AP Syllabus focus: ‘Reaction rate depends on reactant concentrations, temperature, surface area, catalysts, and other environmental conditions.’
Reaction rates vary because particles must collide to react. Any factor that changes collision frequency or the fraction of collisions that are successful will change how quickly reactants are converted to products.
Core idea: collisions control rate
A reaction speeds up when reactant particles:
collide more often (higher collision frequency), and/or
collide more effectively (sufficient energy and proper positioning to rearrange bonds)
Concentration (and pressure for gases)
Higher reactant concentration generally increases rate because more particles occupy a given volume, producing more collisions per unit time.
In solutions, increasing concentration usually increases collision frequency.
For gases, increasing pressure (or decreasing volume) increases rate by increasing the gas concentration.
What you can observe experimentally:
When a reactant is diluted, the reaction often slows noticeably.
Gas-phase reactions often speed up when the container is compressed.
Temperature
Increasing temperature increases reaction rate because particles move faster and collide more frequently, and a larger fraction of collisions have enough energy to react.
Faster particle motion increases collision frequency.
Higher particle energies increase the fraction of effective collisions, so rate often rises sharply with temperature.
Common lab implication:
A reaction mixture warmed slightly can react much faster than the same mixture at room temperature.
Surface area (especially for solids)
For reactions involving a solid reactant, only particles at the surface can collide with particles in a fluid (solution or gas). Increasing surface area increases the number of available collision sites.
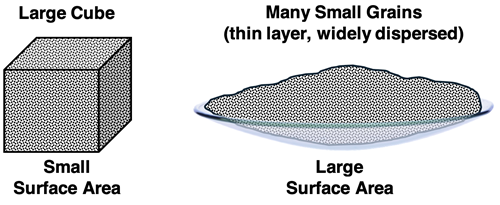
A single large solid piece is contrasted with the same material divided into many smaller grains. Breaking a solid into smaller pieces increases total exposed surface area, increasing the number of interfacial sites where reactant particles can collide per unit time. Source
Powdered solids react faster than large chunks of the same substance.
Stirring can continually expose “fresh” surface and improve contact between phases.
Key idea:
More surface area → more interfacial collisions per unit time → faster rate.
Catalysts
A catalyst increases rate by changing the reaction pathway so a greater fraction of collisions leads to product formation, while the catalyst itself is not used up overall.
Catalyst: A substance that increases reaction rate without being consumed overall; it participates in the process but is regenerated by the end.
Practical consequences:
A catalyst can greatly increase rate even at the same concentration and temperature.
Catalysts are highly specific: a catalyst effective for one reaction may not affect another.
Other environmental conditions
Beyond concentration, temperature, surface area, and catalysts, several conditions can influence collision frequency, contact, or particle reactivity.
Mixing/agitation: Improves contact between reactants and reduces local depletion of reactants near surfaces, often increasing observed rate.
Solvent and phase: Changing solvent can alter how often reactants encounter each other and how stabilised they are before reacting (for example, by changing how well ions are separated).
pH (acidic/basic conditions): Can change the form of reactants (protonated vs deprotonated), changing how readily they collide and react.
Light (photochemical conditions): For light-sensitive reactions, higher light intensity can increase the formation of reactive species, increasing rate.
Ionic strength (in solution): The presence of other ions can influence attractions/repulsions between ionic reactants, affecting how often productive encounters occur.
FAQ
Powdering increases the surface-area-to-volume ratio.
More surface sites are available for collisions with solution/gas particles, so more reactions can occur at once.
A high collision frequency does not guarantee successful collisions.
If few collisions are effective (due to the pathway or reactant structure), the reaction can remain slow.
Stirring reduces concentration gradients near surfaces.
It refreshes reactants at the interface and removes products, increasing effective contact and the observed initial rate.
Gases are highly compressible, so pressure changes significantly change gas concentration.
Liquids/solids compress very little, so concentration changes are minimal.
Yes.
Different solvents can change how often reactants meet and how reactive they are (e.g. by stabilising ions differently), altering the fraction of productive encounters.
Practice Questions
(2 marks) State two factors that can increase the rate of a reaction and, for each, give a brief reason.
1 mark: any correct factor (e.g. higher concentration/pressure, higher temperature, greater surface area, catalyst).
1 mark: correct linked reason (more frequent collisions or greater fraction of successful collisions).
(5 marks) A student reacts calcium carbonate with hydrochloric acid and collects . Describe how the student could change conditions to increase the initial rate, and explain each change in terms of particle collisions.
1 mark: increase acid concentration (or decrease dilution).
1 mark: explanation: more particles per unit volume more collisions.
1 mark: increase temperature.
1 mark: explanation: faster particles and/or higher fraction of successful collisions.
1 mark: increase surface area of (crushed/powdered) and explanation: more exposed sites more collisions at the surface.

