e.g., using $P \propto n$ at constant $T,V$). The coefficient ratios still link the species’ changes.
Both obey the same stoichiometric scaling. Average rates use $\Delta[\ ]/\Delta t$ over an interval; instantaneous rates use slopes $d[\ ]/dt$ at a specific time.
Practice Questions
(2 marks) For , write an expression relating the reaction rate to and to .
1 mark:
1 mark:
(5 marks) For , the instantaneous rate of disappearance of A is .
(a) Express the reaction rate in terms of . (2 marks)
(b) Express in terms of . (3 marks)
(a) 1 mark:
(a) 1 mark: equivalently
(b) 1 mark:
(b) 1 mark: rearrangement to
(b) 1 mark: correct sign (shows if A is disappearing)

